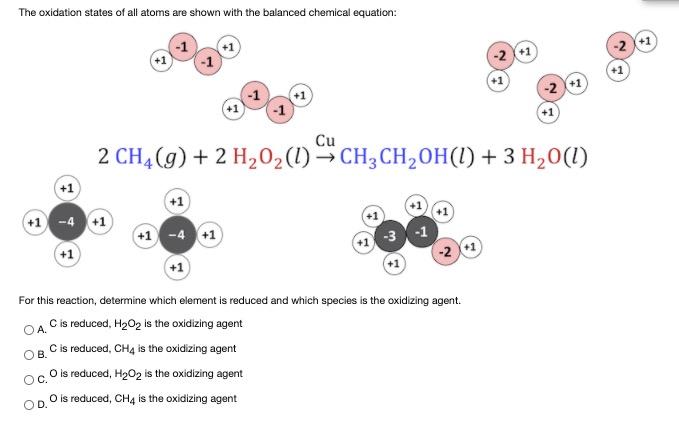
The oxidation states of all atoms are shown with the balanced chemical equation: +1 -2 +1...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The oxidation states of all atoms are shown with the balanced chemical equation: +1 -2 +1 -2 (+1 (+1 -1 -2 +1 Cu 2 CH4 (g) + 2 H202(1)→CH;CH,OH(I) + 3 H20(1) (+1 -4 +1 +1 -4 +1 -3 (+1) -2 +1 (+1 For this reaction, determine which element is reduced and which species is the oxidizing agent. OA Cis reduced, H202 is the oxidizing agent C is reduced, CH4 is the oxidizing agent OB. O is reduced, H202 is the oxidizing agent OC. O is reduced, CH4 is the oxidizing agent The oxidation states of all atoms are shown with the balanced chemical equation: +1 -2 +1 -2 (+1 (+1 -1 -2 +1 Cu 2 CH4 (g) + 2 H202(1)→CH;CH,OH(I) + 3 H20(1) (+1 -4 +1 +1 -4 +1 -3 (+1) -2 +1 (+1 For this reaction, determine which element is reduced and which species is the oxidizing agent. OA Cis reduced, H202 is the oxidizing agent C is reduced, CH4 is the oxidizing agent OB. O is reduced, H202 is the oxidizing agent OC. O is reduced, CH4 is the oxidizing agent
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The oxidation states of the halogens vary from -1 to +7. Identify compounds of chlorine that have -1, +1, +3, +5, and +7 oxidation states. How does the oxyacid strength of the halogens vary as the...
-
Which species in each pair is a better oxidizing agent under standard-state conditions? (a) Br2 or Au3+. (b) H2 or Ag+. (c) Cd2+ or Cr3+. (d) O2 in acidic media or O2 in basic media.
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
__________ is the process that occurs when a job applicant does research on an organization and concludes there is enough of a fit between themselves and a job opening to warrant submitting their...
-
From Eqs. 13.23 and 13.24 and the knowledge that Cp > Cv what can you conclude about the slopes of constant v and constant P curves in a T-s diagram? Notice that we are looking at functions T(s, P or...
-
A coal suspension is found to behave as a power law fluid, with a flow index of 0.4 , a specific gravity of 1.5 , and an apparent viscosity of $90 \mathrm{cP}$ at a shear rate of $100...
-
What are the three factors that affect lateral stability?
-
The following data are for four independent process-costing departments. Inputs are added continuously. Required: Compute the equivalent units of production for each of the preceding departments...
-
If f(x)=(8x3+9) and g(x) = -(9x + 19), then d dx [(2)-9(2)] -0
-
6.2. Data has been collected from a chemical reactor. The inlet concentration was the only input variable that changed when the data was collected. The input and output data is given in Table Q6.2....
-
Show that a paraboloidal wave centered at the point P1 is converted by a lens of focal length f into a paraboloidal wave centered about P2, where 1/z1+1/z2=1/f. [hint: using U1, U2 and t] Pz P1 Z1
-
Diana Corporation provides the following information for 2017: Calculate the total manufacturing costs incurred in 2017. Beginning inventory of direct materials, 1/1/2017 Purchases of direct...
-
Donna Corporation manufactures custom cabinets for kitchens. It uses a normalcosting system with two direct-cost categoriesdirect materials and direct manufacturing laborand one indirect-cost pool,...
-
Let us continue with the above illustration of Jai Investment and Finance Ltd. Assume that the company disposed off its holding to Imam Saheb Ltd. on a cum-right basis @ Rs. 846 per share. Imam Saheb...
-
If Usha Corporation Ltd. were to continue with the classification of its investment in Bhonsle as current investments, and the fair market value as on the balance sheet date was Rs. 42,07,500, at...
-
Usha Corporation Ltd. purchases 12,000 10% secured redeemable bonds of the face value of Rs. 100 each of Andhra Pradesh Power Finance Corporation Ltd. on 1st January for a consideration of Rs....
-
20. What is the output of the following program segment? (4) long long num = 5; int i; for (i = 1; i < 3; i++) num = num + num * (num - i); bob cout < < num < < " "; "; rollet ad lo } 10 cout < <...
-
What types of questions can be answered by analyzing financial statements?
-
A glass vessel contains 28 g nitrogen gas. Assuming ideal behavior, which of the processes listed below would double the pressure exerted on the walls of the vessel? a. Adding enough mercury to fill...
-
Consider the following enthalpy changes: F2(g) + HF(g) FHF2(g) ÎH = 155 kJ/ mol H2O(g) + HOH(g) H2O---HOH (in ice) ÎH = 21 kJ/ mol How do the strengths of hydrogen bonds vary with the...
-
A chemical brearhalyzer test works because ethyl alco¬hol in the breath is oxidized by the dichromate ion (orange) to form acetic acid and chromium(III) ion (green). The balanced reaction is You...
-
Option price relations can be seen by looking at real-time pricing, market data, and data for exchange-traded call and put options found on OMON, CALL, and PUT. Using the OMON screen for selected a...
-
Suppose XYZ stock is trading at \(\$ 60\), XYZ 60 European puts expiring in one year are trading at \(\$ 2\), and the annual risk-free rate is \(3 \%\). Using the put-call parity model, determine the...
-
Suppose XYZ stock is trading \(\$ 60, X Y Z\) 6o European calls expiring in one year are trading at \(\$ 3\), and the annual risk-free rate is \(3 \%\). Using the put-call parity model determine the...

Study smarter with the SolutionInn App


