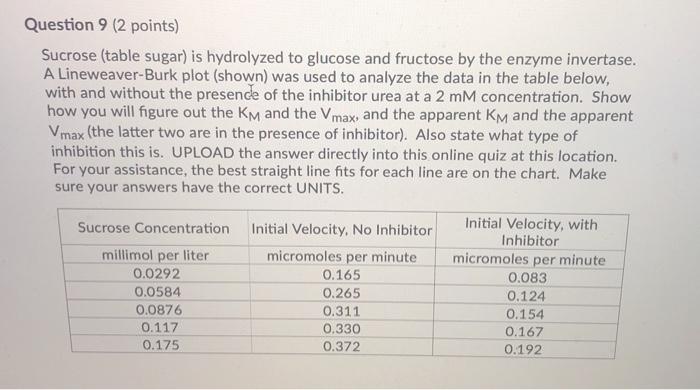
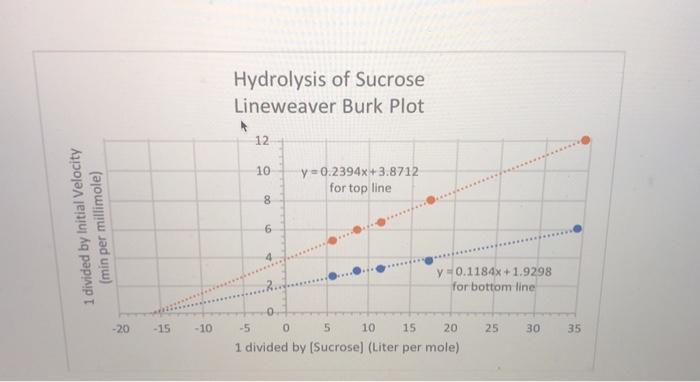
Question 9 (2 points) Sucrose (table sugar) is hydrolyzed to glucose and fructose by the enzyme...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Question 9 (2 points) Sucrose (table sugar) is hydrolyzed to glucose and fructose by the enzyme invertase. A Lineweaver-Burk plot (shown) was used to analyze the data in the table below, with and without the presence of the inhibitor urea at a 2 mM concentration. Show how you will figure out the KM and the Vmax, and the apparent KM and the apparent Vmax (the latter two are in the presence of inhibitor). Also state what type of inhibition this is. UPLOAD the answer directly into this online quiz at this location. For your assistance, the best straight line fits for each line are on the chart. Make sure your answers have the correct UNITS. Sucrose Concentration millimol per liter 0.0292 0.0584 0.0876 0.117 0.175 Initial Velocity, No Inhibitor micromoles per minute 0.165 0.265 0.311 0.330 0.372 Initial Velocity, with Inhibitor micromoles per minute 0.083 0.124 0.154 0.167 0.192 1 divided by Initial Velocity (min per millimole) -20 -15 -10 Hydrolysis of Sucrose Lineweaver Burk Plot 12 10 8 6 y=0.2394x+3.8712 for top line y 0.1184x+1.9298 for bottom line -5 0 5 10 15 20 1 divided by [Sucrose] (Liter per mole). 25 30 35 Question 9 (2 points) Sucrose (table sugar) is hydrolyzed to glucose and fructose by the enzyme invertase. A Lineweaver-Burk plot (shown) was used to analyze the data in the table below, with and without the presence of the inhibitor urea at a 2 mM concentration. Show how you will figure out the KM and the Vmax, and the apparent KM and the apparent Vmax (the latter two are in the presence of inhibitor). Also state what type of inhibition this is. UPLOAD the answer directly into this online quiz at this location. For your assistance, the best straight line fits for each line are on the chart. Make sure your answers have the correct UNITS. Sucrose Concentration millimol per liter 0.0292 0.0584 0.0876 0.117 0.175 Initial Velocity, No Inhibitor micromoles per minute 0.165 0.265 0.311 0.330 0.372 Initial Velocity, with Inhibitor micromoles per minute 0.083 0.124 0.154 0.167 0.192 1 divided by Initial Velocity (min per millimole) -20 -15 -10 Hydrolysis of Sucrose Lineweaver Burk Plot 12 10 8 6 y=0.2394x+3.8712 for top line y 0.1184x+1.9298 for bottom line -5 0 5 10 15 20 1 divided by [Sucrose] (Liter per mole). 25 30 35
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Sucrose is readily hydrolyzed to glucose and fructose in acidic solution. The hydrolysis is often monitored by measuring the angle of rotation of plane polarized light passing through the solution....
