Some bath salts are synthetic cathinones designed to circumvent laws, but have unknown properties and side...
Fantastic news! We've Found the answer you've been seeking!
Question:
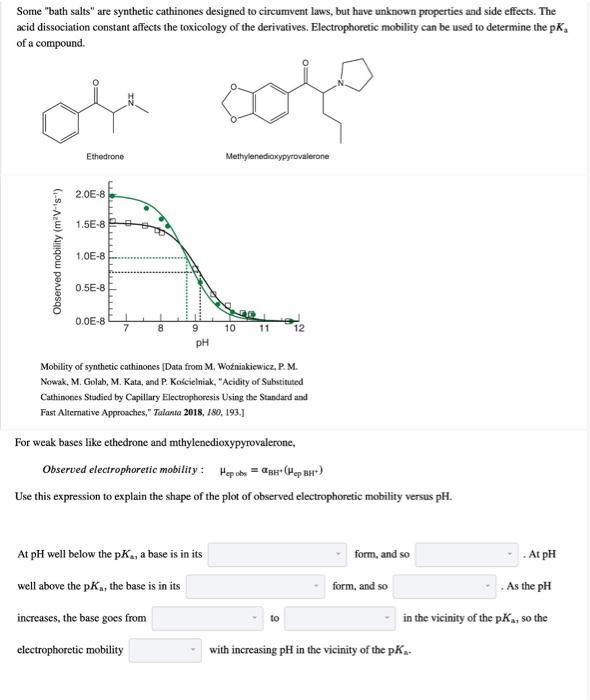

Transcribed Image Text:
Some "bath salts" are synthetic cathinones designed to circumvent laws, but have unknown properties and side effects. The acid dissociation constant affects the toxicology of the derivatives. Electrophoretic mobility can be used to determine the pK, of a compound. Observed mobility (m²V-¹s¹) Ethedrone 2.0E-8 1.5E-8 1.0E-8 0.5E-8 0.0E-8 di 7 8 9 pH electrophoretic mobility Methylenedioxypyrovalerone At pH well below the pK, a base is in its well above the pK₁, the base is in its increases, the base goes from 10 11 Mobility of synthetic cathinones [Data from M. Woźniakiewicz, P. M. Nowak, M. Golah, M. Kata, and P. Kościelniak, "Acidity of Substituted Cathinones Studied by Capillary Electrophoresis Using the Standard and Fast Alternative Approaches," Talania 2018, 180, 193.] For weak bases like ethedrone and mthylenedioxypyrovalerone, Observed electrophoretic mobility: Hep obs= BH (Hep BH) Use this expression to explain the shape of the plot of observed electrophoretic mobility versus pH. 12 to form, and so At pH As the pH in the vicinity of the pK₁, so the form, and so with increasing pH in the vicinity of the pK₂. Electrophoretic mobility is proportional to the charge of the ion and inversely proportional to the size of the ion. Which fully protonated cathinone would be expected to have a greater electrophoretic mobility? methylenedioxypyrovalerone ethedrone Based on the electrophoretic mobility of the fully protonated base (Hep BH-), which curve is for ethedrone and which is for methylenedioxypyrovalerone? the upper curve is methlyenedioxypyrovalerone and the lower curve is ethedrone the upper curve is ethedrone and the lower curve is methlyenedioxypyrovalerone The pK, values for the two cathinones are 8.77 and 9.10. Which acid dissociation constant is for ethedrone? PK₁ = What pH would provide the greatest resolution between the two cathinones? pH <7 pH > 7 OpH=7 Some "bath salts" are synthetic cathinones designed to circumvent laws, but have unknown properties and side effects. The acid dissociation constant affects the toxicology of the derivatives. Electrophoretic mobility can be used to determine the pK, of a compound. Observed mobility (m²V-¹s¹) Ethedrone 2.0E-8 1.5E-8 1.0E-8 0.5E-8 0.0E-8 di 7 8 9 pH electrophoretic mobility Methylenedioxypyrovalerone At pH well below the pK, a base is in its well above the pK₁, the base is in its increases, the base goes from 10 11 Mobility of synthetic cathinones [Data from M. Woźniakiewicz, P. M. Nowak, M. Golah, M. Kata, and P. Kościelniak, "Acidity of Substituted Cathinones Studied by Capillary Electrophoresis Using the Standard and Fast Alternative Approaches," Talania 2018, 180, 193.] For weak bases like ethedrone and mthylenedioxypyrovalerone, Observed electrophoretic mobility: Hep obs= BH (Hep BH) Use this expression to explain the shape of the plot of observed electrophoretic mobility versus pH. 12 to form, and so At pH As the pH in the vicinity of the pK₁, so the form, and so with increasing pH in the vicinity of the pK₂. Electrophoretic mobility is proportional to the charge of the ion and inversely proportional to the size of the ion. Which fully protonated cathinone would be expected to have a greater electrophoretic mobility? methylenedioxypyrovalerone ethedrone Based on the electrophoretic mobility of the fully protonated base (Hep BH-), which curve is for ethedrone and which is for methylenedioxypyrovalerone? the upper curve is methlyenedioxypyrovalerone and the lower curve is ethedrone the upper curve is ethedrone and the lower curve is methlyenedioxypyrovalerone The pK, values for the two cathinones are 8.77 and 9.10. Which acid dissociation constant is for ethedrone? PK₁ = What pH would provide the greatest resolution between the two cathinones? pH <7 pH > 7 OpH=7
Expert Answer:
Answer rating: 100% (QA)
a The plot has a sigmoidal shape like a titration curve So when a BH is equal to 1 full protonated B... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
A get retardation assay can be used to determine if a protein bids to a segment of DNA. When a segment of DNA is bound by a protein. Its mobility will be retarded, and the DNA band will appear higher...
