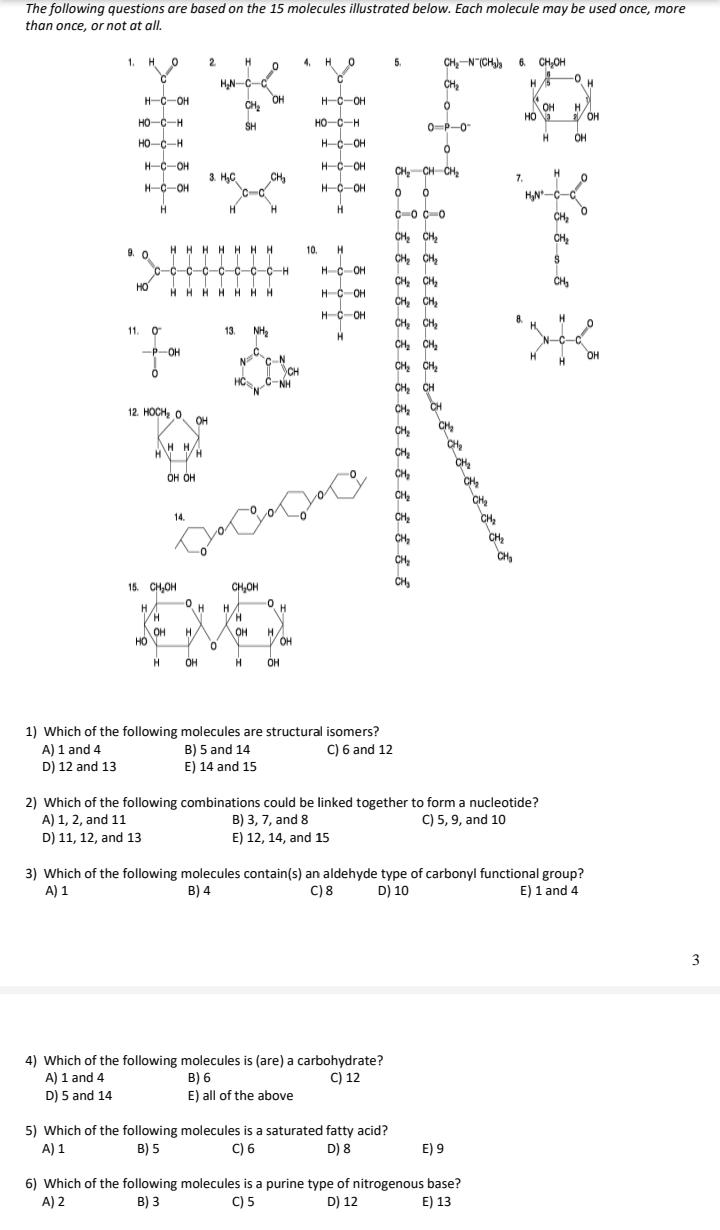
The following questions are based on the 15 molecules illustrated below. Each molecule may be used...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The following questions are based on the 15 molecules illustrated below. Each molecule may be used once, more than once, or not at all. CH,-N (CH, 6. CH,OH CH 1. H O 2 4. H O 5. 0- HN-C-C OH CH H. H-C-OH H-C-OH OH / OH HO-C-H SH HO-C-H 0-P-0 HO-C-H HC-OH H. OH H-C-OH H-C-OH CH, CH CH, 3. H,C CH 7. H-C-OH HC-OH HN-C H H. H. C-o C-0 CH, CH CH2 CH, HHHHHHH 9. 0 c-c-c-c-c-c-c-c-H 10. CH CH2 HC-OH CH CH, CH, CH, CH, CH, CH, HO CH HHHH HHH HC-OH H-C-OH H. 11. 0 13. CH, CH, CH, CH, CH -P-OH C. N C-N CH C-NH OH 12. HOCH, O CH, CH OH CH, H H/ CH, CH, CH, OH OH CH CH, CH, CH, 14. CH, CH, CH 15. CH,OH CH,OH H. H. H OH HO H. H. V OH OH OH H. OH 1) Which of the following molecules are structural isomers? B) 5 and 14 E) 14 and 15 A) 1 and 4 D) 12 and 13 C) 6 and 12 2) Which of the following combinations could be linked together to form a nucleotide? B) 3, 7, and 8 E) 12, 14, and 15 A) 1, 2, and 11 C) 5, 9, and 10 D) 11, 12, and 13 3) Which of the following molecules contain(s) an aldehyde type of carbonyl functional group? C) 8 A) 1 B) 4 D) 10 E) 1 and 4 4) Which of the following molecules is (are) a carbohydrate? B) 6 E) all of the above C) 12 A) 1 and 4 D) 5 and 14 5) Which of the following molecules is a saturated fatty acid? C) 6 A) 1 B) 5 D) 8 E) 9 6) Which of the following molecules is a purine type of nitrogenous base? C) 5 A) 2 B) 3 D) 12 E) 13 The following questions are based on the 15 molecules illustrated below. Each molecule may be used once, more than once, or not at all. CH,-N (CH, 6. CH,OH CH 1. H O 2 4. H O 5. 0- HN-C-C OH CH H. H-C-OH H-C-OH OH / OH HO-C-H SH HO-C-H 0-P-0 HO-C-H HC-OH H. OH H-C-OH H-C-OH CH, CH CH, 3. H,C CH 7. H-C-OH HC-OH HN-C H H. H. C-o C-0 CH, CH CH2 CH, HHHHHHH 9. 0 c-c-c-c-c-c-c-c-H 10. CH CH2 HC-OH CH CH, CH, CH, CH, CH, CH, HO CH HHHH HHH HC-OH H-C-OH H. 11. 0 13. CH, CH, CH, CH, CH -P-OH C. N C-N CH C-NH OH 12. HOCH, O CH, CH OH CH, H H/ CH, CH, CH, OH OH CH CH, CH, CH, 14. CH, CH, CH 15. CH,OH CH,OH H. H. H OH HO H. H. V OH OH OH H. OH 1) Which of the following molecules are structural isomers? B) 5 and 14 E) 14 and 15 A) 1 and 4 D) 12 and 13 C) 6 and 12 2) Which of the following combinations could be linked together to form a nucleotide? B) 3, 7, and 8 E) 12, 14, and 15 A) 1, 2, and 11 C) 5, 9, and 10 D) 11, 12, and 13 3) Which of the following molecules contain(s) an aldehyde type of carbonyl functional group? C) 8 A) 1 B) 4 D) 10 E) 1 and 4 4) Which of the following molecules is (are) a carbohydrate? B) 6 E) all of the above C) 12 A) 1 and 4 D) 5 and 14 5) Which of the following molecules is a saturated fatty acid? C) 6 A) 1 B) 5 D) 8 E) 9 6) Which of the following molecules is a purine type of nitrogenous base? C) 5 A) 2 B) 3 D) 12 E) 13
Expert Answer:

