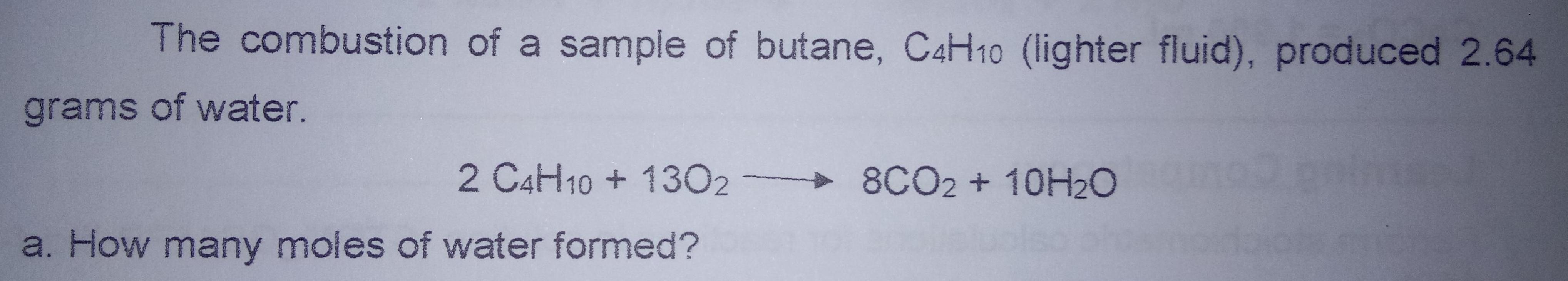
The combustion of a sample of butane, C4H10 (lighter fluid), produced 2.64 grams of water. 2...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
The combustion of a sample of butane, C4H10 (lighter fluid), produced 2.64 grams of water. 2 C4H10 + 1302 8CO2 + 10H2O a. How many moles of water formed? b.How many moles of butane burned? c. How many grams of butane burned? d. How much oxygen was used up in moles? e. How much oxygen was used up in grams? The combustion of a sample of butane, C4H10 (lighter fluid), produced 2.64 grams of water. 2 C4H10 + 1302 8CO2 + 10H2O a. How many moles of water formed? b.How many moles of butane burned? c. How many grams of butane burned? d. How much oxygen was used up in moles? e. How much oxygen was used up in grams?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The combustion of a fuel usually results in an increase in pressure when the volume is held constant, or an increase in volume when the pressure is held constant, because of the increase in the...
-
The combustion of a hydrocarbon fuel with air results in a mixture of products of combustion having the composition on a volume basis as follows: 4.89 percent carbon dioxide, 6.50 percent water...
-
A power plant is driven by the combustion of a complex fossil fuel having the formula C 11 H7S. Assume the air supply is composed of only N 2 and O 2 with a molar ratio of 3.76:1.00, and the N 2...
-
3. Complete all parts. Let f(x) = I 2+1 on the interval [0, 2). = x 2+1 (a) Explain why the function f(x) on interval [0,2] satisfies the Mean Value Theorem. (b) find all numbers c that satisfy the...
-
Mica Computers provides customers the option to purchase products with three installment payments made over 12 months (equal payments at the end of the 4th, 8th, and 12th months). In January 2011,...
-
FIFO method (continuation of 17-30). Do Problem 17-30 using the FIFO method of process costing. Explain any difference between the cost per equivalent unit in the Assembly Department under the...
-
Reconsider the data from Problem 56. What is the capital recovery cost of Model 334A? Data from problem 56 Octavia Bakery is planning to purchase one of two ovens. The expected cash flows for each...
-
Calgary Corporation produces a liquid solvent in two departments: Mixing and Finishing. Assume that Calgary Corporation provides you with the following information for finishing operations for...
-
A crystal was analyzed using X-ray diffraction with radiation from a copper source. The observed angle was 8.61. Determine the distance between layers of the crystal. d = Element (pm) CO 179 Cr 229...
-
P Company holds an 80% interest in SFr Company, a Swiss company. A trial balance for P Company and SFr Company at December 31, 2020, and other data are given in Problems 13-3 and 13-4. Ignore...
-
The second portion of the assignment requires you to prepare a full company overview that includes detailed information on your companys history executive management team future projections and other...
-
A contract involving a mistake of fact can sometimes be avoided. (True/False)
-
When is a partys reliance on anothers misrepresentation not justifiable?
-
A contract entered into under duress is voidable. (True/False)
-
In selling a warehouse to A&B Enterprises, Ray does not disclose that the foundation was built on unstable pilings. A&B may later avoid the contract on the ground of a. misrepresentation. b. undue...
-
When does the one-year period of the one-year rule begin?
-
How can I solve this question in MATLAB? Using the Newton's laws write the differential equation of a harmonic oscillator of the mass m, which is a system that, when displaced from its equilibrium...
-
Explain the differences and similarities between fringe benefits and salary as forms of compensation.
-
What volume of water would be required to dissolve 10.0 g of silver chloride, AgCl, at 25C? What volume of 0.15 M NH 3 would be required to dissolve the same quantity of silver chloride?
-
What is the de Broglie wavelength of an oxygen molecule, O 2 , traveling at 535 m/s? Is the wavelength much smaller or much larger than the diameter of an atom (on the order of 100 pm)?
-
Calculate the CCl and CC bond lengths in ethyl chloride, C 2 H 5 Cl, using values for the covalent radii from Table 9.4. How do these values compare with the experimental values: CCl, 177; CC, 155...
-
Financial Reporting Problem Marks and Spencer plc (M&S) The financial statements of M&S (GBR) are presented in Appendix A. The companys complete annual report, including the notes to the financial...
-
Cooke Company has a fiscal year ending on September 30. Selected data from the September 30 worksheet are presented below. Instructions (a) Prepare a complete worksheet. (b) Prepare a classified...
-
Let \(X, Y, X_{n}, Y_{n}: \Omega ightarrow \mathbb{R}, n \geqslant 1\), be random variables. a) If, for all n > 1, Xn Yn and if (Xn, Yn) (X, Y), then XIL Y. b) Let X Y such that X, Y ~ B1/2 = (80...

Study smarter with the SolutionInn App