-
The following general equation can be used to determine missing information about balance sheet accounts: Use this equation to determine the missing information in each of the following in dependent...
-
The pulse-echo ultrasonic technique can be used to determine the thickness of a part or structure. By accurately measuring the time it takes for a short ultrasonic pulse to travel through the...
-
Calculate the CGT payable in relation to each of the following disposals, assuming in each case that the annual exemption is fully utilised against other gains, that there are no allowable losses and...
-
Give the proper name for each molecule. a. SeO2 b. Cl2O c. XeF6
-
Marco works three jobs part time to help support his family. He works at two different restaurants as a waiter and as a clerk at a grocery store. Restaurant A paid him \($15,600\) in salary and Marco...
-
A refrigeration system of 15 tons capacity operates on simple vapour compression system cycle using R 22. The evaporator temperature is 5C and condenser temperature is 50C. Calculate (a) the...
-
1. What human resource trends described in this chapter are behind the situation faced by Jill McBride? 2. What advice to McBride would you add, beyond the recommendations given in this case?...
-
2. For Roland Company in Industry F, enter the December 31 adjustments in the following table for prepaid insurance, supplies, accrued wages, accrued revenue, and unearned revenue. Then, complete the...
-
Kofi Allon, who is 32 years old and single, is employed as a technical consultant for a large electronics distributor. He lives at 678 Birch Street, LaMesa, CA 91941. Kofi's Social Security number is...
-
Suppose an auditor is preparing for an audit of the payroll process of an entity an has identified that the entity is labor-intensive an depends largely on hourly compensation. What is the auditor...
-
Your college professors take on what kind of sales role when they select books for their class? a. Order taker b. Order influencer c. Support personnel d. None of the above
-
What is the funds Treynor ratio?
-
CIM refers to using computer software to create two- or three-dimensional models of parts or devices. True or False
-
American Idol judges often have on their desks tall glasses embossed with Coca-Colas logo. This advertising strategy is known as a. product placement. b. viral marketing. c. an infomercial. d. a...
-
What is the settlement method for the futures contract?
-
Suppose that the following equations describes an economy: Consumption function: Investment function: Taxes: Government Spending: C(Y-T) .5(Y-T)+30 I(r)=22-r T=30 G=10 Demand for real money balances:...
-
In a paragraph of approximately 150-200 words, analyze a film or TV/Streaming Show poster of your choosing by focusing on the ways in which representations in the poster are gendered. Include an...
-
Diethylstilbestrol (DES) was given to pregnant women to prevent miscarriage, until it was found that the drug caused cancer in both the mothers and their female children. DES has estradiol activity...
-
An alkyl halide reacts with an alkoxide ion to form a compound whose 1H NMR spectrum is shown here. Identify the alkyl halide and the alkoxide ion. 0 3 4 6 7 8 9 0
-
Which of the double bonds in zingiberene (whose structure is given on the first page of this chapter) is the most reactive in an electrophilic addition reaction?
-
Use the payoff matrix below for the following exercises. The payoff matrix indicates the profit outcome that corresponds to each firms pricing strategy. a. Firms A and B are members of an oligopoly....
-
What is the purpose of a brand name? What would occur if any maker of aspirin could put a Bayer Aspirin label on its product?
-
The South American cocaine industry consists of several families that obtain the raw material, refine it, and distribute it in the United States. There are only about three large families, but there...

Study smarter with the SolutionInn App


