The mechanism for the decomposition of NO2Cl is: By making a steady-state approximation for [Cl], express the
Fantastic news! We've Found the answer you've been seeking!
Question:
The mechanism for the decomposition of NO2Cl is:
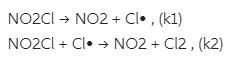
By making a steady-state approximation for [Cl], express the rate of appearance of Cl2 in terms of the concentrations of NO2Cl and NO2.
By making a steady-state approximation for [Cl], express the rate of appearance of Cl2
in terms of the concentrations of NO2Cl and NO2.
By making a steady-state approximation for [Cl], express the rate of appearance of Cl2
in terms of the concentrations of NO2Cl and NO2.
Related Book For 

Posted Date:




