The formation of molecular hydrogen at 3000 K proceeds through the following elementary reaction: 2H (g)...
Fantastic news! We've Found the answer you've been seeking!
Question:

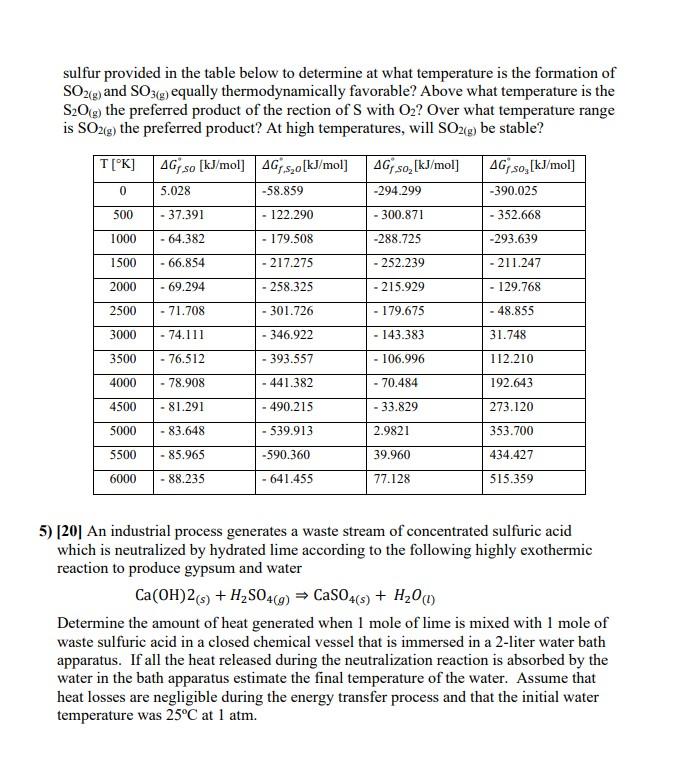
Transcribed Image Text:
The formation of molecular hydrogen at 3000 °K proceeds through the following elementary reaction: 2H (g) + Ar (9) H2(g) + Ar (9) k = 2.2 × 108 mol-2.12.s-1 k, 2.2 x 10 mol-1.L.s-1 Write the rate law for this process with respect to H2. (a) Calculate the rate of the reverse reaction (r=-d[H2(g)]/dt) given [H2(g)] = 4.1×103 mol·L¹ and [Ar(g)] = 4.1×10+ mol·L¹; (b) What concentration of Ar is needed to double the reverse reaction rate computed in (a); (c) Calculate the concentration of [H(g)] needed for the forward reaction rate (r=d[H2(g)]/dt) to be equal to the reverse reaction rate computed under the process conditions given in (a). 2) [20] The decomposition of dinitrogen pentoxide at low temperatures results in the formation of a brown-colored gas with the chemical formula N2O4, known as dinitrogen tetroxide, a harmful pollutant found in the air together with nitrogen dioxide. On the basis of the chemical equation 2N205(g) → 2N204(g) + O2(g) a student assumed that the reaction would be second order. Using the following data, determine whether or not the student's assumption is correct. t/s 0 1200 2400 3600 4800 6000 7200 8400 9600 10800 12000 13000 P(N:Os)/(torr) 268.7 247.2 236.2 227.1 217.8 209.5 201.8 193.2 185.8 178.1 164.9 152.4 3) [10] A piston-cylinder system is used to store 1000 J of work by keeping an ideal gas compressed at a pressure of 4 atm. The stored energy is to be released to the surroundings by allowing the piston to expand isothermally at 25°C to a lower pressure of 1.5 atm. Calculate the amount of gas necessary to carry out the energy transfer if the external pressure is 1 atm. 4) [20] Sulfur oxides are pollutants that contribute to the formation of acid rain, as well as particulate pollution. Use the data on the free energy of formation of the oxides of sulfur provided in the table below to determine at what temperature is the formation of SO2(g) and SO3(g) equally thermodynamically favorable? Above what temperature is the S2O(g) the preferred product of the rection of S with O2? Over what temperature range is SO2(g) the preferred product? At high temperatures, will SO2(g) be stable? AG so [kJ/mol] AGs₂o [kJ/mol] T[°K] 0 5.028 -58.859 500 -37.391 - 122.290 AG so₂ [kJ/mol] -294.299 AG so,[kJ/mol] -390.025 -300.871 - 352.668 1000 - 64.382 - 179.508 1500 - 66.854 -217.275 -288.725 -252.239 2000 - 69.294 - 258.325 - 215.929 2500 - 71.708 -301.726 - 179.675 -293.639 -211.247 - 129.768 - 48.855 3000 - 74.111 -346.922 -143.383 31.748 3500 - 76.512 -393.557 - 106.996 112.210 4000 - 78.908 -441.382 -70.484 192.643 4500 -81.291 -490.215 -33.829 273.120 5000 - 83.648 -539.913 2.9821 353.700 5500 - 85.965 -590.360 39.960 434.427 6000 - 88.235 -641.455 77.128 515.359 5) [20] An industrial process generates a waste stream of concentrated sulfuric acid which is neutralized by hydrated lime according to the following highly exothermic reaction to produce gypsum and water Ca(OH) 2 (s) + H2SO4(9) CaSO4(s) + H2O(1) Determine the amount of heat generated when I mole of lime is mixed with 1 mole of waste sulfuric acid in a closed chemical vessel that is immersed in a 2-liter water bath apparatus. If all the heat released during the neutralization reaction is absorbed by the water in the bath apparatus estimate the final temperature of the water. Assume that heat losses are negligible during the energy transfer process and that the initial water temperature was 25°C at 1 atm. The formation of