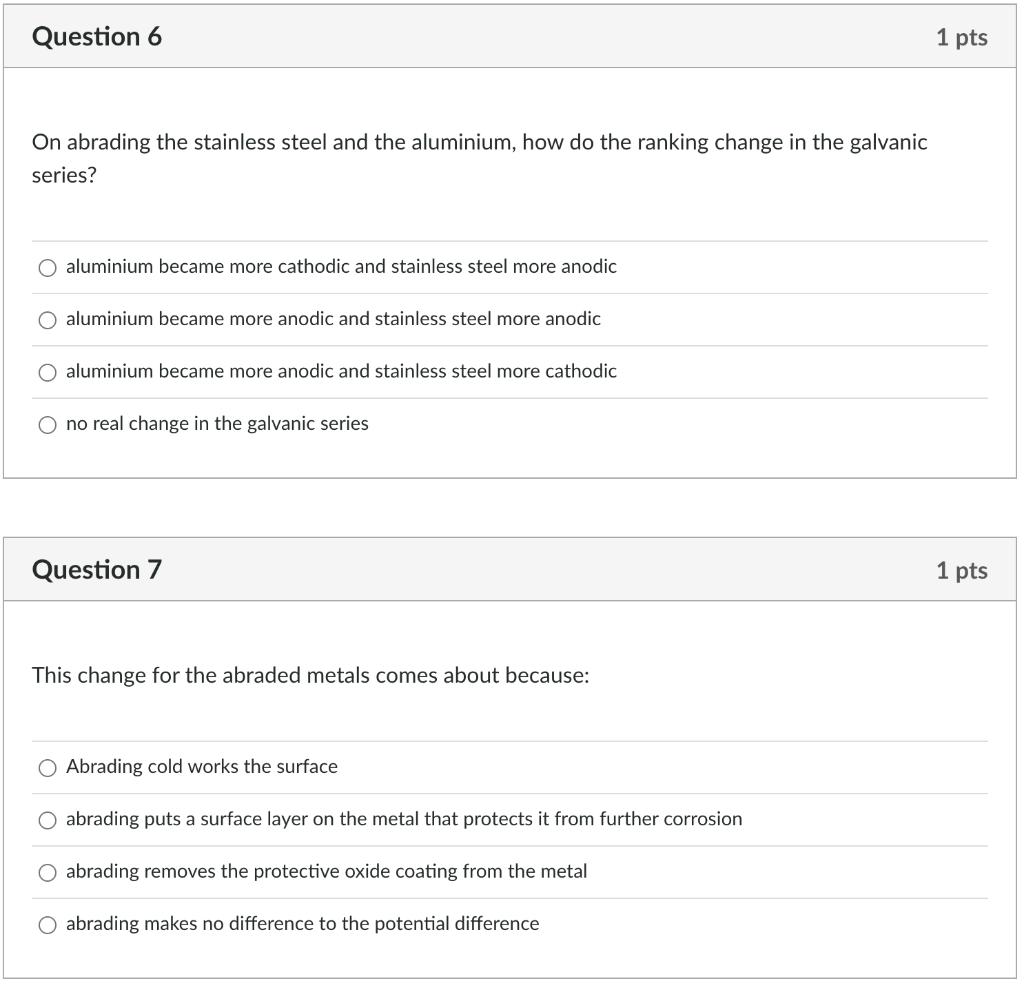
Question 6 On abrading the stainless steel and the aluminium, how do the ranking change in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
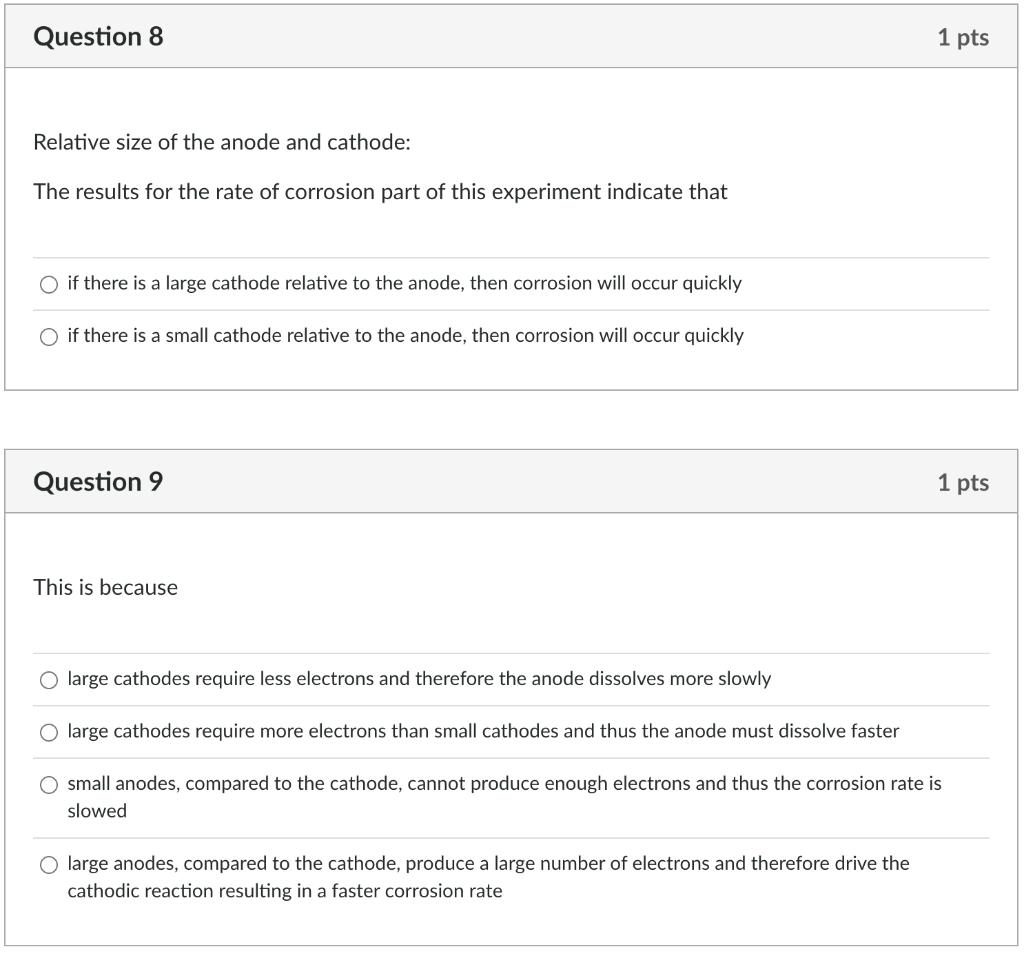
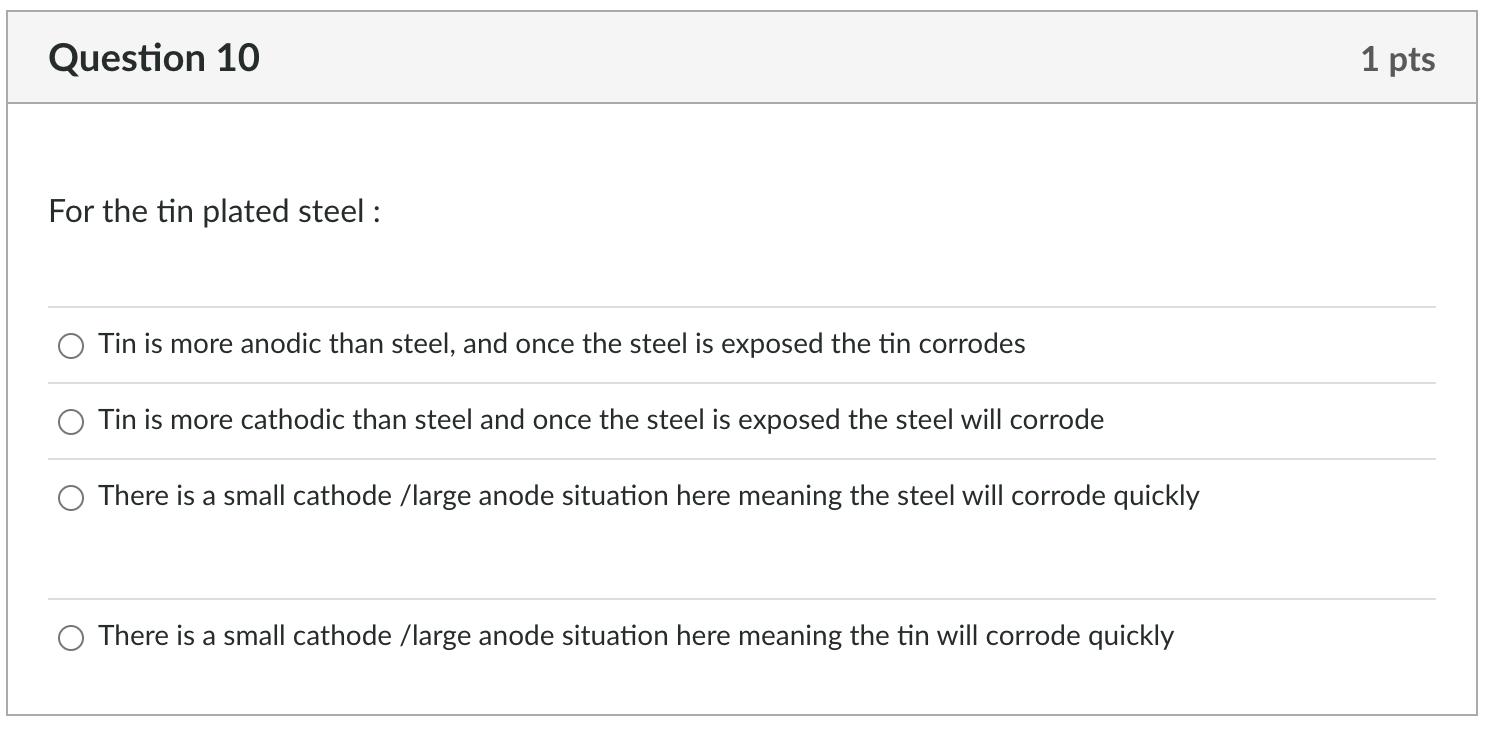
Question 6 On abrading the stainless steel and the aluminium, how do the ranking change in the galvanic series? O aluminium became more cathodic and stainless steel more anodic aluminium became more anodic and stainless steel more anodic O aluminium became more anodic and stainless steel more cathodic no real change in the galvanic series Question 7 This change for the abraded metals comes about because: O Abrading cold works the surface abrading puts a surface layer on the metal that protects it from further corrosion abrading removes the protective oxide coating from the metal O abrading makes no difference to the potential difference 1 pts 1 pts Question 8 Relative size of the anode and cathode: The results for the rate of corrosion part of this experiment indicate that O if there is a large cathode relative to the anode, then corrosion will occur quickly if there is a small cathode relative to the anode, then corrosion will occur quickly Question 9 This is because 1 pts O large anodes, compared to the cathode, produce a large number of electrons and therefore drive the cathodic reaction resulting in a faster corrosion rate 1 pts O large cathodes require less electrons and therefore the anode dissolves