The following data was collected after running a calorimetric experiment using three salts. Table 1: AHsol...
Fantastic news! We've Found the answer you've been seeking!
Question:
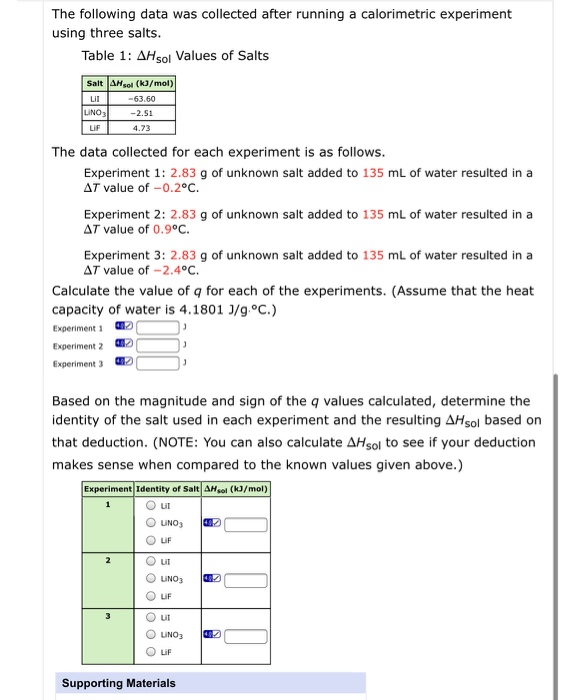
Transcribed Image Text:
The following data was collected after running a calorimetric experiment using three salts. Table 1: AHsol Values of Salts Salt AHsol (kJ/mol) Lil LINO3 -63.60 -2.51 LIF 4.73 The data collected for each experiment is as follows. Experiment 1: 2.83 g of unknown salt added to 135 ml of water resulted in a AT value of -0.2°C. Experiment 2: 2.83 g of unknown salt added to 135 mL of water resulted in a AT value of 0.9°c. Experiment 3: 2.83 g of unknown salt added to 135 mL of water resulted in a AT value of -2.4°c. Calculate the value of q for each of the experiments. (Assume that the heat capacity of water is 4.1801 J/g.°C.) Experiment 1 Experiment 2 Experiment 3 Based on the magnitude and sign of the q values calculated, determine the identity of the salt used in each experiment and the resulting AHsol based on that deduction. (NOTE: You can also calculate AHsol to see if your deduction makes sense when compared to the known values given above.) Experiment Identity of Salt AHsol (kɔ/mol) LINO3 LIF O ut O UNO3 LIF O ut O UNO3 Lif Supporting Materials O O 0 O O The following data was collected after running a calorimetric experiment using three salts. Table 1: AHsol Values of Salts Salt AHsol (kJ/mol) Lil LINO3 -63.60 -2.51 LIF 4.73 The data collected for each experiment is as follows. Experiment 1: 2.83 g of unknown salt added to 135 ml of water resulted in a AT value of -0.2°C. Experiment 2: 2.83 g of unknown salt added to 135 mL of water resulted in a AT value of 0.9°c. Experiment 3: 2.83 g of unknown salt added to 135 mL of water resulted in a AT value of -2.4°c. Calculate the value of q for each of the experiments. (Assume that the heat capacity of water is 4.1801 J/g.°C.) Experiment 1 Experiment 2 Experiment 3 Based on the magnitude and sign of the q values calculated, determine the identity of the salt used in each experiment and the resulting AHsol based on that deduction. (NOTE: You can also calculate AHsol to see if your deduction makes sense when compared to the known values given above.) Experiment Identity of Salt AHsol (kɔ/mol) LINO3 LIF O ut O UNO3 LIF O ut O UNO3 Lif Supporting Materials O O 0 O O
Expert Answer:
Answer rating: 100% (QA)
First of all Convert Ansol of each salt om KJmof to ... View the full answer

