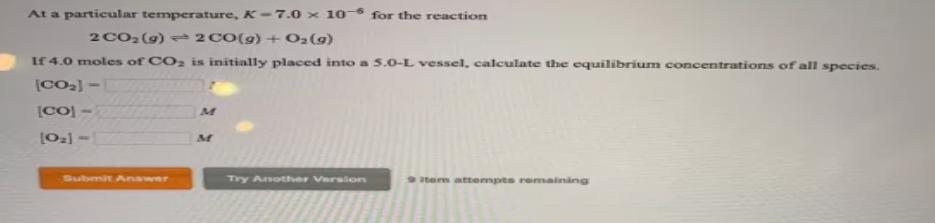
At a particular temperature, K-7.0 x 10- for the reaction 2 CO2(9) 2 CO(g) + O2(9)...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
At a particular temperature, K-7.0 x 10- for the reaction 2 CO2(9) 2 CO(g) + O2(9) If 4.0 moles of CO2 is initially placed into a 5.0-L vessel, calculate the equilibrium concentrations of all species. [CO) - [021 - Submit Anawer Try Anothher Versiors tem attempts remaining At a particular temperature, K-7.0 x 10- for the reaction 2 CO2(9) 2 CO(g) + O2(9) If 4.0 moles of CO2 is initially placed into a 5.0-L vessel, calculate the equilibrium concentrations of all species. [CO) - [021 - Submit Anawer Try Anothher Versiors tem attempts remaining
Expert Answer:
Answer rating: 100% (QA)
Eablewin 2C0cg 2004 Ozca Givew Ozcg 4 mole of Cor in 5L Vessel i K 70x106 So conceutratio... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
For the reaction 2 C6HsCOOH(s) + 13 02(g) 12 CO2 (g) + 6 H20 (g), -Uo= -772.7 k) mol-1 at 298 K. Calculate -1Ho.
-
Calculate the half-life, in years, for the reaction 2 X Y when the starting concentration of X is 6 M and the rate constant is 3.6 10-3 M-1 s-1.
-
The following data were obtained for the reaction 2 ICl ( g ) + H 2 ( g ) I 2 ( g ) + 2 HCl ( g ) Experiment [ICl] 0 (M) [H 2 ] 0 (M) Initial Rate (M/s) 1 0.0015 0.0015 3.7 x 10 -7 2 0.0030 0.0015...
-
A car is randomly selected at a traffic safety checkpoint, and the car has 6 cylinders. Determine whether the given values are from a discrete or continuous data set.
-
The personnel director for a small manufacturing company has collected the data found in the file Dat9-19.xls on your data disk describing the salary (Y) earned by each machinist in the factory along...
-
Following graduation, you meet Mr. Big at a country club. Mr. Big is a venture capitalist who has a sizable stock portfolio concentrated in a single firm, Blue Hat. After several weeks of golfing...
-
There is a route through the city of Konigsberg that a person may pass over each bridge exactly once and return to the starting point. Determine whether each statement is always true, sometimes true,...
-
Clonex Labs, Inc., uses a process costing system. The following data are available for one department for October: The department started 175,000 units into production during the month and...
-
Selected accounts from Westeros Company's adjusted trial balance for the year ended December 3 1 follow. \ table [ [ Sales , $ 6 2 , 0 0 0 , Depreciation expense,$ 1 3 , 0 0 0
-
What theory explains why in 2008 the US government injected $25bn to rescue The Federal National Mortgage Association (Fannie Mae)? Briefly explain this theory and discuss the impact on any two...
-
What is the relationship between capital flow and currency regime? EXPLAIN.
-
Name four characteristics of a future ATC/ATM system that are necessary to provide capacity to meet the future air trafficdemands,and briefly describe each capability.(with references)
-
After reading Principles of Microeconomics and reflecting on the earlier video (also linked below), discuss how economics impacts you in a personal way. How can knowledge about the economy help you...
-
You must present a logical argument on three questions regarding the issues using what we have learned in the text, including references to cases that pertain to the issues. You next written...
-
Calculate the pH at the following points in the titration of 25.00 ml of 0.250 M HNO 3 with 0.250 M NaOH. Before the addition of any NaOH After the addition of 10.00 ml of 0.250 M NaOH After the...
-
Spicy Salsa Inc. complains to the Federal Trade Commission (FTC) about an ad broadcast by Tangy Taco Sauce Company, Spicys competitor. The FTC investigates and concludes that the ad is deceptive. T
-
(a) As Section 17.3 discusses, high-frequency sound waves exhibit less diffraction than low-frequency sound waves do. However, even high-frequency sound waves exhibit much more diffraction under...
-
Some automobiles and buses have been equipped to burn propane (C3H8) as a fuel. Compare the amount of energy that can be obtained per gram of C3H8(g) with that per gram of gasoline, assuming that...
-
Calculate H for the reaction N2H4(l) + O2(g) N2(g) + 2H2O(l) given the following data: 2NH3(g) + 3N2O(g) 4N2(g) + 3H2O(l) H = 1010.kJ N2O(g) + 3H2(g) N2H4(l) + H2O(l) H = 317 kJ 2NH3(g) + O2(g) ...
-
Given the following two standard reduction potentials, M 3+ + 3e M o = 20.10 V M 2+ + 2e M o = 20.50 V determine for the standard reduction potential of the half reaction M 3+ + e M 2+ (You...
-
Graph specific costs (Learning Objective 1) Graph these cost behavior patterns over a relevant range of 0 to 10,000 units: a. Variable expenses of \(\$ 8\) per unit b. Mixed expenses made up of fixed...
-
Continuation of S6-14: absorption costing (Learning Objective 7) (Appendix) Refer to the Sportade example on pages 332-338 and the data and your answer to S6-14. 1. Prepare the April income statement...
-
Prepare income statement using variable costing (Learning Objective 7) (Appendix) Consider the Sportade example on pages 332-338. Suppose that during April, the company produces 10,000 cases of...

Study smarter with the SolutionInn App


