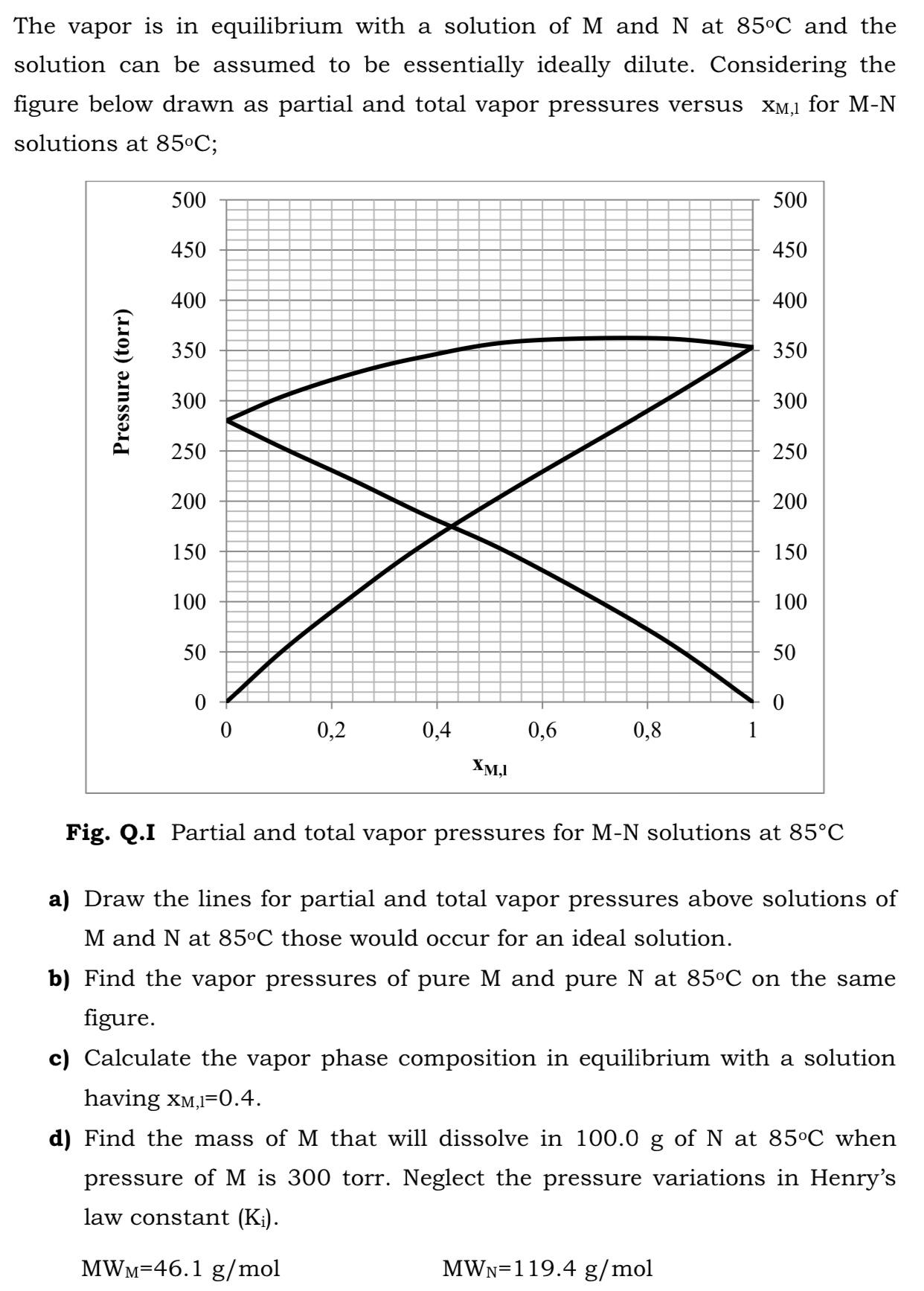
The vapor is in equilibrium with a solution ofM and N at 85C and the solution...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The vapor is in equilibrium with a solution ofM and N at 85°C and the solution can be assumed to be essentially ideally dilute. Considering the figure below drawn as partial and total vapor pressures versus XM,1 for M-N solutions at 85°C; 500 500 450 450 400 400 350 350 300 300 250 250 200 200 150 150 100 100 50 50 0,2 0,4 0,6 0,8 XM,I Fig. Q.I Partial and total vapor pressures for M-N solutions at 85°C a) Draw the lines for partial and total vapor pressures above solutions of M and N at 85°C those would occur for an ideal solution. b) Find the vapor pressures of pure M and pure N at 85°C on the same figure. c) Calculate the vapor phase composition in equilibrium with a solution having xM,I=0.4. d) Find the mass of M that will dissolve in 100.0 g of N at 85°C when pressure of M is 300 torr. Neglect the pressure variations in Henry's law constant (K¡). MWM=46.1 g/mol MWN=119.4 g/mol Pressure (torr) The vapor is in equilibrium with a solution ofM and N at 85°C and the solution can be assumed to be essentially ideally dilute. Considering the figure below drawn as partial and total vapor pressures versus XM,1 for M-N solutions at 85°C; 500 500 450 450 400 400 350 350 300 300 250 250 200 200 150 150 100 100 50 50 0,2 0,4 0,6 0,8 XM,I Fig. Q.I Partial and total vapor pressures for M-N solutions at 85°C a) Draw the lines for partial and total vapor pressures above solutions of M and N at 85°C those would occur for an ideal solution. b) Find the vapor pressures of pure M and pure N at 85°C on the same figure. c) Calculate the vapor phase composition in equilibrium with a solution having xM,I=0.4. d) Find the mass of M that will dissolve in 100.0 g of N at 85°C when pressure of M is 300 torr. Neglect the pressure variations in Henry's law constant (K¡). MWM=46.1 g/mol MWN=119.4 g/mol Pressure (torr)
Expert Answer:
Answer rating: 100% (QA)
a The experimental and Idea l partial vapour pressures of Solu... View the full answer

Related Book For 

Foundations of Financial Management
ISBN: 978-1259194078
15th edition
Authors: Stanley Block, Geoffrey Hirt, Bartley Danielsen
