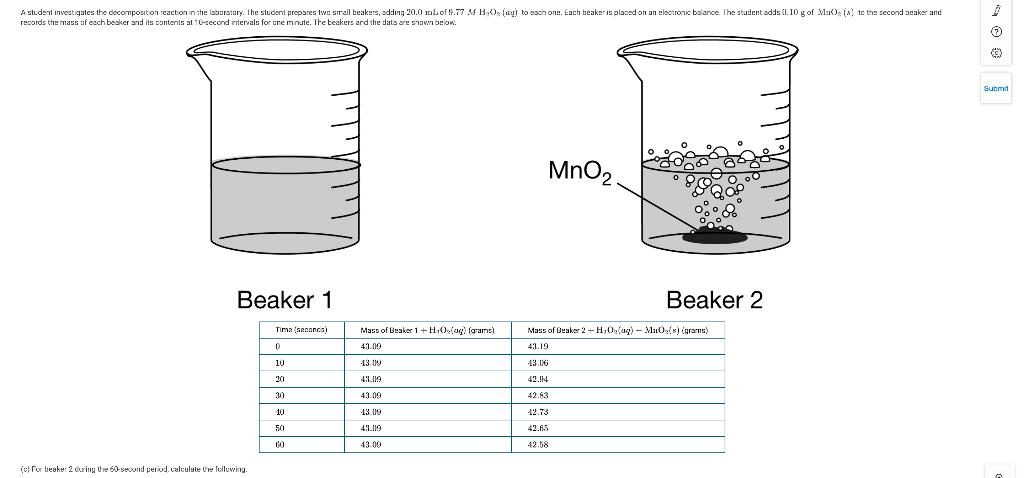
(i) The number of moles of O2(g) that was produced (ii) The mass ofH2O2(aq)that decomposed (d) The...
Question:
(i) The number of moles of O2(g) that was produced
(ii) The mass of H2O2(aq) that decomposed
(d) The student continues the experiment for an additional minute. For beaker 2, will the mass of H2O2(aq) consumed during the second minute be greater than, less than, or equal to the mass of H2O2(aq) consumed during the first minute? Explain your answer referring to the data in the table.
(e) Based on the data, the student claims that the catalyzed reaction has zeroth-order kinetics. Do you agree with the student’s claim? Justify your answer.
(f) A second student did the experiment using larger volumes of 9.77MH2O2(aq) . The student noticed that the reaction in beaker 2 proceeded extremely rapidly, causing some of the liquid to splash out of the beaker onto the lab table. The student claims that as a result of the loss of the liquid from the beaker, the calculated number of moles of O2(g) produced is greater than the actual number of moles of O2(g) produced during the first 60 seconds. Do you agree with the student? Justify your answer.
(g) The hydrogen peroxide used in this experiment can be prepared by the reaction of solid ammonium peroxydisulfate, (NH4)2S2O8, with water. The products are hydrogen peroxide (H2O2) and ammonium bisulfate (NH4HSO4). Write the balanced equation for the reaction.