During the titration process if a drop of standardized NaOH adheres to the inside of the...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
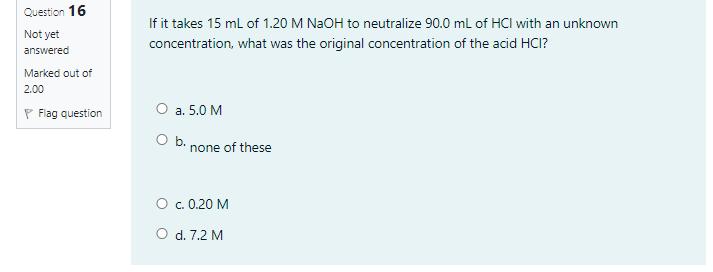
During the titration process if a drop of standardized NaOH adheres to the inside of the titrand container and is never washed down into the solution. The percentage of acetic acid in vinegar sample determined would O a. not affected O b. decrease O c. increase Question 16 If it takes 15 ml of 1.20 M NAOH to neutralize 90.0 ml of HCI with an unknown Not yet concentration, what was the original concentration of the acid HCI? answered Marked out of 2.00 P Flag question O a. 5.0 M O b. none of these O c. 0.20 M O d. 7.2 M During the titration process if a drop of standardized NaOH adheres to the inside of the titrand container and is never washed down into the solution. The percentage of acetic acid in vinegar sample determined would O a. not affected O b. decrease O c. increase Question 16 If it takes 15 ml of 1.20 M NAOH to neutralize 90.0 ml of HCI with an unknown Not yet concentration, what was the original concentration of the acid HCI? answered Marked out of 2.00 P Flag question O a. 5.0 M O b. none of these O c. 0.20 M O d. 7.2 M
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Acetic acid in vinegar results from the bacterial oxidation of ethanol. C2H5OH(l) + O2(g) CH3COOH(l) + H2O(l) What is So for this reaction? Use standard entropy values. (See Appendix C for data.)
-
If a drop of water is placed on a slide and examined under a microscope, the number x of a specific type of bacteria present has been found to have a Poisson probability distribution. Suppose the...
-
(a) If a drop of liquid has capacitance 1.00 pF, what is its radius? (b) If another drop has radius 2.00 mm, what is its capacitance? (c) What is the charge on the smaller drop if its potential is...
-
The slurry of Prob. 29.3 is to be filtered in a press having a total area of 8 m 2 and operated at a constant pressure drop of 2 atm. The frames are 36 mm thick. Assume that the filter medium...
-
Franchise headquarters claims it randomly selected the local franchises that received surprise visits during the past year. The sequence of visits is as shown below for male (M) versus female (F)...
-
Read the examples of cost overruns in What Went Wrong? feature in this chapter. Find at least two articles related to cost overruns for hosting Olympic games. Summarize the reasons for the overruns....
-
On June 15, 2010, Baileys Department Store purchased $4,300 of inventory on account from one of its suppliers. The terms were 3/15, n/45, FOB shipping point. On June 18 Baileys Department Store paid...
-
The following data appeared in the accounting records of Royale Manufacturing Company, which uses an average cost production system: Started in process . . . . . . . . . . . . . . . . . . . . . . . ....
-
An Australian commercial bank is looking to issue $1 billion of bonds into the wholesale funding markets to fund its ever-increasing demand for credit growth which is fuelling yet another house price...
-
In Exercise 3.108 on page 215, we see that the home team was victorious in 70 games out of a sample of 120 games in the FA premier league, a football (soccer) league in Great Britain. We wish to...
-
Write a Research Proposal on ESG Integration in the Banking: How the Banking Sector will adapt in the Era of Global Volatility?
-
One of the loans included in Sloppy Mortgage Corporations loans at January 1, 2006, was a pay-option ARM loan of \($900,000\) taken out on January 1, 2006, when the value of the house was...
-
The continued acceleration of merger and acquisition (M&A) activity remains strong. As companies find it more challenging to increase revenue growth from within their organizations, big mergers...
-
Which of the following is a signal that a company may be using SPEs to overstate its earnings or understate its debt? (a) When references in the notes to the financial statements regarding...
-
On your own or with a classmate, choose one of the following products and create an advertisement that illustrates how your company creates time, place, and form utility in its delivery of the...
-
The Secondary Mortgage Market Enhancement Act of 1984: (a) Allowed the GSEs (Fannie Mae and Freddie Mac) to sell MBSs (mortgagebacked securities) backed by FHA and VA loans. (b) Allowed institutional...
-
When would a preferred stock with a stated maturity most likely see its largest decline in price? Select answer from the options below Never, as a stated maturity date for preferred stock is not...
-
Suppose the market is semistrong form efficient. Can you expect to earn excess returns if you make trades based on? a. Your brokers information about record earnings for a stock? b. Rumors about a...
-
Acetic acid, CH3COOH, freezes at 16.6oC. The heat of fusion, Hfus, is 69.0 J/g. What is the change of entropy, S, when 1 mol of liquid acetic acid freezes to the solid?
-
It is found that a gas undergoes a zero-order decomposition reaction in the presence of a nickel catalyst. If the rate constant for this reaction is 8.1 102 mol/(Ls), how long will it take for the...
-
A voltaic cell consists of a silversilver ion half-cell and a nickelnickel(II) ion half-cell. Silver ion is reduced during operation of the cell. Sketch the cell, labeling the anode and cathode and...
-
If debentures are included as part of capital employed, what figure is used for return? How and why does it differ from the return figure used for calculating return on ordinary shareholders capital...
-
What information (in addition to the current ratio, quick ratio, capital gearing ratio and interest times cover) would you want to assess a companys solvency?
-
How could a company increase its profitability when sales are falling (assuming that it is unable to increase sales)?

Study smarter with the SolutionInn App



