The standard heat of combustion ( AH, ) of liquid 2,3,3-trimethylpentane [C3H18] is reported in a...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
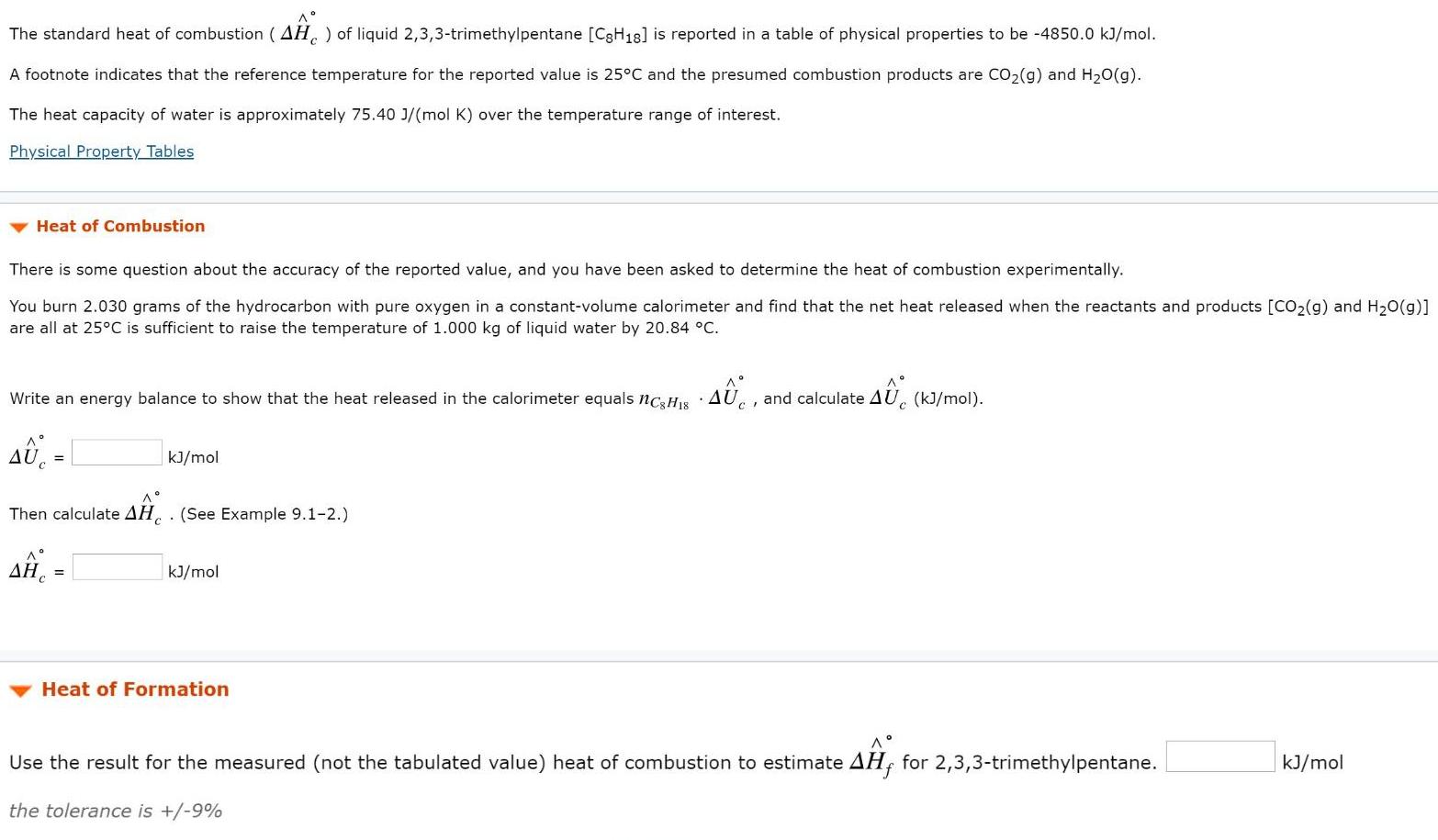
The standard heat of combustion ( AH, ) of liquid 2,3,3-trimethylpentane [C3H18] is reported in a table of physical properties to be -4850.0 kJ/mol. A footnote indicates that the reference temperature for the reported value is 25°C and the presumed combustion products are CO2(g) and H20(g). The heat capacity of water is approximately 75.40 J/(mol K) over the temperature range of interest. Physical Property Tables Heat of Combustion There is some question about the accuracy of the reported value, and you have been asked to determine the heat of combustion experimentally. You burn 2.030 grams of the hydrocarbon with pure oxygen in a constant-volume calorimeter and find that the net heat released when the reactants and products [CO2(g) and H20(g)] are all at 25°C is sufficient to raise the temperature of 1.000 kg of liquid water by 20.84 °C. Write an energy balance to show that the heat released in the calorimeter equals nc, Hs and calculate AU. (k)/mol). kJ/mol Then calculate AH . (See Example 9.1-2.) kJ/mol Heat of Formation A° Use the result for the measured (not the tabulated value) heat of combustion to estimate AH, for 2,3,3-trimethylpentane. kJ/mol the tolerance is +/-9% The standard heat of combustion ( AH, ) of liquid 2,3,3-trimethylpentane [C3H18] is reported in a table of physical properties to be -4850.0 kJ/mol. A footnote indicates that the reference temperature for the reported value is 25°C and the presumed combustion products are CO2(g) and H20(g). The heat capacity of water is approximately 75.40 J/(mol K) over the temperature range of interest. Physical Property Tables Heat of Combustion There is some question about the accuracy of the reported value, and you have been asked to determine the heat of combustion experimentally. You burn 2.030 grams of the hydrocarbon with pure oxygen in a constant-volume calorimeter and find that the net heat released when the reactants and products [CO2(g) and H20(g)] are all at 25°C is sufficient to raise the temperature of 1.000 kg of liquid water by 20.84 °C. Write an energy balance to show that the heat released in the calorimeter equals nc, Hs and calculate AU. (k)/mol). kJ/mol Then calculate AH . (See Example 9.1-2.) kJ/mol Heat of Formation A° Use the result for the measured (not the tabulated value) heat of combustion to estimate AH, for 2,3,3-trimethylpentane. kJ/mol the tolerance is +/-9%
Expert Answer:

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder,? Ronald W. Rousseau,? Lisa G. Bullard
Posted Date:
Students also viewed these chemical engineering questions
-
The standard heat of combustion of liquid n-octane to form CO 2 and liquid water at 25C and 1 atm is H c = 5471 kJ/mol. (a) Briefly explain what that means. Your explanation may take the form When...
-
The standard heat of combustion (Hc) of liquid 2.3,3-trimethylpentane [C8H18] is reported in a table of physical properties to be 4850 kJ/mol. A footnote indicates that the reference temperature for...
-
The standard heat of the reaction 4 NH 3 (g) + 5O 2 (g) 4 NO (g) + 6 H 2 O (g) is H t = 904.7 kJ/mol (a) Briefly explain what that means. Your explanation may take the form When ______ (specify...
-