Related Book For 

Intermediate accounting
ISBN: 978-0077647094
7th edition
Authors: J. David Spiceland, James Sepe, Mark Nelson
Posted Date:
Students also viewed these chemical engineering questions
-
The following questions are based on the 15 molecules illustrated below. Each molecule may be used once, more than once, or not at all. 1. H O 2 4. H O 5. CH,-N (CH, 6 CH,OH CH 0- HN-C-C OH CH H....
-
The following questions are adapted from a variety of sources including questions developed by the AICPA Board of Examiners and those used in the Kaplan CPA Review Course to study property, plant,...
-
The following questions are adapted from a variety of sources including questions developed by the AICPA Board of Examiners and those used in the Kaplan CPA Review Course to study property, plant,...
-
What is true about business cycles? Multiple select question. They vary in duration and intensity. They follow an identical pattern. They are made up of alternating rises and declines. "Ups" are...
-
What is meant by an issue or issuer being placed on a credit watch?
-
Calculate the rate of cooling (kW) required to bring 300 kg/mm of carbon monoxide from 450C to 50C, performing the calculation (a) Using Table B.2 and (b) Using Table B.8. Considering how much...
-
What ethical standards paralegals must follow?
-
Into what two categories does the FASAB divide government assets? How are each of the two accounted for?
-
Consider a standing normal shock in a steady one-dimensional ow from left to right. Sketch, on an x-t diagram, the shock itself, particle paths and both families of characteristics in the...
-
Passion Company is trying to decide whether or not to acquire Desiree Inc. The following balance sheet for Desiree Inc. provides information about book values. Estimated market values are also...
-
Given a state or local government describe the accounting standards that apply to Colleges and Universities and Health Care Organizations. Demonstrate the correct financial reporting as mandated by...
-
Evaluate the role of Six Sigma methodologies in achieving operational excellence and reducing process defects, focusing on the DMAIC (Define, Measure, Analyze, Improve, Control) framework and its...
-
Discuss the implementation of quality management systems (QMS), such as ISO 9001, in organizations, highlighting the importance of process standardization, continual improvement, and internal audits...
-
Assuming a 12% annual interest rate, determine the present value of a five-period annual annuity of $6.200 under each of the following situations: Note: Use tables, Excel, or a financial calculator....
-
Evaluate the thermodynamic properties and behavior of complex fluids, such as liquid crystals, surfactant solutions, and polymeric materials, in terms of phase transitions, self-assembly phenomena,...
-
Discuss the principles of process intensification (PI) and its role in achieving sustainable and efficient chemical processing, focusing on the reduction of equipment size, energy consumption, and...
-
3. Let f(x, y, z) = x - 23 = and let v (5,2,1). 2y a) Find the directional derivative of the function f at the point (1,2,3) in the direction of v. That is, compute Duf(1,2,3), where u is a unit...
-
Provide a draft/outline of legal research involving an indigenous Canadian woman charged with assault causing bodily harm under (Sec 267b) of the Criminal Code, where the crown wants a 12-month jail...
-
At the beginning of 2013, a company adopts the dollar-value LIFO inventory method for its one inventory pool. The pools value on that date was $1,400,000. The 2013 ending inventory valued at year-end...
-
At December 31, 2013, the financial statements of Hollingsworth Industries included the following: Net income for 2013 ...................... $560 million Bonds payable,10%, convertible into 36...
-
During 2013, WMC Corporation discovered that its ending inventories reported on its financial statements were misstated by the following amounts: 2011 . understated by . $120,000 2012 . overstated by...
-
A written representation from a clients management which, among other matters, acknowledges responsibility for the fair presentation of financial statements, should normally be signed by the a. Chief...
-
Which of the following expressions is least likely to be included in a clients representation letter? a. No events have occurred subsequent to the balance sheet date that require adjustments to, or...
-
What are computer-assisted audit programs?

Study smarter with the SolutionInn App


