Show calculation Show calculation Show calculations Show calculations Show calculations of percent error. of molality. of...
Fantastic news! We've Found the answer you've been seeking!
Question:

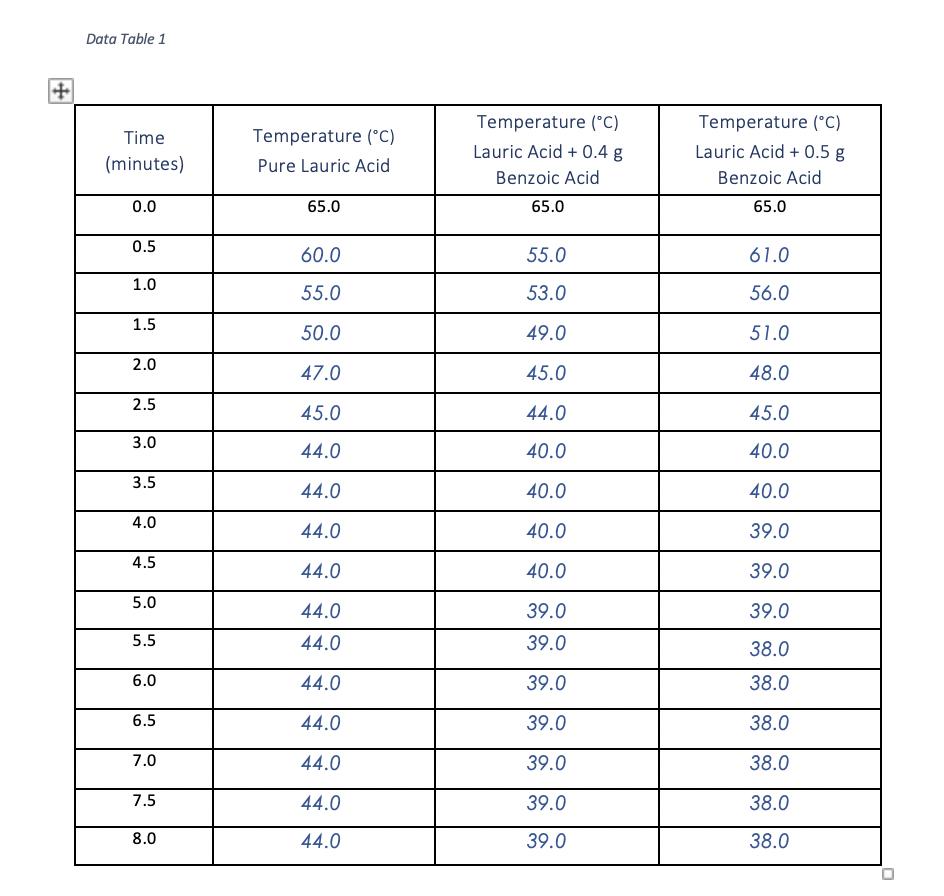
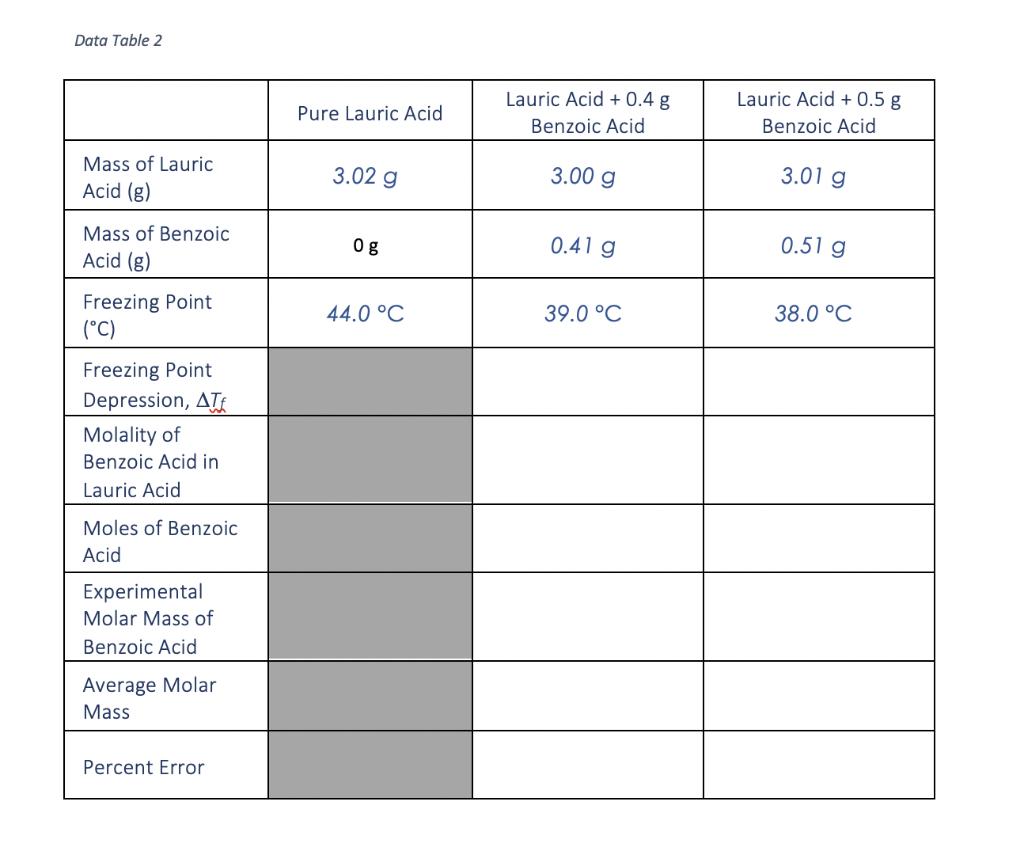
Transcribed Image Text:
Show calculation Show calculation Show calculations Show calculations Show calculations of percent error. of molality. of moles benzoic acid. of molar mass. of average molar mass. Data Table 1 Time (minutes) 0.0 0.5 1.0 1.5 2.0 2.5 3.0 3.5 4.0 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 Temperature (°C) Pure Lauric Acid 65.0 60.0 55.0 50.0 47.0 45.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 Temperature (°C) Lauric Acid + 0.4 g Benzoic Acid 65.0 55.0 53.0 49.0 45.0 44.0 40.0 40.0 40.0 40.0 39.0 39.0 39.0 39.0 39.0 39.0 39.0 Temperature (°C) Lauric Acid + 0.5 g Benzoic Acid 65.0 61.0 56.0 51.0 48.0 45.0 40.0 40.0 39.0 39.0 39.0 38.0 38.0 38.0 38.0 38.0 38.0 U Data Table 2 Mass of Lauric Acid (g) Mass of Benzoic Acid (g) Freezing Point (°C) Freezing Point Depression, AT Molality of Benzoic Acid in Lauric Acid Moles of Benzoic Acid Experimental Molar Mass of Benzoic Acid Average Molar Mass Percent Error Pure Lauric Acid 3.02 g og 44.0 °C Lauric Acid + 0.4 g Benzoic Acid 3.00 g 0.41 g 39.0 °C Lauric Acid + 0.5 g Benzoic Acid 3.01 g 0.51 g 38.0 °C Show calculation Show calculation Show calculations Show calculations Show calculations