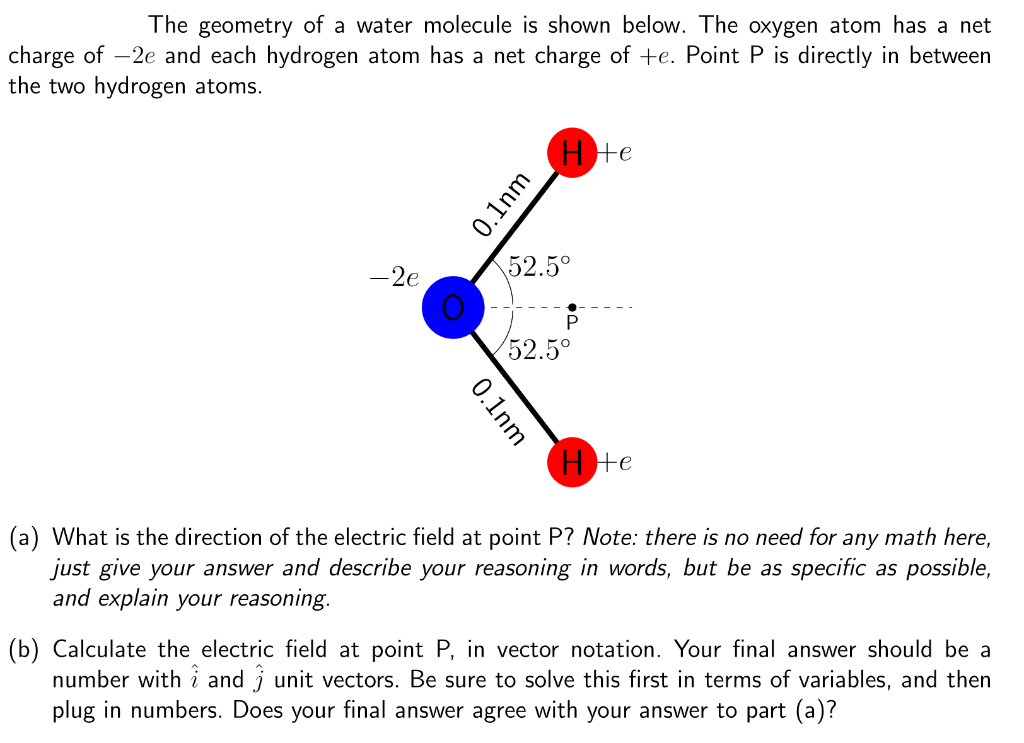
The geometry of a water molecule is shown below. The oxygen atom has a net charge...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The geometry of a water molecule is shown below. The oxygen atom has a net charge of -2e and each hydrogen atom has a net charge of +e. Point P is directly in between the two hydrogen atoms. - 2e 0.1nm 0.1nm Hote 52.5⁰ P 52.5⁰ He (a) What is the direction of the electric field at point P? Note: there is no need for any math here, just give your answer and describe your reasoning in words, but be as specific as possible, and explain your reasoning. (b) Calculate the electric field at point P, in vector notation. Your final answer should be a number with and unit vectors. Be sure to solve this first in terms of variables, and then plug in numbers. Does your final answer agree with your answer to part (a)? The geometry of a water molecule is shown below. The oxygen atom has a net charge of -2e and each hydrogen atom has a net charge of +e. Point P is directly in between the two hydrogen atoms. - 2e 0.1nm 0.1nm Hote 52.5⁰ P 52.5⁰ He (a) What is the direction of the electric field at point P? Note: there is no need for any math here, just give your answer and describe your reasoning in words, but be as specific as possible, and explain your reasoning. (b) Calculate the electric field at point P, in vector notation. Your final answer should be a number with and unit vectors. Be sure to solve this first in terms of variables, and then plug in numbers. Does your final answer agree with your answer to part (a)?
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
A water molecule is shown schematically in FIGURE 3-40. The distance from the center of the oxygen atom to the center of a hydrogen atom is 0.96 Ã, and the angle between the hydrogen atoms is...
