Shen and Smith [Ind. Eng. Chem. Fundam., 7, 100- 105 (1968)] measured equilibrium-adsorption isotherms at four...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
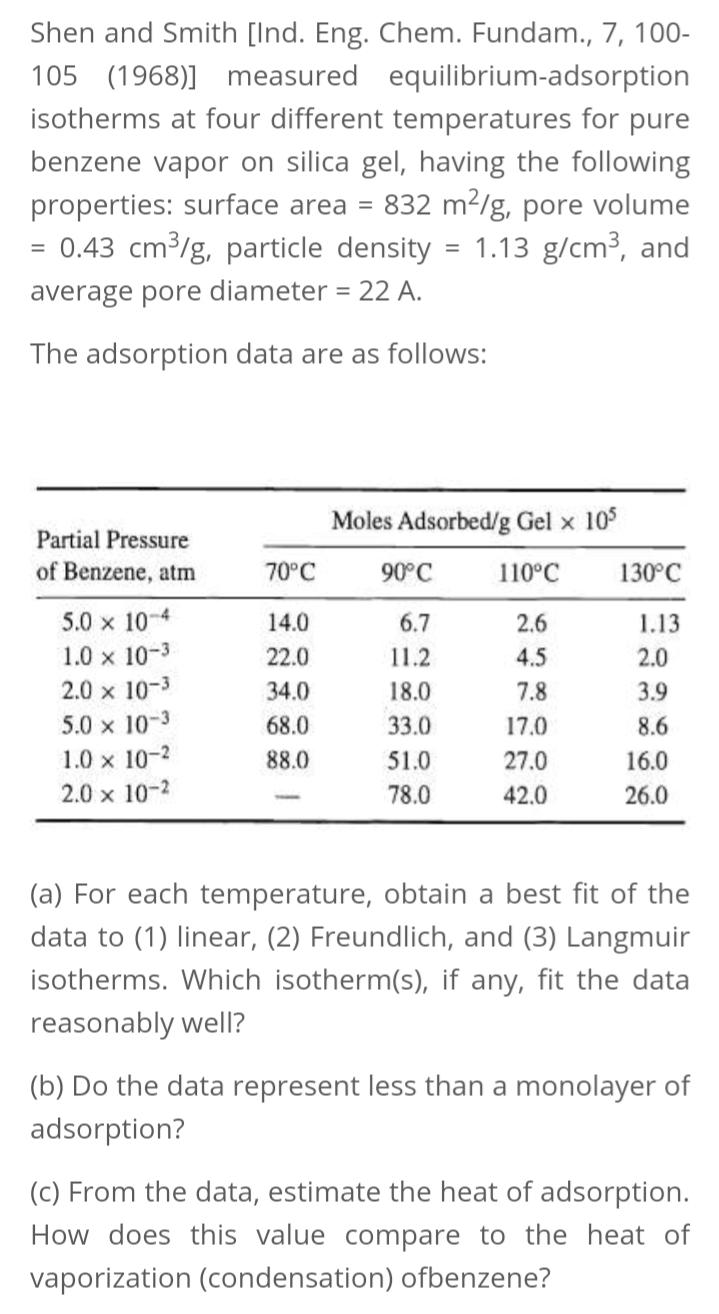
Shen and Smith [Ind. Eng. Chem. Fundam., 7, 100- 105 (1968)] measured equilibrium-adsorption isotherms at four different temperatures for pure benzene vapor on silica gel, having the following properties: surface area = 832 m2/g, pore volume = 0.43 cm³/g, particle density = 1.13 g/cm3, and average pore diameter = 22 A. The adsorption data are as follows: Moles Adsorbed/g Gel x 10 Partial Pressure of Benzene, atm 70°C 90°C 110°C 130°C 5.0 x 10-4 14.0 6.7 2.6 1.13 1.0 x 10-3 2.0 x 10-3 22.0 11.2 4.5 2.0 34.0 18.0 7.8 3.9 5.0 x 10-3 1.0 x 10-2 68.0 33.0 17.0 8.6 88.0 51.0 27.0 16.0 2.0 x 10-2 78.0 42.0 26.0 (a) For each temperature, obtain a best fit of the data to (1) linear, (2) Freundlich, and (3) Langmuir isotherms. Which isotherm(s), if any, fit the data reasonably well? (b) Do the data represent less than a monolayer of adsorption? (c) From the data, estimate the heat of adsorption. How does this value compare to the heat of vaporization (condensation) ofbenzene? Shen and Smith [Ind. Eng. Chem. Fundam., 7, 100- 105 (1968)] measured equilibrium-adsorption isotherms at four different temperatures for pure benzene vapor on silica gel, having the following properties: surface area = 832 m2/g, pore volume = 0.43 cm³/g, particle density = 1.13 g/cm3, and average pore diameter = 22 A. The adsorption data are as follows: Moles Adsorbed/g Gel x 10 Partial Pressure of Benzene, atm 70°C 90°C 110°C 130°C 5.0 x 10-4 14.0 6.7 2.6 1.13 1.0 x 10-3 2.0 x 10-3 22.0 11.2 4.5 2.0 34.0 18.0 7.8 3.9 5.0 x 10-3 1.0 x 10-2 68.0 33.0 17.0 8.6 88.0 51.0 27.0 16.0 2.0 x 10-2 78.0 42.0 26.0 (a) For each temperature, obtain a best fit of the data to (1) linear, (2) Freundlich, and (3) Langmuir isotherms. Which isotherm(s), if any, fit the data reasonably well? (b) Do the data represent less than a monolayer of adsorption? (c) From the data, estimate the heat of adsorption. How does this value compare to the heat of vaporization (condensation) ofbenzene?
