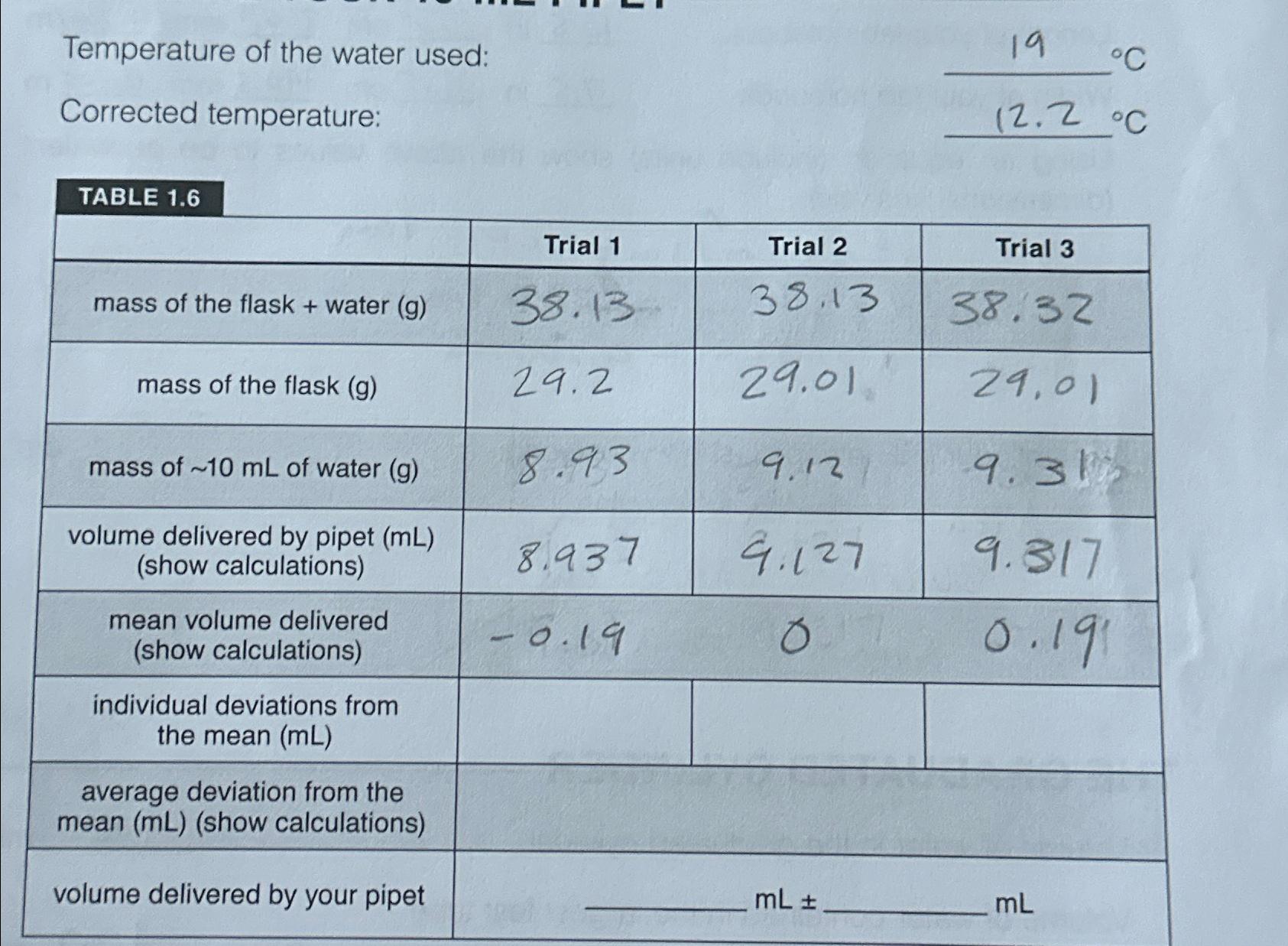
Temperature of the water used: Corrected temperature: TABLE 1.6 mass of the flask + water (g)...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Temperature of the water used: Corrected temperature: TABLE 1.6 mass of the flask + water (g) mass of the flask (g) mass of ~10 mL of water (g) volume delivered by pipet (mL) (show calculations) mean volume delivered (show calculations) individual deviations from the mean (mL) average deviation from the mean (mL) (show calculations) volume delivered by your pipet Trial 1 38.13 29.2 8.93 8.937 -0.19 Trial 2 38.13 29.01 9.12 9.127 o mL 19 C 12.2 C Trial 3 38.32 29,01 9.31 9.317 0.191 mL Temperature of the water used: Corrected temperature: TABLE 1.6 mass of the flask + water (g) mass of the flask (g) mass of ~10 mL of water (g) volume delivered by pipet (mL) (show calculations) mean volume delivered (show calculations) individual deviations from the mean (mL) average deviation from the mean (mL) (show calculations) volume delivered by your pipet Trial 1 38.13 29.2 8.93 8.937 -0.19 Trial 2 38.13 29.01 9.12 9.127 o mL 19 C 12.2 C Trial 3 38.32 29,01 9.31 9.317 0.191 mL
Expert Answer:
Answer rating: 100% (QA)
The image youve provided is of a worksheet displaying a set of measurements taken from an experiment involving a pipet The worksheet is structured as ... View the full answer

Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Posted Date:
Students also viewed these chemical engineering questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
Using the Internet, explore the most useful mobile applications for a business or industry of your choice. Also explore mobile applications for this business or industry that are not currently...
