Some chemical compounds are listed in the first column of the table below. Each compound is...
Fantastic news! We've Found the answer you've been seeking!
Question:
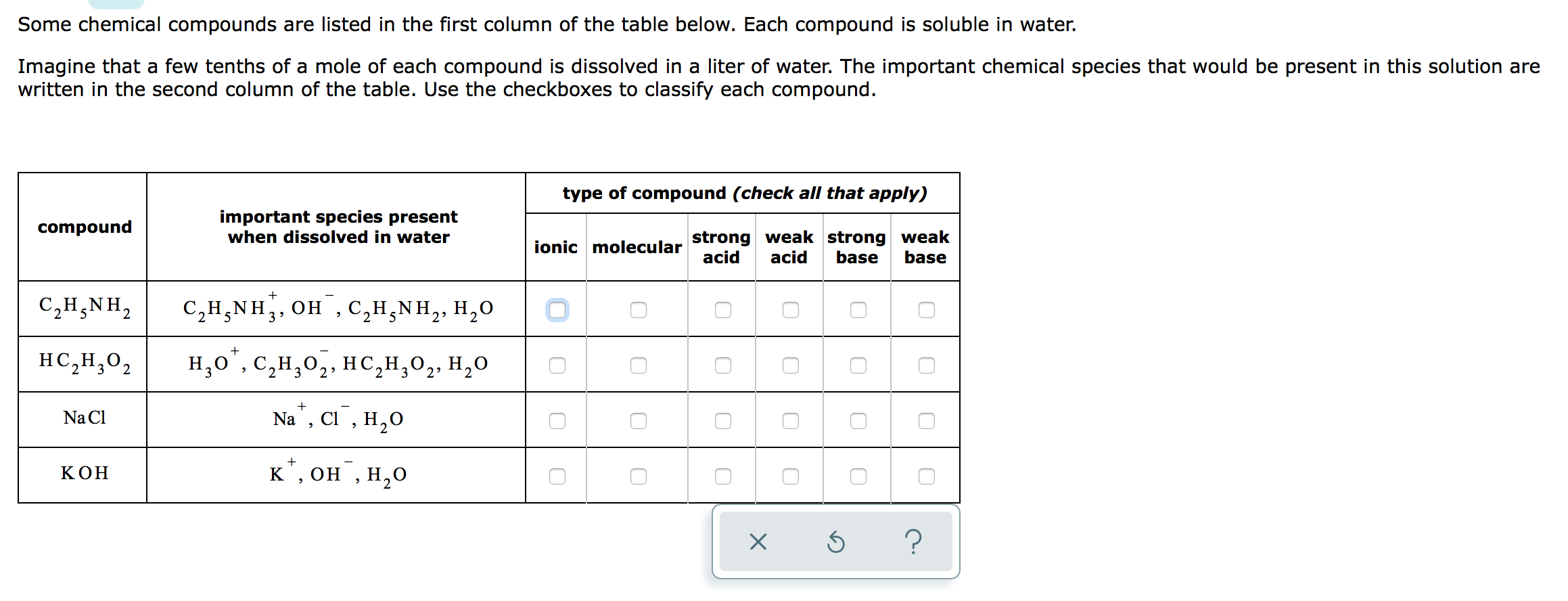
Transcribed Image Text:
Some chemical compounds are listed in the first column of the table below. Each compound is soluble in water. Imagine that a few tenths of a mole of each compound is dissolved in a liter of water. The important chemical species that would be present in this solution are written in the second column of the table. Use the checkboxes to classify each compound. compound C,HẠNH, HC,H,O2 Na Cl KOH important species present when dissolved in water + C₂H₂NH, OH, C₂H₂NH₂, H₂O ΝΗ + H,O*,C,H,O,, HC,H,O,, H,O 2 3 Na, Cl, H₂O + к†, он`, H2O type of compound (check all that apply) strong weak strong weak acid acid base base ionic molecular 0 0 0 0 X 0 0 Ś 0 0 0 0 0 ? Some chemical compounds are listed in the first column of the table below. Each compound is soluble in water. Imagine that a few tenths of a mole of each compound is dissolved in a liter of water. The important chemical species that would be present in this solution are written in the second column of the table. Use the checkboxes to classify each compound. compound C,HẠNH, HC,H,O2 Na Cl KOH important species present when dissolved in water + C₂H₂NH, OH, C₂H₂NH₂, H₂O ΝΗ + H,O*,C,H,O,, HC,H,O,, H,O 2 3 Na, Cl, H₂O + к†, он`, H2O type of compound (check all that apply) strong weak strong weak acid acid base base ionic molecular 0 0 0 0 X 0 0 Ś 0 0 0 0 0 ? Some chemical compounds are listed in the first column of the table below. Each compound is soluble in water. Imagine that a few tenths of a mole of each compound is dissolved in a liter of water. The important chemical species that would be present in this solution are written in the second column of the table. Use the checkboxes to classify each compound. compound C,HẠNH, HC,H,O2 Na Cl KOH important species present when dissolved in water + C₂H₂NH, OH, C₂H₂NH₂, H₂O ΝΗ + H,O*,C,H,O,, HC,H,O,, H,O 2 3 Na, Cl, H₂O + к†, он`, H2O type of compound (check all that apply) strong weak strong weak acid acid base base ionic molecular 0 0 0 0 X 0 0 Ś 0 0 0 0 0 ? Some chemical compounds are listed in the first column of the table below. Each compound is soluble in water. Imagine that a few tenths of a mole of each compound is dissolved in a liter of water. The important chemical species that would be present in this solution are written in the second column of the table. Use the checkboxes to classify each compound. compound C,HẠNH, HC,H,O2 Na Cl KOH important species present when dissolved in water + C₂H₂NH, OH, C₂H₂NH₂, H₂O ΝΗ + H,O*,C,H,O,, HC,H,O,, H,O 2 3 Na, Cl, H₂O + к†, он`, H2O type of compound (check all that apply) strong weak strong weak acid acid base base ionic molecular 0 0 0 0 X 0 0 Ś 0 0 0 0 0 ?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Use the data in the first column of Table 11.5 on page 699. These data give the boiling points of water at 17 different locations from Forbes experiment. Let F be the distribution from which these...