Single PlantwideandMultiple Production Department Factory Overhead Rate Methodsand Product Cost Distortion Eclipse Motor Company manufactures two types of specialty electric motors, a commercial...
-
When sodium ethoxide reacts with 1-(chloromethyl)oxirane (also called epichlorohy drin), labeled with 14C as shown by the asterisk in I, the major product is II. Provide a mechanistic explanation for...
-
The probability that Sam parks in a no-parking zone and gets a parking ticket is 0.06, and the probability that Sam cannot find a legal parking spot and has to park in the no-parking zone is 0.20. On...
-
Suppose there is a stock and a bond governed by the equations It is desired to construct a portfolio of these two securities that gives the maximum expected \(\log\) of return. However, although...
-
On January 1, 2013, Tonge Industries had outstanding 440,000 common shares (par $l) that originally sold for $20 per share, and 4,000 shares of 10% cumulative preferred stock (par $100), convertible...
-
6) Consider following C function: } int fun (int n) int i, result = 1; for (i=1 i
-
Go to the websites for project management tools (besides Microsoft Project), such as Apptivo (www.apptivo.com), GanttProject (www.ganttproject.biz), Gantter (www.gantter.com), and smartsheet...
-
Goodlife fitness chain wants to identify the type of people who are currently using the gym and the physical fitness activities they are doing. Gyms will compare this data with existing demographics...
-
Please explain why you think placing unearned client funds in an office operating account would not be ethical and would not be a safe practice. Please explain how you would handle the situation if...
-
The following are independent situations. a. A company orders and receives 10 personal computers for office use for which it signs a note promising to pay $20,000 within three months. b. A company...
-
Read the Change Metaphor Entitled Who Moved My Cheese and Critically Reflect on the Following Points 1) Being a metaphor in the story, what the Cheese represents in the real life change situation?...
-
How does data management within an information system impact the effectiveness and success of laboratory projects?
-
What role does data security and privacy play in the context of laboratory project information systems?
-
Periodic inventory using FIFO, LIFO, and weighted average cost methods The units of an item available for sale during the year were as follows: Jan. 1 Aug. 13 Inventory Purchase 10 units at $46 $460...
-
What is the amount of total interest dollars earned on a $5,000 deposit earning 6% for 20 years?
-
You are performing an experiment to measure the specific internal energy of a gas relative to a reference state of 25C and 1 atm (at which conditions U is arbitrarily set equal to 0). The gas is...
-
A stream of ethylene glycol vapor at its normal boiling point and 1 atm flowing at a rate of 175 kg/mm is to be condensed at constant pressure. The product stream from the condenser is liquid glycol...
-
A solution of diphenyl (MW = 154.2) in benzene is formed by mixing 56.0 g diphenyl with 550.0 mL of benzene. Estimate the effective vapor pressure of the solution at 30C and the melting and boiling...
-
Give an example of a local wind pattern. Give an example of a global wind pattern.
-
What drives surface currents?
-
Do surface currents affect water temperature, air temperature, or both?

Study smarter with the SolutionInn App