-
Facebook is a corporation. Why do you think the firm uses this form of ownership?
-
Find the derivative of the function. f (t) = 3 2 t /t
-
Consider two alternatives, each of which will accomplish the same EPA-mandated pollution control. Using an incremental IRR analysis, determine the preferred alternative assuming MARR is 20...
-
A larger and more modem main post office is to be constructed at a new location in Davis, California. Growing suburbs caused a shift in the population density from where it was 40 years ago when the...
-
A point P is equidistant from R(-2, 4) and S(6, -4) and its x-coordinate is twice its y-coordinate. (i) Find the coordinates of P. (ii) Hence, determine whether P, R and S are collinear, showing your...
-
Light-It-Up Company maintains and repairs warning lights, such as those found on radio towers and lighthouses. Light-It-Up Company prepared the following end-of-period spreadsheet at August 31, 20Y5,...
-
Each of the following is a benefit of economic growth: a. an expansion in the range of goods and services available for purchase b. reductions in infant mortality c. increased income inequality
-
please provide real case that happen in Malaysia or other country that can show reinvestment risk on bonds.
-
(c) The Dublin based 'Urban Movement' company manufactures a foldable electric scooter. (i) Describe, with the aid of a diagram(s), a suitable mechanism for folding the scooter handle. (ii) Outline...
-
create a class within a project that throws exceptions for blank or empty inputs. test.java import java.util.Scanner; public class test { public static void main(String[] args) { //accepting the...
-
I need one jar and one java file for this. It should calculate the first 1000 prime numbers as an array of longs. Then it needs to write the array of longs to a file named Lab8Primetime, with each...
-
2. (11 marks total) Ethanol (CHOH) burns in a domestic furnace at a pressure of 101.3 kPa according to the following stoichiometric equation: CHOH + 4.0 (O+3.76 N) 2.0 CO+ 3.0 HO + 1.0 O + 15.04 N...
-
Lets assume that you have been asked to calculate risk-based capital ratios for a bank with the following accounts: Cash = $15 million Government securities = $17 million Mortgage loans = $40 million...
-
We all experience emotions, but some people disguise their true feelings better than others. Do you think this is a helpful or harmful thing to do? Under what conditions do you think it would be most...
-
Calculate the pH of a solution prepared by adding exactly 10.0 mL of a 14.8 M KOH solution to 200 mL of water, then adding water until the volume of solution is exactly 250 mL.
-
H is -108 kJ for the formation of phosgene at the normal reaction temperature in Bhopal. In which direction does the system react if temperature is increased? CO(g) + Cl(g) COCI(g)
-
Suppose you have an exothermic reaction with H = -15 kJ and a S of -150 J/K. Calculate G and K eq at 10, 100, and 1000 K. For each reaction, an equilibrium constant at 298 K is given. Calculate G ...
-
Explain two ways a fixed-for-fixed currency swap can be valued.
-
Explain the difference between the credit risk and the market risk in a swap.
-
The time-series graph in Figure 24 depicts the number of residents in the United States living in poverty. Why might this graph be considered misrepresentative? Approach Look for any characteristics...

Study smarter with the SolutionInn App