more slowly large cathodes require more electrons than small cathodes and thus the anode must dissolve faster small anodes, compared to the cathode, cannot produce enough electrons and thus the corrosion rate is slowed Question 10 For the tin plated steel: Tin is more anodic than steel, and once the steel is exposed the tin corrodes Tin is more cathodic than steel and once the steel is exposed the steel will corrode There is a small cathode /large anode situation here meaning the steel will corrode quickly There is a small cathode /large anode situation here meaning the tin will corrode quickly 1 pts Question 6 On abrading the stainless steel and the aluminium, how do the ranking change in the galvanic series? O aluminium became more cathodic and stainless steel more anodic aluminium became more anodic and stainless steel more anodic O aluminium became more anodic and stainless steel more cathodic no real change in the galvanic series Question 7 This change for the abraded metals comes about because: O Abrading cold works the surface abrading puts a surface layer on the metal that protects it from further corrosion abrading removes the protective oxide coating from the metal O abrading makes no difference to the potential difference 1 pts 1 pts Question 8 Relative size of the anode and cathode: The results for the rate of corrosion part of this experiment indicate that O if there is a large cathode relative to the anode, then corrosion will occur quickly if there is a small cathode relative to the anode, then corrosion will occur quickly Question 9 This is because 1 pts O large anodes, compared to the cathode, produce a large number of electrons and therefore drive the cathodic reaction resulting in a faster corrosion rate 1 pts O large cathodes require less electrons and therefore the anode dissolves more slowly large cathodes require more electrons than small cathodes and thus the anode must dissolve faster small anodes, compared to the cathode, cannot produce enough electrons and thus the corrosion rate is slowed Question 10 For the tin plated steel: Tin is more anodic than steel, and once the steel is exposed the tin corrodes Tin is more cathodic than steel and once the steel is exposed the steel will corrode There is a small cathode /large anode situation here meaning the steel will corrode quickly There is a small cathode /large anode situation here meaning the tin will corrode quickly 1 pts
Expert Answer:
Answer rating: 100% (QA)
Question 6 Answer Aluminum become more anodic and stainless steel become mo... View the full answer

Related Book For 

Concepts in Federal Taxation
ISBN: 9780324379556
19th Edition
Authors: Kevin E. Murphy, Mark Higgins, Tonya K. Flesher
Posted Date:
Students also viewed these chemistry questions
-
The Lazy Lion Restaurant was owned and operated by Leon Lowe. Although the property upon which the restaurant was located was owned by Lowe, it was also subject to a mortgage in favour of Ursula...