molecular hydrogen at 3000 °K proceeds through the following elementary reaction: 2H (g) + Ar (9) H2(g) + Ar (9) k = 2.2 × 108 mol-2.12.s-1 k, 2.2 x 10 mol-1.L.s-1 Write the rate law for this process with respect to H2. (a) Calculate the rate of the reverse reaction (r=-d[H2(g)]/dt) given [H2(g)] = 4.1×103 mol·L¹ and [Ar(g)] = 4.1×10+ mol·L¹; (b) What concentration of Ar is needed to double the reverse reaction rate computed in (a); (c) Calculate the concentration of [H(g)] needed for the forward reaction rate (r=d[H2(g)]/dt) to be equal to the reverse reaction rate computed under the process conditions given in (a). 2) [20] The decomposition of dinitrogen pentoxide at low temperatures results in the formation of a brown-colored gas with the chemical formula N2O4, known as dinitrogen tetroxide, a harmful pollutant found in the air together with nitrogen dioxide. On the basis of the chemical equation 2N205(g) → 2N204(g) + O2(g) a student assumed that the reaction would be second order. Using the following data, determine whether or not the student's assumption is correct. t/s 0 1200 2400 3600 4800 6000 7200 8400 9600 10800 12000 13000 P(N:Os)/(torr) 268.7 247.2 236.2 227.1 217.8 209.5 201.8 193.2 185.8 178.1 164.9 152.4 3) [10] A piston-cylinder system is used to store 1000 J of work by keeping an ideal gas compressed at a pressure of 4 atm. The stored energy is to be released to the surroundings by allowing the piston to expand isothermally at 25°C to a lower pressure of 1.5 atm. Calculate the amount of gas necessary to carry out the energy transfer if the external pressure is 1 atm. 4) [20] Sulfur oxides are pollutants that contribute to the formation of acid rain, as well as particulate pollution. Use the data on the free energy of formation of the oxides of sulfur provided in the table below to determine at what temperature is the formation of SO2(g) and SO3(g) equally thermodynamically favorable? Above what temperature is the S2O(g) the preferred product of the rection of S with O2? Over what temperature range is SO2(g) the preferred product? At high temperatures, will SO2(g) be stable? AG so [kJ/mol] AGs₂o [kJ/mol] T[°K] 0 5.028 -58.859 500 -37.391 - 122.290 AG so₂ [kJ/mol] -294.299 AG so,[kJ/mol] -390.025 -300.871 - 352.668 1000 - 64.382 - 179.508 1500 - 66.854 -217.275 -288.725 -252.239 2000 - 69.294 - 258.325 - 215.929 2500 - 71.708 -301.726 - 179.675 -293.639 -211.247 - 129.768 - 48.855 3000 - 74.111 -346.922 -143.383 31.748 3500 - 76.512 -393.557 - 106.996 112.210 4000 - 78.908 -441.382 -70.484 192.643 4500 -81.291 -490.215 -33.829 273.120 5000 - 83.648 -539.913 2.9821 353.700 5500 - 85.965 -590.360 39.960 434.427 6000 - 88.235 -641.455 77.128 515.359 5) [20] An industrial process generates a waste stream of concentrated sulfuric acid which is neutralized by hydrated lime according to the following highly exothermic reaction to produce gypsum and water Ca(OH) 2 (s) + H2SO4(9) CaSO4(s) + H2O(1) Determine the amount of heat generated when I mole of lime is mixed with 1 mole of waste sulfuric acid in a closed chemical vessel that is immersed in a 2-liter water bath apparatus. If all the heat released during the neutralization reaction is absorbed by the water in the bath apparatus estimate the final temperature of the water. Assume that heat losses are negligible during the energy transfer process and that the initial water temperature was 25°C at 1 atm.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these finance questions
-
The expected rate of return of an equity index fund is 12% and the standard deviation of return of this fund is 25%. The rate of a Treasury bill is 4% in the market. You have $2,000,000 investment...