-
Table 11* lists data for the enzyme invertase treated with the substrate sucrose. We want to model these data with a Michaelis-Menten function. (A) Plot the points in Table 11 on graph paper and...
-
Glucose and fructose are simple sugars with the molecular formula C6HI206 Sucrose, or table sugar, is a complex sugar with molecular formula C12H220I1 that consists of a glucose unit covalently bound...
-
In evaluating employee performance and determining salary raises, a company uses a large number of subjective and objective criteria gathered from numerous evaluators that are then combined together...
-
Average annual interest rates (banks prime lending) in the United States from 1966 through 2009 are shown in the following time series graph. a) What components do you see in this series? Heres an...
-
1. Describe Burberry's business-level strategy. 2. How is Burberry managing its relationship with customers? 3. Is the differentiation strategy appropriate for Burberry? Why or why not? Now or in the...
-
Whistle-blowers often question themselves as to whether they are doing the right thing. Terms such as snitch and tattletale are often associated with an individual who exposes wrongdoing. Do you...
-
Jerry and Lois Gower own and operate the Gower Painting Co. The couple is insured under a Homeowners 3 policy with no special endorsements. The policy has a $100,000 per occurrence limit for personal...
-
Last year a company had sales of $560,000, operating costs of $336,000, and year-end assets of $725,000. The debt-to-total-assets ratio was 25%, the interest rate on the debt was 4%, and the tax rate...
-
Butler, Inc., operates at capacity and makes wooden playground equipment. Although Butlers swing sets and play forts are a matching set, they are sold individually and so the sales mix is not 1:1....
-
The Earth orbit the Sun in 365 days, and the distance betweenthe Earth and the Sun is 149.6 million km. Given the orbital radiusof a planet is 57.9 million km, what is its predicted orbitalperiod in...
-
GOM240-STRATEGIC MANAGEMENT OF CUSTOMER RELATIONSHIPS ASSIGNMENT 3: B2B PARTNERSHIPS PITCH DECK Grade Value: BACKGROUND / DESCRIPTION Business-to-business (B2B) partnerships occur when 2 or more...
-
Consider a project with the following data: Annual fixed costs = $225,000. Price is $45/unit and variable costs are $21/unit. The project will last 5 years and is depreciated on a straight-line basis...
-
Company A is a publicly trading firm and wants to acquire company B (company B is not in the same line of business and is a private firm). Company A has 80,000 shares outstanding and Company C, a...
-
Case assignment in this module consists of two parts: Part A: After you've read through the articles and related materials, think about their content carefully and compose a short (3- to 4-page)...
-
Bond A has a coupon rate of 7.52 percent, a yield-to-maturity of 11.94 percent, and a face value of 1,000 dollars; matures in 14 years; and pays coupons annually with the next coupon expected in 1...
-
For this sample data of number of hours per week spent on homework by 19 students: 20, 16, 20, 9, 16, 25, 16, 10, 16, 8, 10, 61, 21, 26, 16, 16, 11, 20, 11 . What is the sample mean? What is the...
-
Show that gj concave AHUCQ Abadie For nonnegative variables, we have the following corollary.
-
One of the principal methods for obtaining the electronic spectra of unstable radicals is to study the spectra of comets, which are almost entirely due to radicals. Many radical spectra have been...
-
From data in Table 18.1 calculate the molar polarization, relative permittivity, and refractive index of methanol at 20e. Its density at that temperature is 0.7914 g cm-1,
-
A gas mixture consists of 320 mg of methane, 175 mg of argon, and 225 mg of neon. The partial pressure of neon at 300 K is 8.87 kPa. Calculate (a) The volume and (b) The total pressure of the mixture.
-
Refer to the balance sheet data given in Exercise E13-3B. a. Prepare common-size balance sheets for each year (use total assets as the base amount for computing percentages). b. Compare the...
-
Purple Company has a current ratio of 2.2 on December 31. On that date its current assets are as follows: Purple Company's current liabilities at the beginning of the year were \(\$ 195,000\) and...
-
The following information is available for Virginia Company: Calculate the following ratios for 2019: a. Gross profit percentage b. Return on sales c. Asset turnover d. Return on assets e. Return on...

Study smarter with the SolutionInn App