Suppose the following is known about pet ownership among citizens of a city 80% of...
Fantastic news! We've Found the answer you've been seeking!
Question:
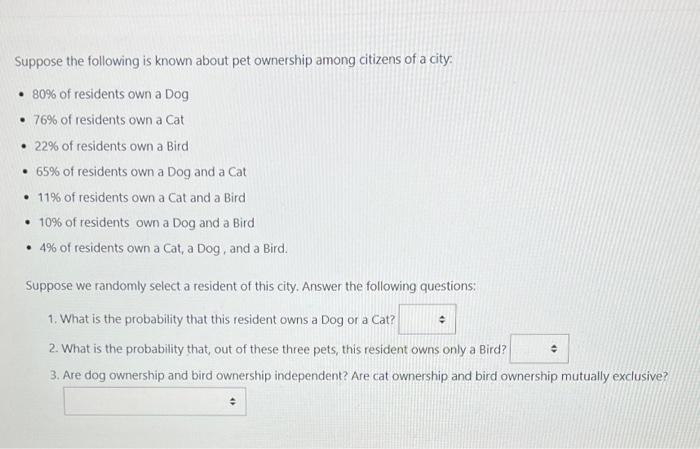
Transcribed Image Text:
Suppose the following is known about pet ownership among citizens of a city • 80% of residents own a Dog • 76% of residents own a Cat • 22% of residents own a Bird • 65% of residents own a Dog and a Cat • 11% of residents own a Cat and a Bird • 10% of residents own a Dog and a Bird • 4% of residents own a Cat, a Dog, and a Bird. Suppose we randomly select a resident of this city. Answer the following questions: 1. What is the probability that this resident owns a Dog or a Cat? 2. What is the probability that, out of these three pets, this resident owns only a Bird? 3. Are dog ownership and bird ownership independent? Are cat ownership and bird ownership mutually exclusive? Suppose the following is known about pet ownership among citizens of a city • 80% of residents own a Dog • 76% of residents own a Cat • 22% of residents own a Bird • 65% of residents own a Dog and a Cat • 11% of residents own a Cat and a Bird • 10% of residents own a Dog and a Bird • 4% of residents own a Cat, a Dog, and a Bird. Suppose we randomly select a resident of this city. Answer the following questions: 1. What is the probability that this resident owns a Dog or a Cat? 2. What is the probability that, out of these three pets, this resident owns only a Bird? 3. Are dog ownership and bird ownership independent? Are cat ownership and bird ownership mutually exclusive?
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
The following is known as the thermite reaction [Figure 5.8(b)]: This highly exothermic reaction is used for welding massive units, such as propellers for large ships. Using standard enthalpies of...
-
The following is known as the transcendental production function (TPF), a generalization of the well-known CobbDouglas production function: Y i = β 1 L β2 k β3 e...
-
Suppose the following is 2017 information for PepsiCo, Inc. and The Coca-Cola Company. Instructions Compute free cash flow for both companies and compare. (S in millions) PepsiCo Coca-Cola Net cash...
-
On 1/1/22, lmani and Ahmad formed AI Partnership, a limited partnership. lmani will be the general partner and Ahmad will be a limited partner. He will not participate in management of the...
-
Prepare a ten year professional development plan that will prepare you for a career in forensic accounting.
-
A solid mixture weighing 0.05485 g contained only ferrous ammonium sulfate and ferrous chloride. The sample was dissolved in 1 M H 2 SO 4 , and the Fe 2+ required 13.39 mL of 0.01234 M Ce 4+ for...
-
Identify the five objectives of a sales presentation and develop an example of how a sales presentation would accomplish each one.
-
Tex Hardware sells many of its products overseas. The following are some selected transactions. 1. Tex sold electronic subassemblies to a firm in Denmark for 120,000 Danish kroner (Dkr) on June 6,...
-
EM4.2 Expense Credit Card Required: Complete the following to record Internet services that Mookie The Beagle Concierge incurred. 1. Create an Expense paid with Credit Card. a. Select (+) New icon >...
-
Explain the process by which a Steinway grand piano is constructed as a subsystem of a larger system. From what the text tells you, give some examples of how the production subsystem is affected by...
-
Debating the Good and Harm of Globalization Recently, a university sponsored a roundtable on the broader implications of international business. The participants were an anti-international business...
-
Which one of the following statements about pressure is correct? A Pressure does not need a solid surface to act. B Pressure is a vector quantity that varies with time. C A Newton of force acting...
-
A small mailbag is released from a helicopter that is descending steadily at 1.00 m/s. (a) After 5.00 s, what is the speed of the mailbag? m/s (b) How far is it below the helicopter? m (c) What are...
-
1. Calculate the electric field created by the constant pl0 C/m infinitely long linear charge density on the z-axis shown in Figure 1, using Coulomb's law. zt t x Ps0 C/m Figure 1: Infinitely long...
-
The position of an electron is given by 7 = 4.59ti - 8.32127+ 1.66k, with t in seconds and in meters. At t = 4.20 s, what are (a) the x-component, (b) the y-component, (c) the magnitude, and (d) the...
-
8) In the xy-plane, an infinite conducting plane grounded. At a distance of d above the plane along the z-axis, there is a +q charge. a) Using the image method, write the electric potential. Show...
-
The following selected data is from the financials of Aztec Corporation: Total Assets Total Current Liabilities Total Long-term Liabilities Total Stockholders' Equity Interest Expense Income Tax...
-
Determine which of the following limits exist. Compute the limits that exist. lim x-0 1- + 3x X
-
Silicon for computer chips is grown in large cylinders called "boules" that are 300 mm in diameter and 2 m in height. The density of silicon is 2.33 g/cm3. Silicon wafers for making integrated...
-
Explain why increasing concentrations of CO2 in the atmosphere affect the quantity of energy leaving Earth but do not affect the quantity of energy entering from the Sun.
-
(a) What conditions must be met if a molecule with polar bonds is non-polar? (b) What geometries will signify non-polar molecules for AB2, AB3, and AB4 geometries?
-
Give an example of a local wind pattern. Give an example of a global wind pattern.
-
What drives surface currents?
-
Do surface currents affect water temperature, air temperature, or both?

Study smarter with the SolutionInn App



