The deferred tax liability was caused by accelerated depreciation and the deferred tax asset is for rentals
Fantastic news! We've Found the answer you've been seeking!
Question:
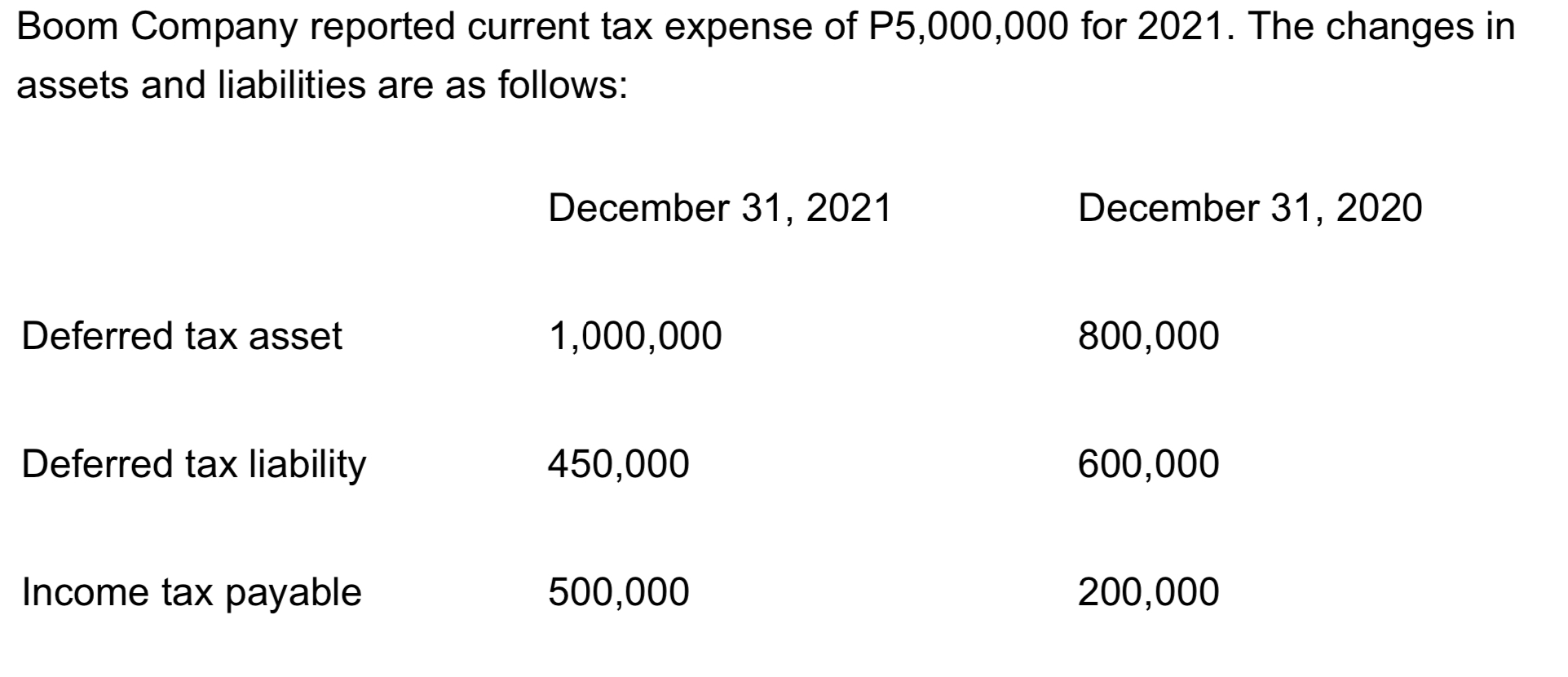
The deferred tax liability was caused by accelerated depreciation and the deferred tax asset is for rentals received in advance. What amount of total expense should be recognized in 2021?
Related Book For 

Financial reporting, financial statement analysis and valuation a strategic perspective
ISBN: 978-0324789416
7th Edition
Authors: James M Wahlen, Stephen P Baginskl, Mark T Bradshaw
Posted Date:




