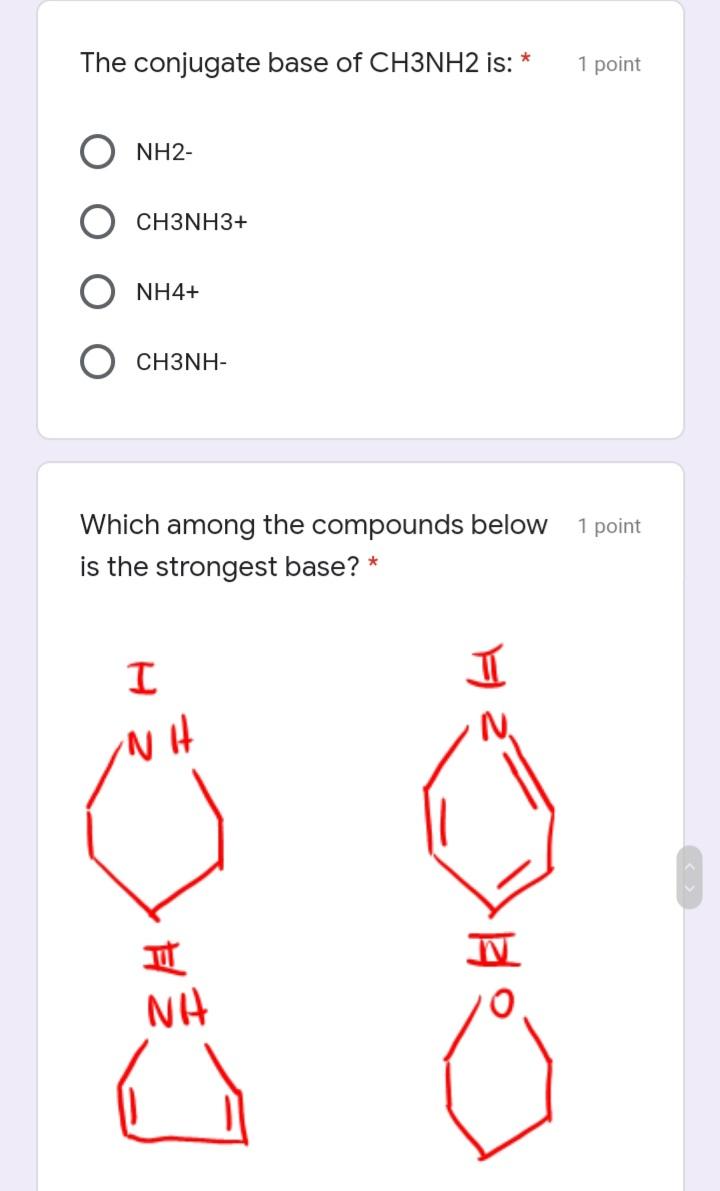
The conjugate base of CH3NH2 is: * 1 point NH2- CH3NH3+ NH4+ CH3NH- Which among the...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
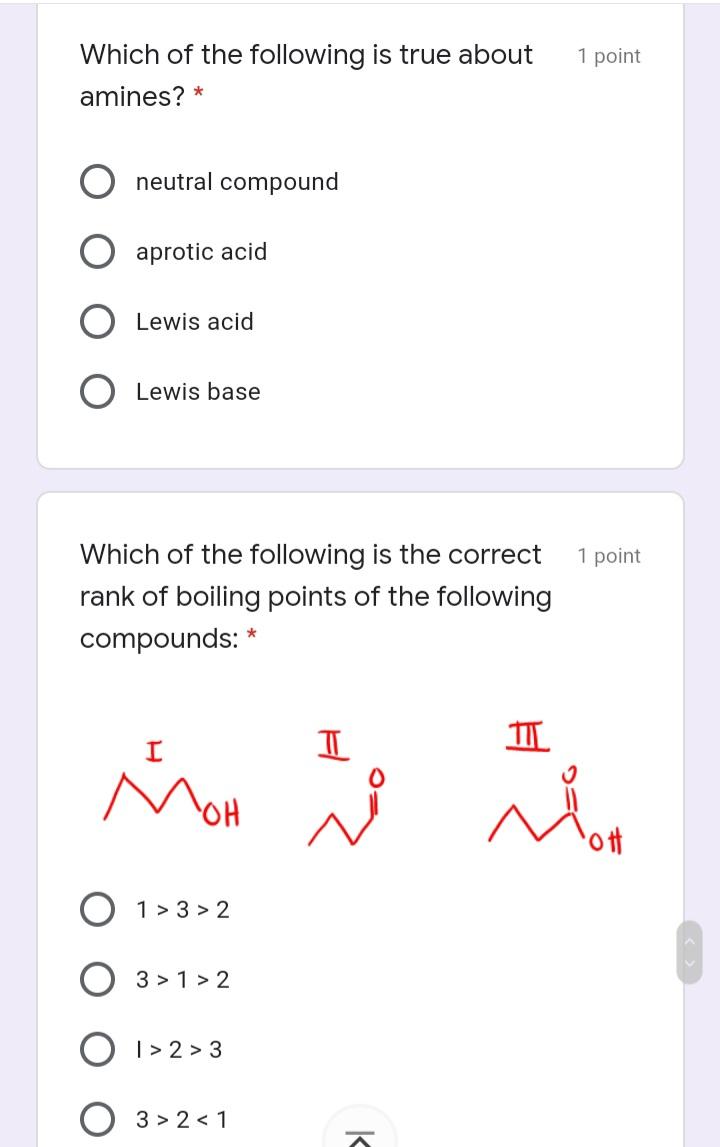
The conjugate base of CH3NH2 is: * 1 point NH2- CH3NH3+ NH4+ CH3NH- Which among the compounds below 1 point is the strongest base? * NH NH Which of the following describes the 1 point correct polarization in acrolein. (The smell of burnt fat (as when cooking oil is heated to its smoke point) is caused by glycerol in the burning fat breaking down into acrolein.) * I H2 C=CH-CH O 8* I H2 C=CH-CH=0 M H2 C-CH- CH=0 V H2 C=CH–CH=0 O IV II Which of the following is true about 1 point amines? * neutral compound aprotic acid Lewis acid Lewis base Which of the following is the correct 1 point rank of boiling points of the following compounds: II MoH ott O 1> 3 > 2 3 > 1 > 2 O I> 2 > 3 3 > 2 < 1 The conjugate base of CH3NH2 is: * 1 point NH2- CH3NH3+ NH4+ CH3NH- Which among the compounds below 1 point is the strongest base? * NH NH Which of the following describes the 1 point correct polarization in acrolein. (The smell of burnt fat (as when cooking oil is heated to its smoke point) is caused by glycerol in the burning fat breaking down into acrolein.) * I H2 C=CH-CH O 8* I H2 C=CH-CH=0 M H2 C-CH- CH=0 V H2 C=CH–CH=0 O IV II Which of the following is true about 1 point amines? * neutral compound aprotic acid Lewis acid Lewis base Which of the following is the correct 1 point rank of boiling points of the following compounds: II MoH ott O 1> 3 > 2 3 > 1 > 2 O I> 2 > 3 3 > 2 < 1
Expert Answer:
Answer rating: 100% (QA)
1 Option D Conjugate base is formed by the loss of on... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The conjugate base of diethyl malonate can serve as a nucleophile to attack a wide range of electrophiles. Identify the product that is formed when the conjugate base of diethyl malonate reacts with...
-
Show the resonance structures for the conjugate base of phenol.
-
Show the resonance structures for the conjugate base of the Meta isomer of nitro-phenol and confirm that the nitro group is less effective at stabilizing this anion than it is in the case of the Para...
-
Calculate the directional derivative in the direction of v at the given point. Remember to use a unit vector in your directional derivative computation. g(x, y, z) = xe-y, v = (1, 1, 1), P = (1,2,0)
-
Classify each of the following transactions as increasing, decreasing, or having no effect on cash flows: a. Prepaying rent for the month b. Accruing the wages owed to employees at the end of the...
-
On August 2, 2015, Jun Co. receives a $6,000, 90-day, 12% note from customer Ryan Albany as payment on his $6,000 account. Prepare Juns journal entry assuming the note is honored by the customer on...
-
\(C_{P}-C_{V}=R\) is valid for (a) Real gases (b) All gases (c) Ideal gases (d) None of these.
-
