The amount of energy required to break a bond is same as the amount of energy...
Fantastic news! We've Found the answer you've been seeking!
Question:

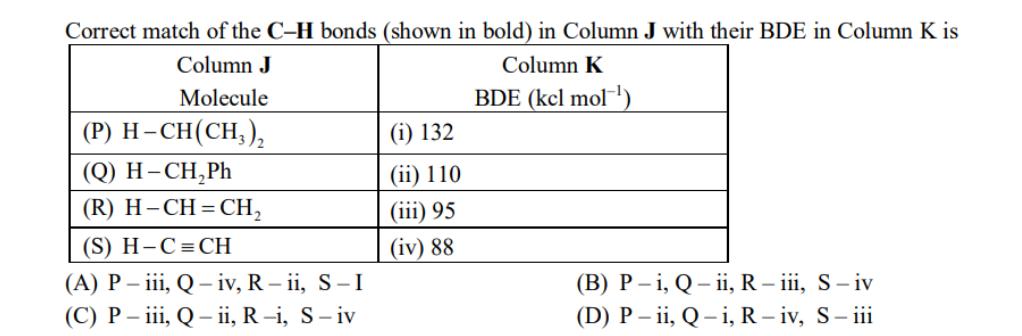
Transcribed Image Text:
The amount of energy required to break a bond is same as the amount of energy released when the same bond is formed. In gaseous state, the energy required for homolytic cleavage of a bond is called Bond Dissociation Energy (BDE) or Bond Strength. BDE is affected by s-character of the bond and the stability of the radicals formed. Shorter bonds are typically stronger bonds. BDEs for some bonds are given below: H₂C-H(g) H₂C (g) H(g) AH = 105 kcal mol-1 CI-CI (g) H₂C-Cl (g) H-Cl (g) Ci (g) + H₂C (g) + H*(g) + Cl (g) AH° = 58 kcal mol-¹ Ci (9) AH = 85 kcal mol-¹ ci (g) AH = 103 kcal mol-¹ Correct match of the C-H bonds (shown in bold) in Column J with their BDE in Column K is Column J Column K Molecule BDE (kel mol-¹) (P) H-CH(CH3)₂ (Q) H-CH₂Ph (R) H-CH=CH₂ (S) H-C=CH (A) P-iii, Q-iv, R-ii, S-I (C) P-iii, Q-ii, R-i, S-iv (1) 132 (ii) 110 (iii) 95 (iv) 88 (B) P-i, Q-ii, R - iii, S-iv (D) P-ii, Q-i, R-iv, S-iii The amount of energy required to break a bond is same as the amount of energy released when the same bond is formed. In gaseous state, the energy required for homolytic cleavage of a bond is called Bond Dissociation Energy (BDE) or Bond Strength. BDE is affected by s-character of the bond and the stability of the radicals formed. Shorter bonds are typically stronger bonds. BDEs for some bonds are given below: H₂C-H(g) H₂C (g) H(g) AH = 105 kcal mol-1 CI-CI (g) H₂C-Cl (g) H-Cl (g) Ci (g) + H₂C (g) + H*(g) + Cl (g) AH° = 58 kcal mol-¹ Ci (9) AH = 85 kcal mol-¹ ci (g) AH = 103 kcal mol-¹ Correct match of the C-H bonds (shown in bold) in Column J with their BDE in Column K is Column J Column K Molecule BDE (kel mol-¹) (P) H-CH(CH3)₂ (Q) H-CH₂Ph (R) H-CH=CH₂ (S) H-C=CH (A) P-iii, Q-iv, R-ii, S-I (C) P-iii, Q-ii, R-i, S-iv (1) 132 (ii) 110 (iii) 95 (iv) 88 (B) P-i, Q-ii, R - iii, S-iv (D) P-ii, Q-i, R-iv, S-iii
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The amount of energy required to spin-flip a nucleus depends both on the strength of the external magnetic field and on the nucleus. At a field strength of 4.7 1 rf energy of 200 MHz is required to...
