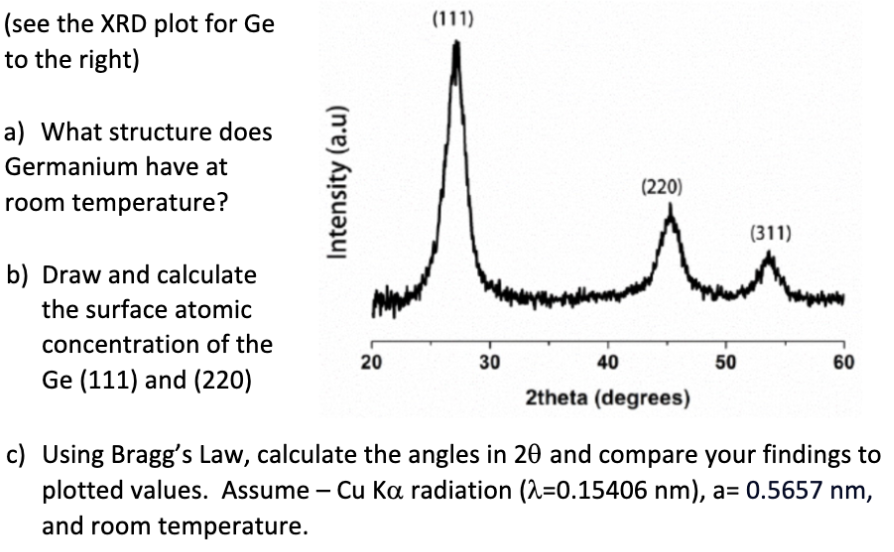
(see the XRD plot for Ge to the right) a) What structure does Germanium have at...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
(see the XRD plot for Ge to the right) a) What structure does Germanium have at room temperature? b) Draw and calculate the surface atomic concentration of the Ge (111) and (220) Intensity (a.u) 20 (111) 30 40 (220) 50 (311) 60 2theta (degrees) c) Using Bragg's Law, calculate the angles in 20 and compare your findings to plotted values. Assume - Cu Ka radiation (2-0.15406 nm), a= 0.5657 nm, and room temperature. (see the XRD plot for Ge to the right) a) What structure does Germanium have at room temperature? b) Draw and calculate the surface atomic concentration of the Ge (111) and (220) Intensity (a.u) 20 (111) 30 40 (220) 50 (311) 60 2theta (degrees) c) Using Bragg's Law, calculate the angles in 20 and compare your findings to plotted values. Assume - Cu Ka radiation (2-0.15406 nm), a= 0.5657 nm, and room temperature. (see the XRD plot for Ge to the right) a) What structure does Germanium have at room temperature? b) Draw and calculate the surface atomic concentration of the Ge (111) and (220) Intensity (a.u) 20 (111) 30 40 (220) 50 (311) 60 2theta (degrees) c) Using Bragg's Law, calculate the angles in 20 and compare your findings to plotted values. Assume - Cu Ka radiation (2-0.15406 nm), a= 0.5657 nm, and room temperature.
Expert Answer:
Answer rating: 100% (QA)
2 m Garmanium 60 has a diamond cubic Cyrstal Structura at rom toppers bre b surface atomic Concentra... View the full answer

Related Book For 

Essentials of Materials Science and Engineering
ISBN: 978-1111576851
3rd edition
Authors: Donald R. Askeland, Wendelin J. Wright
Posted Date:
Students also viewed these physics questions
-
The board of directors of Morales Publishing, Inc., has commissioned a capital structure study. The company has total assets of $40,000,000. It has earnings before interest and taxes of $8,000,000...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Define variable. Also discuss variable initialization.
-
Return to the example of accounting for uncollectibles that begins under the heading "Writing off Uncollectible Accounts". Suppose past experience indicates that the company will fail to collect 2...
-
Give examples of clean sources that can supply energy continuously and examples of others whose output varies with time of day and weather conditions.
-
Tyler Company reported the following costs on its financial statements (in thousands): REQUIRED: Using the reserve disclosure for Tyler Company in problem 13 and the data presented in this problem,...
-
Premier Bank and Trust is considering giving Alou Company a loan. Before doing so, management decides that further discussions with Alou's accountant may be desirable. One area of particular concern...
-
When switch S is thrown to the left in figure, the plates of capacitor 1 acquire a potential differenceVo. Capacitors 2 and 3 are initially uncharged. The switch is now thrown to the right What are...
-
Use PSpice to find V1, V2, and V3 in the network of Fig. 10.128. 8 V. j1012 V, jlOS2 4/0 A
-
An information system(IS)can be defined as a set of interrelated components that collect, process, store, and distribute information to support decision-making and control in an organization. IS...
-
Due to the Covid-19 pandemic, your employer in a meeting with all his staff, announced that he would be deducting staff salary as a way to cope with the company's financial losses. How would you...
-
The recent Netflix show Inventing Anna , based on the convicted con artist Anna Delvey (Sorokin), has been criticized for fictionalizing certain aspects of its story - in the words of its producers,...
-
How is External Business Communication climate created and sustained? How to improve External Business Communications ? What are the indicators or variables (attributes) to measure External Business...
-
Building on the work you have already completed; you are required to identify the correct communication process for two scenarios. You are required to integrate evidence of increased skills and...
-
What sustainability reporting framework focuses on providing guidance to organizations in creating an environmental management system (EMS)? What sustainability reporting framework was a joint...
-
Being a trainee or an employee someday, how will these rights and duties help you in your job?
-
Continuation of Exercise 4-83. (a) What is the probability that the first major crack occurs between 12 and 15 miles of the start of inspection? (b) What is the probability that there are no major...
-
Another form of boron nitride (BN) known as hexagonal boron nitride is used as a solid lubricant. Explain how this may be possible by comparing this situation with that encountered in two forms of...
-
Kevlar (C 14 H 10 N 2 O 2 ) is used in various applications from tires to body armor due to its high strength-to-weight ratio. The polymer is produced from the monomers paraphenylene diamine (C 6 H 8...
-
A polymer bar's dimensions are 1 in. 2 in. 15 in. The polymer has a modulus of elasticity of 600,000 psi. What force is required to stretch the bar elastically from 15 in. to 15.25 in.?
-
Crane l uses 10 kJ of energy to lift a 50 kg box to the roof of a building. Crane 2 uses 20 kl to lift a 100 kg box the same distance. Which crane is more efficient? A. Crane 1 B. Crane 2 C. Both...
-
Christina throws a javelin into the air. As she propels it forward from rest, she does 270 J of work on it. At its highest point, its gravitational potential energy has increased by 70 J. What is the...
-
Two samples of ideal gas, sample 1 and sample 2, have the same thermal energy. Sample l has twice as many atoms as sample 2. What can we say about the temperatures of the two samples? A. T>T B. T = T...

Study smarter with the SolutionInn App


