The rate of effusion of unknown gas was measured and found to be 34.9mL/min. Under the...
Fantastic news! We've Found the answer you've been seeking!
Question:

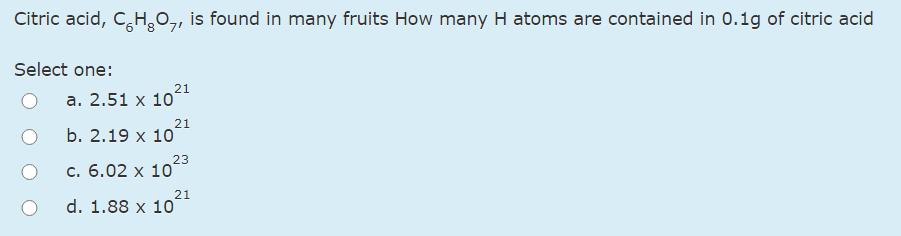

Transcribed Image Text:
The rate of effusion of unknown gas was measured and found to be 34.9mL/min. Under the same conditions, the rate of effusion of o, was found to be 24.6mL/min. Calculate the molar mass of the unknown gas (in g/mol). Select one: а. 54.2 b. 15.9 с. 37.9 d. 43.9 Citric acid, C,H,0,, is found in many fruits How many H atoms are contained in 0.1g of citric acid Select one: 21 а. 2.51 х 10 21 b. 2.19 x 10 23 c. 6.02 x 10 21 d. 1.88 x 10 In the following reaction, what ions, if any, are spectators ions? → AgNO, + KI AgI + 2KNO, Select one: а. К NO (aq)' 3(aq) b. Ag NO (aq)' 3(aq) C. Ag (aq)' (aq) I, + d. K Ag (aq)' (aq) е. К I (aq)' - (aq) The rate of effusion of unknown gas was measured and found to be 34.9mL/min. Under the same conditions, the rate of effusion of o, was found to be 24.6mL/min. Calculate the molar mass of the unknown gas (in g/mol). Select one: а. 54.2 b. 15.9 с. 37.9 d. 43.9 Citric acid, C,H,0,, is found in many fruits How many H atoms are contained in 0.1g of citric acid Select one: 21 а. 2.51 х 10 21 b. 2.19 x 10 23 c. 6.02 x 10 21 d. 1.88 x 10 In the following reaction, what ions, if any, are spectators ions? → AgNO, + KI AgI + 2KNO, Select one: а. К NO (aq)' 3(aq) b. Ag NO (aq)' 3(aq) C. Ag (aq)' (aq) I, + d. K Ag (aq)' (aq) е. К I (aq)' - (aq)
Expert Answer:
Answer rating: 100% (QA)
1 Answer Rate2 Rate1 M1 M2 Where Rate2 Rate of unknown gas 349mLmin Rate1 Rate of O2 gas 246mLmin ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The rate of effusion of unknown gas was measured and found to be 34.9mL/min. Under the same conditions, the rate of effusion of O, was found to be 24.6mL/min. Calculate the molar mass of the unknown...
-
The rate of effusion of a particular gas was measured to be 24.0 mL/ min. Under the same conditions, the rate of effusion of pure methane gas (CH 4 ) is 47.8 mL/ min. What is the molar mass of the...
-
The radius of a sphere was measured and found to be 18 cm with a possible error in measurement of at most 0.01 cm. What is the maximum error in using this value of the radius to compute the volume of...
-
What is the leading cause of death among people with HIV / AIDS in Africa?
-
Why is it important to include a placebo in a scientific study to assess the effectiveness of a drug?
-
The summarized accounts of Hope (Eternal Springs) Ltd for the years 2015 and 2016 are given below. Inventory at 1 January 2015 was 50,000. Required: (a) Calculate the following ratios for 2015 and...
-
Determine whether each of the following is true or false. In each case, assume \(P\) is located at \(t=0\) and \(F\) is located at \(t=n\), and the \(A\) s are spread uniformly over the planning...
-
Peter Rourke, a loan processor at Wentworth Bank, has been timed performing four work elements, with the results shown in the following table. The allowances for tasks such as this are personal, 7%;...
-
10. Label and explain in details the following diagram A 12. 1 Give three functions of E. Zinc E B + C D Copper 13. Give an ideal principle of predicting a performance of a fuel cell and give an...
-
a. During February, $194,500 was paid to creditors on account, and purchases on account were $210,400. Assuming that the February 28 balance of Accounts Payable was $62,500, determine the account...
-
Suppose you finance a $10,000 car over 60 months at an interest rate of 1% per month. 1. How much is your monthly car payment? (Assume that the first payment is one month from now.) 2. How much is...
-
Tax costs may be avoided in its entirety when the transaction is structured as what type of reorganization?
-
Why is EEG equipment inexpensive and portable compared to other neuroimaging techniques?
-
What activity would have triggered red flags? Describe the transactional activity.
-
Why would a company want to use account numbers on its Chart of Accounts?
-
What years did the US begin to use waste management reduction and prevention rather than control?
-
Based on the information on this website, how you can explain about Situation Analysis for this product...
-
Interview managers at three companies in your area about their use of ERP. How have their experiences been similar? What accounts for the similarities and differences?
-
Label the following hydrides as ionic, covalent, or interstitial, and support your answer. The light blue atoms are hydrogen atoms. (b) (c) (a)
-
The melting point of a fictional substance X is 225oC at 10.0 atm. If the density of the solid phase of X is 2.67 g/cm3 and the density of the liquid phase is 2.78 g/cm3 at 10.0 atm, predict whether...
-
It takes 208.4 kJ of energy to remove 1 mole of electrons from the atoms on the surface of rubidium metal. If rubidium metal is irradiated with 254-nm light, what is the maximum kinetic energy the...
-
Solve the matrix equation \([\mathbf{K}]\{\mathbf{Q}\}=\{\mathbf{F}\}\) using forward reduction and backward substitution, where, 5-10 [K] = -13-1 0-1 1 {F}= 0
-
Volunteer work can improve your skills and enhance your rsum. With a partner, seek volunteer opportunities on your campus or in your city. Make a list of volunteer groups that may need help. Here are...
-
What are six brainstorming questions to consider before starting your proposal?

Study smarter with the SolutionInn App