of percent error. of molality. of moles benzoic acid. of molar mass. of average molar mass. Data Table 1 Time (minutes) 0.0 0.5 1.0 1.5 2.0 2.5 3.0 3.5 4.0 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 Temperature (°C) Pure Lauric Acid 65.0 60.0 55.0 50.0 47.0 45.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 44.0 Temperature (°C) Lauric Acid + 0.4 g Benzoic Acid 65.0 55.0 53.0 49.0 45.0 44.0 40.0 40.0 40.0 40.0 39.0 39.0 39.0 39.0 39.0 39.0 39.0 Temperature (°C) Lauric Acid + 0.5 g Benzoic Acid 65.0 61.0 56.0 51.0 48.0 45.0 40.0 40.0 39.0 39.0 39.0 38.0 38.0 38.0 38.0 38.0 38.0 U Data Table 2 Mass of Lauric Acid (g) Mass of Benzoic Acid (g) Freezing Point (°C) Freezing Point Depression, AT Molality of Benzoic Acid in Lauric Acid Moles of Benzoic Acid Experimental Molar Mass of Benzoic Acid Average Molar Mass Percent Error Pure Lauric Acid 3.02 g og 44.0 °C Lauric Acid + 0.4 g Benzoic Acid 3.00 g 0.41 g 39.0 °C Lauric Acid + 0.5 g Benzoic Acid 3.01 g 0.51 g 38.0 °C
Expert Answer:
Answer rating: 100% (QA)
Calculate the freezing point depression T for each solution T Freezing Point of Pure Lauric Acid Fre... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Posted Date:
Students also viewed these mathematics questions
-
1. What responsibilities might you have for ambulatory (Holter) monitoring? 2. Why is a diary a necessary part of Holter monitoring? 3. What does real-time monitoring mean? 4. Why should the patient...
