Suppose that (x, y) = x xy + y 2x + 2y with D...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
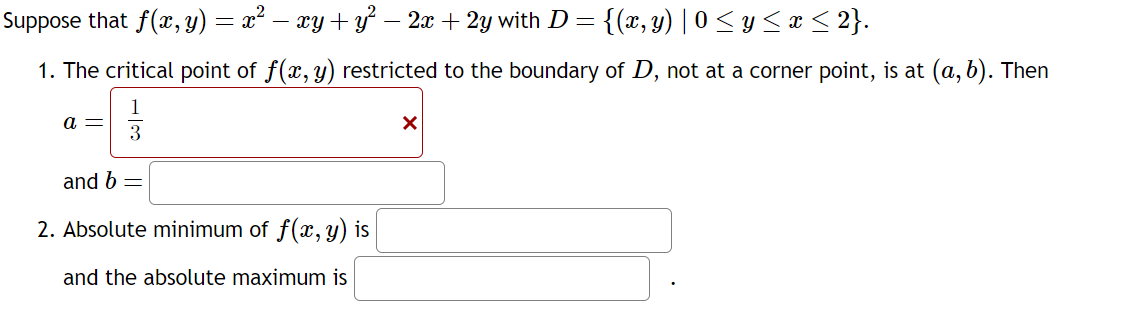
Suppose that ƒ(x, y) = x² − xy + y² − 2x + 2y with D = {(x, y) | 0 ≤ y ≤ x ≤ 2}. 1. The critical point of f(x, y) restricted to the boundary of D, not at a corner point, is at (a, b). Then 1 3 and b = 2. Absolute minimum of f(x, y) is and the absolute maximum is a = X Suppose that ƒ(x, y) = x² − xy + y² − 2x + 2y with D = {(x, y) | 0 ≤ y ≤ x ≤ 2}. 1. The critical point of f(x, y) restricted to the boundary of D, not at a corner point, is at (a, b). Then 1 3 and b = 2. Absolute minimum of f(x, y) is and the absolute maximum is a = X
Expert Answer:
Answer rating: 100% (QA)
16 n xy y 2x2y with D 24 1028 2022 tu ny2 ty x 2y 2 so ty with th 2nY2 n y 2 2 50 x 132 2 A3 13 K ... View the full answer

Related Book For 

College Mathematics For Business Economics, Life Sciences, And Social Sciences
ISBN: 978-0134674148
14th Edition
Authors: Raymond Barnett, Michael Ziegler, Karl Byleen, Christopher Stocker
Posted Date:
Students also viewed these mathematics questions
-
Two people, A and B, walk along the parabola y = x 2 in such a way that the line segment L between them is always perpendicular to the line tangent to the parabola at As position. What are the...
-
Jerry Clark is the sole proprietor of Affordable Digital Hearing, which does business by repairing hearing aids. In June 2006, Clark received a call from Kevin Wilson, a representative of...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Prepare journal entries to record the following grant-related transactions of an enterprise fund activity. Explain how these transactions should be reported in the enterprise funds financial...
-
The wavelength of a resonant mercury line is = 253.65 nm. The mean lifetime of mercury atoms in the state of resonance excitation is z = 0.15s. Evaluate the ratio of the Doppler line broadening to...
-
As a team, find at least four examples of WBSs for various types of projects (different from the examples in this chapter). Discuss the similarities and differences between how the WBSs are...
-
Claire Hunter plans on opening Claire Hunter Floral Designs. She is considering the various types of business organizations and wishes to organize her business with unlimited life and limited...
-
A 100-mm-long, hollow iron cylinder is exposed to a 1000C carburizing gas (a mixture of CO and CO2) at its inner and outer surfaces of radii 4.30 and 5.70 mm, respectively. Consider steady-state...
-
Mouret Corporation uses the following activity rates from its activity based costing to assign overhead cost of products activity cross pools setting up batches activity rate $ 9 2 . 6 8 per batch...
-
The Farr Quality Bathroom Tile Company has developed the following standards for one of its products: The company produced 1,200 units and sold all of the units at the budgeted sales price of $85....
-
Cleveland Clinic's case study - Video What are the case/simulation issues? What are the most valuable lessons you have learned? How can you apply what you have learned to your work, to your studies,...
-
the potential function f log lityy. Hind the complex omplex potential function
-
Requirement 1. Prepare the journal entry for production completed in June. (Prepare a single journal entry for this event. Record debits first, then credits. Select the explanation on the last line...
-
A mercury manometer can be used to measure the water level in a largetank. Given the manometer configuration below, determine the height of water in the tank(H). Assume that the (gage) pressure at...
-
bco + represents the and Q = 8xty-y fend o complex potential Id an electrical field
-
Exhibit 45.8 provides some guidance on two scenarios: bullish and bearish cash flow forecasts. Based on the two scenarios, model cash flows for American Greetings for fiscal years 2012 through 2015...
-
Daniel Hudson has been approached by a larger software company that is interested in possibly acquiring eHarbour. Daniel Hudson and the other partners of eHarbour are somewhat excited about this...
-
Show, if u(x, y) and v(x, y) are harmonic functions, that u + v must be a harmonic function but that uv need not be a harmonic function. Is e"e" a harmonic function?
-
Find a formula for the function W(x 1 , x 2 , x 3 , x 4 ) of four variables that gives the total volume of oil that can be carried in four oil tankers of capacities x 1 , x 2 , x 3 , and x 4 ,...
-
In Problem find f(x) and find the value(s) of x where f(x) = 0. f(x) x + 1
-
In Problem if a matrix is in reduced form, say so. If not, explain why and indicate a row operation that completes the next step of GaussJordan elimination. 3 6 0 2 05 -1 -3 9. -2
-
Calculate the acentric factor for ethanol. The vapour pressure of ethanol can be estimated from the following equation: \[ \log _{10} P^{\text {Sat }}=8.1122-\frac{1592.864}{t+226.184} \] where...
-
For liquid acetone at \(20^{\circ} \mathrm{C}\) and \(1 \mathrm{bar}\), \[ \beta=1.487 \times 10^{-3} /{ }^{\circ} \mathrm{C} \quad \alpha=62 \times 10^{-6} / \mathrm{bar} \quad V=1.287...
-
Determine the molar volume of ammonia vapour and ammonia liquid at \(321.55 \mathrm{~K}\) and 1.95 \(\mathrm{MPa}\). Ammonia is assumed to follow van der Waals equation of state.

Study smarter with the SolutionInn App