-
Assume that the transactions listed in the first column of the table below are anticipated by U.S. firms that have no other foreign transactions. Place an X in the table wherever you see possible...
-
Each numbered item in the first column is an example of a nonfinancial performance measure. Each item identified by a letter in the second column is a goal, objective, or other aspect of performance....
-
Consider a property investment that you finance with 20% down payment. For the remaining, you borrow 2'300'000 at a 6% rate monthly amortized loan for 25 years. This property, with 2% of EBITDA as...
-
Diamondback, Inc., has the following stockholders equity: Preferred stock, 1%, $12 par, 40,000 shares authorized and issued...................................... $ 480,000 Common stock, $8 par,...
-
Repeat Prob. 340 for a = 6 mm, b = 18 mm, d = 12 mm, and F = 4 kN. Repeat Prob. 340, A pin in a knuckle joint carrying a tensile load F deflects somewhat on account of this loading, making the...
-
Defendant Monty J. Person began working for Garage Solutions, LLC, in March 2015. Three months into his employment, Person was sent by the owner of Garage Solutions, Mark Fontenot, to Rexburg, Idaho,...
-
Beginning inventory, purchases, and sales data for prepaid cell phones for July are as follows: a. Assuming that the perpetual inventory system is used, costing by the LIFO method, determine the cost...
-
Need help with this relational model, please. I am having trouble understanding how to create the relational model.
-
You are discussing your 401(k) with Dan Ervin when he mentions that Sarah Brown, a representative from Bledsoe Financial Services, is visiting East Coast Yachts today. You decide that you should meet...
-
Consider a stock with a price of 100 and annual volatility = 0.2. Suppose that all interest rates are 1% p.a. with continuous compounding. For sim- plicity, suppose that all options are for 1 share...
-
I have to create hospital employee tracking system for the SVV hospitals. You will create a Java project to automate the adding, deleting and displaying the list of UAB hospital employees. The set of...
-
I need help with making these following adjustments to my program: Add a user-defined exception that can be thrown by one of the methods as part of the validation or error checking. The main method...
-
Mr. Semi is a new business owner who has recently secured four projects (X, Y, Z, and W). Each project will take 15 days to complete. However, one of the major challenges he faces is time management...
-
A hand truck is used to move two kegs, each of mass 40 kg. Neglecting the mass of the hand truck, determine (a) the vertical force P that should be applied to the handle to maintain equilibrium when ...
-
Can not figure out what is wrong with my Java code private char suit; private int value; //+PlayingCard (s;char, v;int) public playingCard(char s, int v){ suit = s; value = v; } //get suit public...
-
Your broker advised you to add some shares of a particular company to your portfolio. The broker indicated that you could expect a 10% return on your investment in 5 years and you decide to analyze...
-
An item of depreciable machinery was acquired on 1 July 2009 for $120,000 by cash It is expected to have a useful life of 10 years and zero salvage value On 1 July 2012, it was decided to revalue the...
-
The current spot rate of the Singapore dollar (S$) is $.50. The following option information is available: Call option premium on Singapore dollar (S$) = $.015. Put option premium on Singapore...
-
Jacksonville Corp. is a U.S.-based firm that needs $600,000. It has no business in Japan but is considering one-year financing with Japanese yen because the annual interest rate would be 5 percent...
-
Tarheel Co. plans to determine how changes in U.S. and Mexican real interest rates will affect the value of the U.S. dollar. a. Describe a regression model that could be used to achieve this purpose....
-
Flaherty Ltd sells two types of shoes, mens shoes and womens shoes. During the financial year ended 30 June 2019, fixed costs were $460 800 and sales were in the ratio of three units (pairs) of mens...
-
Newbery and Newstead is a public accounting firm specialising in auditing local medium-sized businesses. Fees charged for each audit are determined on the basis of identifiable hours worked on an...
-
Hotbox Ltd produces pizza boxes using two processes cutting and packaging. The production budget for the year ending 30 June 2019 estimated raw materials use of $400 000, factory overhead of $270...

Study smarter with the SolutionInn App