Related Book For 

Probability and Statistical Inference
ISBN: 978-0321923271
9th edition
Authors: Robert V. Hogg, Elliot Tanis, Dale Zimmerman
Posted Date:
Students also viewed these accounting questions
-
Although we often assume that the heat capacity of a substance is not temperature dependent, this is not strictly true, as shown by the following data for ice: Use these data to calculate graphically...
-
Among the data collected for the World Health Organization air quality monitoring project is a measure of suspended particles, in g/m3. Let X and Y equal the concentration of suspended particles in...
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
During the year, the total actual cost of inventory produced for The Wood Legacy was $1,500,000 (assume no beginning or ending inventory balances). Actual manufacturing overhead was $300,000. Direct...
-
Kenney Chesney decides to invest $ 43,630 today because he would like to build a guitar factory for $ 100,000 in 17 years. Interest is compounded annually. What annual interest rate must he receive...
-
log 18 Use a calculator to evaluate each expression. Round your answers to three decimal places.
-
The trial balance of URNO 1 Child Care, Inc., does not balance. The following errors are detected: a. Cash is understated by $5,000. b. A \($3,000\) debit to Accounts receivable was posted as a...
-
Ten-year-old Marla says her classmate Bernadette will never get good grades because she's lazy. Jane believes that Bernadette tries but can't concentrate because her parents are divorcing. Why is...
-
Consider the following indifference curve graphs for five different applicants for a pay for performance job. Pay A B C D E Productivity/ Effort (Q) a. Define the marginal disutility of effort (also...
-
As Mel Smith was doing his year-end accounting, he noticed that the bookkeeper had made errors in recording several transactions. The erroneous transactions are as follows: (a) A check for $700 was...
-
Ace Shoe Company sells heel replacement kits for men's shoes. It has fixed costs of $10 million and unit variable costs of $5 per pair. If the company charges $15 per pair, how many pairs must it...
-
Chalk is calcium carbonate, CaCO 3 . (Its basicallymade of tiny shells of ancient sea creatures.) The density ofchalk is 2.499 g/cm 3 . Calcium carbonate reactswith hydrochloric acid to make calcium...
-
Reflecting upon your learnings of Digital Business Analysis core knowledge areas and skillset and Plan future professional development to address areas for improvement. Develop strategies for own...
-
Exercise 13-1 Below are two classes that store some information about two different types of art pieces, Paintings and Sculptures. Each class has an instance variable containing the artist's name (a...
-
How has the development of advanced numerical modeling tools like the finite difference method (FDM) and the distinct element method (DEM) revolutionized the analysis of soil-structure interaction,...
-
Consider the structural problem shown in Figure 4.1a y X L N Figure 4.1a The "slab" of linearly elastic material is subjected to shear loads applied on the external surface and parallel to the...
-
What are some ways that synergistic communication can positively affect organizational communication? Furthermore, how does SC relate to the achieving of (or the not achieving of) the overall goals...
-
Write a paper about medication error system 2016.
-
In 2012, Red Rose tea randomly began placing 1 of 12 English porcelain miniature figurines in a l00-bag box of the tea, selecting from 12 nautical figurines. (a) On the average, how many boxes of tea...
-
Suppose that a third group of nurses was observed along with groups I and II of Exercise 9.2-1, resulting in the respective frequencies 130, 75, 136, 33, 61, and 65. Test H0: pi1 = pi2 = pi3, i = 1,...
-
Show that the expression in the exponent of Equation 4.5-2 is equal to the function q(x, y) given in the text.
-
With your instructor's consent, identify a company and perform a background review of it to identify high-risk areas for an upcoming audit. Utilize all the electronic sources that have information...
-
According to the AICPA's ethical standards, an auditor would be considered in- dependent in which of the following instances? a. The auditor has an automobile loan from a client bank. b. The auditor...
-
Explain how "privileged communication" differs from confidential information.

Study smarter with the SolutionInn App