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
At Stevens Manufacturing, workers are organized in teams that combine their efforts to produce complex units of a final product. Stevens is using a facility layout known as a(n): consolidated process...
-
Consider the diode bridge rectifier shown below with the voltage regulating capacitor in parallel with the load resistor. 60Hz 120V 10:1 C-470F IL R=1kQ a) For the input signal, sketch waveforms of...
-
The vapor pressure of the benzene at different temperatures is given in the table on the side. Match this Clapeyron equation (linear model). Clapeyron equation: log P = A + B/T Here T is absolute...
-
In view of the information provided below provide suggestions for the efficient management of trade debtors. INFORMATION MANAGEMENT OF TRADE DEBTORS Debtor management is central to the effective cash...
-
A company that produces 8-ounce low-fat yogurt cups wanted to estimate the mean number of calories for such cups. A random sample of 10 such cups produced the following numbers of calories. Construct...
-
Form the scalar products BC and B²C, where B = B², and use the results obtained to prove the identity cos α cos β = 1/2 cos (α + β)...
-
The transactions for Dancing Antz, Inc., for the month of September 2010 have been posted to the accounts as follows: Requirements 1. Prepare the journal entries that served as the sources for the...
-
Lehmans net leverage ratio was not reported within the companys audited financial statements but rather in the companys financial highlights table and MD&A section of its annual report. What...
-
Two years after Martin Marcotte's mother was injured, she passed away as a result of complications arising from her disability. Mrs. Ruth Marcotte left behind a small life insurance policy. The...
-
David R. and Ella M. Cole (ages 39 and 38, respectively) are husband and wife who live at 1820 Elk Avenue, Denver, CO 80202. David is a self-employed consultant, specializing in retail management and...
-
Q. Explain anomalous electronic configuration of copper and chromium Chromium
-
The skier departs the horizontal ski run with a speed of v=110 km/h, soaring into the air. Upon landing, the slope he/she encounters forms a 45 angle with the horizontal plane. Calculate the skier's...
-
A drone has a speedometer and a compass as part of its instrumentation. The speedometer tells you the speed of the drone and the compass tells you the direction the drone moves. While the drone is...
-
My industry is NAIC code is 541213 Tax Preparation in Chicago, Illinois Section A: Business Concept: (50 points) Please describe the purpose of the selected company, including a detailed description...
-
A swimmer competes in a 100 m race. He completes the length of the 50.0 m swimming pool in 39.5 s, and makes the return lap in 42.0 s. (Assume that the swimmer moves in a straight line through his...
-
1. Suppose you measure three independent variables as x = 10 2, y = 7 1, = 40 3, and use these values to compute = + 2/ + sin (4) What should be your answer for and its uncertainty? 2. The Type K...
-
You estimated the single index (market) model for stocks A and B with the following results: Return on Stock A: RA = 0.06 + 0.5RM + CA Return on Stock B: RB = 0.04 + 1.5R + eB Where RM is return on...
-
1. Using the information from Problem 16-4B, prepare a statement of cash flows for Lim Garden Supplies Inc. using the direct method of presenting cash flows from operating activities. 2. How does Lim...
-
Indicate which of the following is independent of the path by which a change occurs: (a) The change in potential energy when a book is transferred from table to shelf (b) The heat evolved when a cube...
-
Explain how a redox reaction involves electrons in the same way that a neutralization reaction involves protons.
-
Look up and compare the normal boiling points and normal melting points of H2O and H2S. (a) Based on these physical properties, which substance has stronger intermolecular forces? What kind of...
-
Estimate the gravitational force and the acceleration due to gravity on a body of \(1.25 \mathrm{~kg}\) mass on the earth's surface. The radius and mass of the earth are \(6370 \mathrm{~km}\) and...
-
A reactor contains a gas mixture of \(25 \mathrm{~kg} \mathrm{NH}_{3}, 15 \mathrm{~kg} \mathrm{CO}\) and \(10 \mathrm{~kg} \mathrm{C}_{2} \mathrm{H}_{2}\). Calculate the total number of moles of the...
-
Estimate the gravitational force on a body of \(1.5 \mathrm{~kg}\) mass on the earth's surface, given that the radius and mass of the earth are \(6000 \mathrm{~km}\) and \(6 \times 10^{24}...

Study smarter with the SolutionInn App



