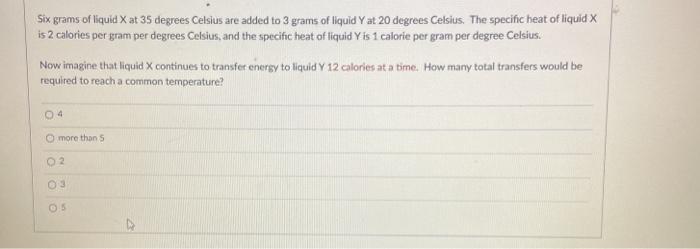
Six grams of liquid X at 35 degrees Celsius are added to 3 grams of liquid...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Six grams of liquid X at 35 degrees Celsius are added to 3 grams of liquid Y at 20 degrees Celsius. The specific heat of liquid X is 2 calories per gram per degrees Celsius, and the specific heat of liquid Y is 1 calorie per gram per degree Celsius. Now imagine that liquid X continues to transfer energy to liquid Y 12 calories at a time. How many total transfers would be required to reach a common temperature? 04 O more than S 02 Six grams of liquid X at 35 degrees Celsius are added to 3 grams of liquid Y at 20 degrees Celsius. The specific heat of liquid X is 2 calories per gram per degrees Celsius, and the specific heat of liquid Y is 1 calorie per gram per degree Celsius. Now imagine that liquid X continues to transfer energy to liquid Y 12 calories at a time. How many total transfers would be required to reach a common temperature? 04 O more than S 02
Expert Answer:
Answer rating: 100% (QA)
Solution Let the common temperature be t Thus Heat lost ... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
The specific heat of a material is given in a strange unit to be c = 3.60 kJ/kg = F. The specific heat of this material in the SI units of kJ/kg C is (a) 2.00 kJ/kg C (d) 4.80 kJ/kg C (b) 3.20...
-
The specific heat capacity of silver is 0.24 JoC-1g-1. a. Calculate the energy required to raise the temperature of 150.0 g Ag from 273 K to 298 K. b. Calculate the energy required to raise the...
-
The specific heat capacity of ice is about 0.5 cal/gC. Supposing that it remains at that value all the way to absolute zero, calculate the number of calories it would take to change a 1-g ice cube at...
-
Selected answer is incorrect During substantive procedures, performing analytical procedures satisfies which primary audit objective: Cutoff Accuracy Existence Completeness 2 answers
-
What is the utilitarian idea of fairness and what is wrong with it?
-
Find the most current 10K (SEC annual report) and 10Q (SEC quarterly report) for Starbucks, either through the companys Web site or the SEC Web site. Find the auditors report for the annual financial...
-
Bernardino Santos-Rodiguez was piloting a boat owned by his friend, Raul Viera-Torres, in waters near Guayama, Puerto Rico. The boat was equipped with a hydraulic steering system manufactured by...
-
KC Corporation manufactures an air-freshening device called GoodAir, which it sells to six merchandising firms. The list price of a GoodAir is $ 30, and the full manufacturing costs are $ 18....
-
Show that the two drawings represent the same graph by labeling the vertices and edges of the right-hand drawing to correspond to those of the left-hand drawing. 5 el 06 Veq 5 e2 V2 ez 3 U5 (a) es 1...
-
The Farr Quality Bathroom Tile Company has developed the following standards for one of its products: The company produced 1,200 units and sold all of the units at the budgeted sales price of $85....
-
A plane wave (em) is bavelling along the Positive z-direction. The maximum electric field along the x-direction is lov/m. The approximate maxinuum values of power per unit area & magnetic induction...
-
On October 31, 2015, Pete's Print Shop purchased a copy machine for $27,600. Pete's Print Shop expects the machine to last for three years and to have a residual value of $1,500. Compute depreciation...
-
Fifteen years ago, Hailey invested $5,000 and locked in an annual interest rate of 6 percent for 30 years ( ending 15 years from now ). Aidan can make a 15-year investment today and lock in an...
-
1. Sales Revenue: a. The price is $10 and the volume sold was 345,000 units. What is sales revenue? b. Sales revenue is $55,110 and the price is $11. What is the volume sold? c. The volume sold was...
-
Information about the ending inventories of Charleston Chair Company, who applies the LIFO method, is shown below: Current Normal Replacement Selling Cost of Profit Year Cost Cost Price Completion...
-
A pension fund manager is holding a 10-year 10% coupon bond in the funds portfolio, and the interest rate is currently 10%. What loss would the fund be exposed to if the interest rate rises to 11%...
-
Based upon the analyses, now explain what you will do in Period-1, for example: In order to increase awareness of SYPR among Professionals I plan to ............ and reallocate the budget...
-
Fahrad Inc. sells all of its product on account. Fahrad has the following accounts receivable payment experience: Percent paid in the month of sale .........10 Percent paid in the month after the...
-
The heat capacity of a bomb calorimeter was determined by burning 6.79 g of methane (energy of combustion = 802 kJ/ mol CH4) in the bomb. The temperature changed by 10.8oC. a. What is the heat...
-
Consider the following gas samples: 4.0 g of hydrogen gas, 4.0 g of helium gas, 1.0 mole of fluorine gas, 44.0 g of carbon dioxide gas, and 146 g of sulfur hexafluoride gas. Arrange the gas samples...
-
Consider the following reaction at some temperature: H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0 Some molecules of H2O and CO are placed in a 1.0- L container as shown below. When equilibrium is reached,...
-
Consider the electrical circuit shown below where \(v_{i}(t)=\beta\), for \(t \geq 0\). Find the dynamic system response \(v_{o}(t)\). VC C ww R V;(t) R ww R3 A First-Order Electrical System Vo (t) +
-
Find the dynamic system response for the spring mass damping system shown in Figure 3.2, where \(f(t)=\beta, x(0)=0\) (i.e. there is no initial energy stored). b f(t) k Figure 3.2 A first-order...
-
Consider a first-order system with a forcing function \(f(t)\) such that input-output differential equation is given by \[\begin{equation*}\dot{y}+3 y=f(t) \text {, } \tag{3.7}\end{equation*}\] where...
Financial And Managerial Accounting For Undergraduates 1st Edition - ISBN: 9781618532763 - Free Book

Study smarter with the SolutionInn App