-
Sometimes during ambulatory monitoring the electrodes may become loose or disconnected, and the patient will need to know how to handle this situation. Check the policy of the facility where you are...
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
The flexural rigidity of a rod-like structure like a microtubule is a measure of its bending stiffness. Flexural rigidity is given by EL, where E is the Young?s modulus and L is the geometrical...
-
National Tennis Racquet Co. produces and sells three models: Required a. What is the weighted average contribution margin per unit? b. Calculate the break-even point in units assuming the current...
-
Moshi Mining is set to open a gold mine in northern Tanzania. The mine will cost 6 million rand to open and will have an economic life of 12 years. It will generate a cash inflow of 1 million rand at...
-
What would not be an abusive earnings management scheme according to the SEC? a. A big bath b. Channel stuffing c. Postponing repairs and maintenance expenses d. Cookie-jar accounting e. a. and c....
-
Adirondack Paper Mills, Inc., operates paper plants in Augusta, Maine, and Tupper Lake, New York. Warehouse facilities are located in Albany, New York, and Portsmouth, New Hampshire. Distributors are...
-
International financial management. Answer the following questions about the capital mobility: A. In your opinion, why does a country like Venezuela impose capital controls? [7 marks] B. Explain the...
-
Prove the following analogs to Stein's Lemma, assuming appropriate conditions on the function g. (a) If X ~ gamma(α, β), then E(g(X)(X-aβ)= βE...
-
Which document authorizes reducing the balance owed by a customer? A. Monthly statement B. All four documents can be used to authorize reducing the balance owed by a customer. C. Sales invoice D....
-
Malware attacks are mainly on PC systems (running Microsoft Windows). What other platforms does Malware attacks?
-
On January 1, 2016, Poplar Company acquired 80% of the common stock of Sequoia Company for $400,000. On this date, Sequoia had total owners' equity of $400,000. The excess of cost over book value was...
-
What you need to produce for the assignment: a three thousand word report including two sections. 1: Website Development In section 1 of report you should demonstrate the following: A clear...
-
By focusing on cleaning up messes after they happen or as they arise, traditional policing tends to fall short when it comes to stopping crime before it occurs. That's wherecommunity-oriented...
-
how Microsoft Windows features could be used in the healthcare industry?
-
The manager of a company's Cash Account sees that there is a deficit balance in the forecasted cash budget. You would likely advise the manager to:
-
Consider the circuit of Fig. 7.97. Find v0 (t) if i(0) = 2 A and v(t) = 0. 1 3 ett)
-
A situation often encountered with thermodynamic data is that different sources may present different correlations for a given quantity. In this case study, for example, two different expressions are...
-
A liquid mixture containing 40.0 wt% n-octane and the balance n-decane flows into a tank mounted on a balance. The mass in kg indicated by the scale is plotted against time. The data fall on a...
-
The composition of a coal is determined by a proximate analysis. The coal is first finely ground and air-dried. Samples of the dried coal are then subjected to several operations, with the sample...
-
Rewrite the solution to Problem 3 and identify the initial value Green's functions. Data from Problem 3 Find product solutions, \(u(x, t)=b(t) \phi(x)\), to the wave equation satisfying the boundary...
-
Established less than 50 years ago, Honda Motor Co. Ltd. of Japan is a leading international manufacturer of automobiles and the largest manufacturer of motorcycles in the world. As a Japanese...
-
Rewrite the solution to Problem 2 and identify the initial value Green's function. Data from Problem 2 Find product solutions, \(u(x, t)=b(t) \phi(x)\), to the heat equation satisfying the boundary...

Study smarter with the SolutionInn App