-
Calculate the amount of energy required to spin-flip a proton in a spectrometer operating at 300MHz. Does increasing the spectrometer frequency from 200 to 300 MHz increase or decrease the amount of...
-
The amount of energy collected from every solar panel on a photovoltaic power plant. Determine whether the data set is a population or a sample. Explain your reasoning.
-
In Exercises 8594, find all values of x satisfying the given conditions. y = 2x + x 8x + 2 and y = 6.
-
Frequency inverters have been used for many years for speech scrambling. Indeed, a voice signal x (n) becomes unintelligible if we invert its spectrum as shown in figure(a) Determine how frequency...
-
Raymond Co. began operations in January 2011. The information below is for Raymond Co.'s operations for the three months from January to March (the first quarter) of 2012: Expenses for Quarter 1...
-
The National Railroad allocates all central corporate overhead costs to its divisions. Some costs, such as specified internal auditing and legal costs, are identified on the basis of time spent....
-
Jewett Online Company has the following liability accounts after posting adjusting entries: Accounts Payable $63,000, Unearned Ticket Revenue $24,000, Estimated Warranty Liability $18,000, Interest...
-
radebook Quiz Me: Quiz Me: 11.7-2 Graph systems of linear inequalities. stents Resource Library e Resources Options Graph the system of inequalities. 3x + y 2 6x + 2y > 20 nication Tools Choose the...
-
The lifetime (in months) of a battery is modeled by a random variable X that has pdf f ( x ) = K x 1 ( x > 0 ) where K = ln ( 1 / ) for an unknown parameter ( 0 , 1 ) . (Here 1 ( x > 0 )...
-
What is the density in g dm of SO at 20 C and 750 mm Hg pressure?
-
You're sunbathing on the island shown on the map below. The island is six miles from shore at the closest point, and the nearest store is a convenience store seven miles down the beach. If you can...
-
Discuss the advanced methodologies used to analyze slope stability and prevent landslides in steep terrain. What role do geotechnical monitoring systems, retaining structures, and drainage solutions...
-
Zeke the Plumber follows the accrual accounting revenuerecognition principle. Zeke performed services for a client on July10, notified the client that he did the job the next day July 11th.The...
-
Bonus - Reverse Engineering (30pts) (a) Reverse engineer the behavior of the sequential circuit shown below. Show the state machine table and the FSM state machine diagram. (10pts) a clk s1...
-
Beginning work in process (50% complete) 45,000 units Units received from Dept 1. 255,000 units Finished and transferred 270,000 units Ending work in process (50% complete) 25,000 units All materials...
-
EXERCISE 7-12 Uncertain Cash Flows LO 7-4 The Cambro Foundation, a nonprofit organization, is planning to invest $104,950 in a project that will last for three years. The project will produce net...
-
Refer to Example 9.15. Add the following functionality to this program: Allow the user to enter the cost of a gallon of gas on each trip and use a function, Cost() to calculate the cost of purchasing...
-
Zirconium is one of the few metals that retains its structural integrity upon exposure to radiation. The fuel rods in most nuclear reactors therefore are often made of zirconium. Answer the following...
-
Discuss the relationship between wmax and the magnitude and sign of the free energy change for a reaction. Also discuss wmax for real processes.
-
Consider the relationship The equilibrium constant for some hypothetical process was determined as a function of temperature (in kelvins) with the results plotted below. From the plot, determine the...
-
The statements of financial position for Maxi Ltd and Mini Ltd are set out below. On 1 April last year, Maxi Ltd bought 1,500,000 shares of Mini Ltd for a total consideration of 5 million. At that...
-
A year ago Pod Ltd bought 225,000 1 fully paid ordinary shares of Pea Ltd for a consideration of 500,000. Pea Ltds share capital and share premium were each the same as at todays date. Simplified...
-
It has been suggested that too much information might be as bad as too little information for users of annual reports. Explain.

Study smarter with the SolutionInn App