-
What is the mass of a water molecule in atomic mass units?
-
The geometry of a compound microscope, which consists of two converging lenses, is shown in Fig. 23.29. (More detail on microscopes is given in Chapter 25.) The objective lens and the eyepiece lens...
-
Harry Bhel carries a business as a sole proprietorship. During its 2022 fiscal period, its first year of operations, the business had cash sales of $123,000. It also has sales on account of $46,000,...
-
Describe the events of mitosis in sequence.
-
Your baby cousin Hubert loves lollipops and bouncy balls. Look at the indifference curves in Figure EP-1 that represent his preferences among various bundles of the two goods. Assuming the...
-
What stakeholder affected by their behavior was Gilead weighting very lightly when it decided what to tell the FDA about the medicines it wished to have permission to sell?
-
On June 10, Naveen Company purchased $8,000 of merchandise from Jarrah Company, FOB shipping point, terms 2/10, n/30. Naveen pays the freight costs of $400 on June 11. Damaged goods totaling $300 are...
-
Development economics studies the transformation of emerging nations into more prosperous one and it seeks to understand and shape the country's macro and microeconomics policies in order to lift...
-
Ross Gellar has come to you for some help in computing his income from his investments. He is trying to plan for the payment of his 2019 taxes and wants an idea as to how much that tax bill will be....
-
IRS. a. Indicate whether the following statements are "True" or "False" regarding the reporting responsibilities Abby Sue might have as church treasurer. 1.) Filing Form 1023 [Application for...
-
Identify at least 4 different stakeholders that operate within the real estate environment in relation to the role they fulfill developer rental agent managing agent engine etc.
-
I have this data frame with the years and values, can you make a model to predict the value of meats,fish,fruits and vegetables and grains? based on the current values of the table and historical...
-
Alicia's Accessories is a global manufacturer of jewelry and hair accessories. The company currently relies on the COSO Internal Control-Integrated Framework to manage its corporate governance and...
-
11 in 6-8 Consider the structure below. You may also refer to Atachment C for better clarity on dimensions. BEDROOM 13-9 BEDROOM -TAAN VAIN ATTACHMENT C 6-8 BATH L 5-9 KITCHEN 5 DINING W/D 955 sq ft...
-
Draw the diagram for the decoder question below ( ( Digital System ) ) :b . . Decoders 3 - to 8 : Combine two 3 - to 8 decoders to create a 4 - to 1 6 decoder. Connect inputs A and B to the...
-
Wood Products Inc. expects to have a cash balance of $25,000 on January 1. Relevant information is as follows (See image below): Required (a) Prepare a cash budget for January, February, and March....
-
Why do markets typically lead to an efficient outcome for buyers and sellers?
-
The temperature dependence of the diffusion coefficient D (cm2/s) is given by an Arrhenius type equation: D = D0e(- Ea/RT) where D0 (cm2/s) is pre-exponential constant, Ea (J/mol) is activation...
-
A simply supported beam is subjected to distributed loads w1 and w2 as shown. The bending moment as a function of x is given by the following equations: M(x) = RAx W1x2/2 for 0 M(x) = RAx w1a/2 (2x ...
-
Create three row vectors: a = [3 -1 5 11 -4 2] b = [7 -9 2 13 1 -2] c = [-2 4 -7 8 0 9] (a) Use the three vectors in a MATLAB command to create a 3 4 matrix such that the first, second, and third...
-
A reaction in which the pre-exponential term is independent of temperature is found to be a 100 times faster at \(200^{\circ} \mathrm{C}\) than it is at \(25^{\circ} \mathrm{C}\). Calculate the...
-
The rate of formation of nitric oxide (NO) is controlled by the three reversible chemical reactions Use the steady state approximation for the nitrogen atom concentration and the assumption of...
-
The structure of ethylene is \(\mathrm{H}_{2} \mathrm{C}=\mathrm{CH}_{2}\). Estimate the enthalpy of reaction when \(1 \mathrm{kmol}\) of ethylene is completely oxidised. Compare the value obtained...

Study smarter with the SolutionInn App