-
The Hartford Group is a partnership owned and operated by June and Joyce. June owns a 60% interest, and her basis in the partnership is $33,000. Joyce owns a 40% interest, and her basis is $18,000....
-
Pyramid Company is a small rug retailer owned and operated by Rosemary Endecott. After the accounts have been adjusted on January 31, the following selected account balances were taken from the...
-
Look again at the financial forecasts for Growth-Tech given in Table 4.3. This time assume you know that the opportunity cost of capital is r = .12 (discard the .099 figure calculated in the text)....
-
Find the PSD of the process Let Wn be an IID sequence of zero- mean Gaussian random variables with variance. Define a discrete- time random process, X[ n] = pX[ n 1]+ Wn, n = 1, 2, 3, where X[ 0] =...
-
When might a firm desire to locate close to its suppliers? Its customers?Somewhere in-between? What are the impacts on cost and customer service?
-
Owing to economical sample sizes and ease of application, probability-proportional to-size (PPS) sampling has become the most widely used statistical sampling plan in practice today, even by local...
-
Podsednik Company has gathered the following information Units in beginning work in process ..............0 Units started into production ..................36,000 Units in ending work in process...
-
You allow yourself 46 min to drive 23mi to the airport, but you're caught in heavy traffic and average only 15mi/ for the first 13min. a) what must your average speed be on the rest of the trip if...
-
Glenn Foreman, president of Oceanview Development Corporation, is considering submitting a bid to purchase property that will be sold by sealed bid at a county tax foreclosure. Glenns initial...
-
Let V = R be equipped with the operations x1 X2 ko x1 x2. Y Y2 = = - 3 x + Y x2 + y2 + 1 kx - 3k +3] kx + k-1 Prove, by definition (showing all axioms hold in general) that V is a vector space. Note:...
-
In what ways do the characteristics of small firms impact upon the marketing approaches and activities that they adopt?
-
How can community entrepreneurship benefit from the business, voluntary and public sectors?
-
What strategies are associated with success in small businesses?
-
Where do you think community entrepreneurship is more relevant in poor or rich communities? In urban or rural communities?
-
Why do you think some entrepreneurs engage in community entrepreneurship rather than using their skills and effort to create a commercial venture?
-
Frivolous, Inc., recently purchased an asset that will be very effective in the early years of its useful life, but the effectiveness of which will diminish significantly as maintenance charges...
-
Determine whether the lines are parallel, perpendicular, or neither. 2x + 3y = -12, 2y - 3x = 8
-
The New Tech Corporation contributes some of its inventory of scientific equipment to the computer department of Great University during the current year. At the date of the contribution, the...
-
ABC Company owns a chain of furniture stores. How much loss can ABC Company deduct in each of the following cases? Explain. a. ABC closes a store in a depressed part of the county. Rather than move...
-
Larry is a professional gambler, specializing in dog racing. He spends 50 to 60 hours per week studying racing forms and placing bets at the track. During the current year, Larry has winnings of...
-
Explain how objects interact by exchanging messages.
-
Describe the relationship between dynamic modeling, behavioral modeling, and structural modeling.
-
Comment on this statement: Dynamic modeling is about interaction.

Study smarter with the SolutionInn App