A tornado struck the only manufacturing plant of Toledo Farm Implements (TFI) on June 1. All work-in-process inventory was destroyed, but a few records were salvaged from the wreckage and from the...
-
What is the present value of $100,000 to be received in 20 years? Your required rate of return is 8% per year. What is the annual interest rate required for 20,000 to grow into 30,000 in 15 years?
-
The trial balance of Alagoass Boutique at December 31 shows Inventory R$21,000, Sales Revenue R$156,000, Sales Returns and Allowances R$4,000, Sales Discounts R$3,000, Cost of Goods Sold R$92,400,...
-
Why do applications for business credit typically require a more thorough investigation than applications for consumer credit and what sources of information can be tapped to provide this more...
-
SportZ has invoices for materials purchased from Platinum Steel Incorporated. The invoices are for $4,242 due 60 days ago, $12,567 due in 30 days, and $18,451 due in 140 days. If SportZ pays all of...
-
Kaiyu has a $40,000 car loan at 12% for 36 months, on which she makes monthly payments of $1,328.57. After making her 10th payment, she wants to know the amount to pay the loan off. What is the...
-
1 A soup recipe calls for 3 cups of chicken broth and will make enough to serve 10 people. How much broth is required if the recipe is modified to serve 6 people? The recipe will require cups of...
-
The following scatterplot is created using one of the datasets in the income and housing data in the spreadsheet linked above. 40 35 30 25 20 20 15 10 5 0 500 550 600 650 Which variables are used in...
-
7. In the coin-flipping situation of problem 1, what is P(H = 4| H is even)?
-
Select all that apply - When supplies are purchased on credit it means that: Multiple select question. a liability has been incurred. the Accounts Payable account will be increased. the business will...
-
List four items of financial information you consider to be important to a manager of a business that has been operating for a year.
-
The hydrogen's of the hydrocarbons can b randomly replaced by chlorine by reaction of the hydrocarbon with Cl2 in the presence of light. An unknown compound, A, with the formula C4H10, produces two...
-
Show the products of thesereactions: CH3 Br CH,CH,OH CH,OH a) Ph- -Br b) Ph CH; CH, Br . CH;OH Br d) ELOH Ph
-
Explain which of these reactions would have the faster rate: CI CH3 CI CH,CH,CH, + CH;-N: CH3 or CH;-NH2 CH,CH,CH, +
-
The upper curve in the boiling point diagram is called (a) The saturated vapour curve (b) The dew point curve (c) The saturated liquid curve (d) Both (a) and (b).
-
A vertical cylinder containing helium gas is filled with a piston of \(50 \mathrm{~kg}\) mass and crosssectional area of \(0.025 \mathrm{~m}^{2}\). If the atmospheric pressure outside the cylinder is...
-
The Joule-Thomson coefficient for any gas at inversion point is (a) 1 (b) 0 (c) 2 (d) 3

Study smarter with the SolutionInn App