-
Density is not routinely measured in the atmosphere. Why is this not a problem for atmospheric scientists?
-
Determine the total amount of tax due and the amount of the alternative minimum tax in each of the following situations: a. Wilbur Corporation's regular tax liability is $180,000, and its tentative...
-
Everton Manufacturing Company had an excellent year. The company hired a new marketing director in January. The new director's great motivational appeal inspired the sales staff, and, as a result,...
-
Solve Problem 8.23 if one end of the shaft is fixed and the other free. Data From Problem 8.23:- Find the natural frequencies for torsional vibration of a fixed-fixed shaft.
-
The July 1, 20X5, trial balance for the Bond Redemption and Interest Debt Service Fund of the County of Hawaii, Hawaii, is presented here. The resources of the fund are committed to debt service. The...
-
Side view of a car is presented in Figure 1. Dimensions of the car side is as follows: ?Length of the Car = (4000 + X) mm ?Height of the Car = (1500 + X) mm ?Wheel Diameter, D = 500 mm ?Width of the...
-
9. Write the static method hidePicture that takes two pictures (source and secret) and hides the secret in source using the algorithm previously discussed, returning the new picture. Add code to main...
-
Assume that you are an observer standing at a point along a three-lane roadway (in one direction). All vehicles in lane 1 are travelling at 40 km/h, all vehicles in lane 2 are travelling at 60 km/h,...
-
The income statement of Baron Inc. reports net sales of $3,749.9 million for the current year. The balance sheet reports accounts receivable, net of $535.3 million at December 31 of the current year...
-
Sabine owns a small rental apartment building and uses an outside professional management company to look for tenants, handle the leases, make repairs and collect rents. During the year, the property...
-
A property sells for a net price of $250,000. Its adjusted basis is $175,000. The terms include closing costs down and a $ 250,000 wraparound contract for deed payable not less than interest only...
-
To the nearest cent, what is the future value of annuity earning 5.8%, when quarterly deposits of $600 are made for 25 years? (Identify or find each) A = d = As a decimal, r = n = t = >> AJ
-
In 2023, Deon and NeNe are married filing jointly. Deon and NeNe's taxable income (all ordinary) is $1,412,000, and they itemize their deductions as follows: real property taxes of $17,500,...
-
Choose companies or industries for which JIT would be totally inappropriate? Explain your reasoning with examples. Choose companies or industries for which JIT would be totally inappropriate Choose...
-
According to a New York Times columnist, The estate tax affects a surprisingly small number of people. In 2003, . . . just 1.25 percent of all deaths resulted in taxable estates, with most of them...
-
Can a firm sustain its operations by maximizing stockholders wealth at the expense of other stakeholders ?
-
In an effort to better understand how her investments are affected by market factors, Michelle Delatorre, the professional tennis player introduced in the Integrative Problem in Chapter, has posed...
-
What type of information can investors get from stock and bond quotation?
-
A simplified model of a petroleum pump is shown in Fig. 1.99, where the rotary motion of the crank is converted to the reciprocating motion of the piston. Find the equivalent mass,...
-
Find the equivalent mass of the system shown in Fig. 1.100. Sphere, mass m k 000 No slip Bell crank lever,- mass moment of inertia Jo 90 k1 m M 00000 x(t) FIGURE 1.100 A bell crank lever connected to...
-
Figure 1.101 shows an offset slider-crank mechanism with a crank length \(r\), connecting rod length \(l\), and offset \(\delta\). If the crank has a mass and mass moment of inertia of \(m_{r}\) and...

Study smarter with the SolutionInn App