Posted Date:
Students also viewed these chemical engineering questions
-
When returns from a project can be assumed to be normally distributed, such as those shown in Figure (represented by a symmetrical, bell-shaped curve), the areas under the curve can be determined...
-
Soil water with 10^-3 N alkalinity is in equilibrium with a soil gas in which PCO2 = 10^-1 atm. Calculate the pH and total dissolved inorganic carbon concentration (M).
-
A water is in equilibrium with Ca3(PO4)2(s). The solution contains other cations and anions, but no other weak acids or bases or sources of Ca and PO4, and has a pH of 8.6. Ignore complexes. (a) What...
-
Explain these points in detail w.r.t application of control in auditing with examples. 1: Relate data capture control with internal control in auditing with example 2: Relate validation control with...
-
Crable and Tesch, partners in a systems consulting firm, budgeted the following professional labor hours for the year ended December 31, 2016: Partners . . . . . . . . . . . . . . . . . . . . . . . ....
-
If no real engine can be as efficient as a Carnot engine operating between the same two temperatures, what is the point of developing and using Eq. (20.14)? Eq. (20.14) Tc TH Tc TH (efficiency of a...
-
Wilmot Real Estate Co. had the following balance sheet at year-end 2015: In early 2016, the company took out a \($200,000\) two-year bank loan to finance new real estate investments. The loan...
-
In the year just ended, Callaway Lighting had sales of $5,000,000 and incurred a cost of goods sold equal to $4,500,000. The firms operating expenses were $130,000 and its increase in retained...
-
The Cost of Capital: Cost of New Common Stock If a firm plans to issue new stock, flotation costs (investment bankers' fees) should not be ignored. There are two approaches to use to account for...
-
Constructing a distribution of demand during reorder lead time is complicated if the lead time itself is variable. Consider the following distribution for a reorder point inventory system. a. What is...
-
Carmelita Inc., has the following information available: Costs from Costs from Beginning Inventory Current Period Direct materials $4,200 $25,900 Conversion costs 6,400 153,300 At the beginning of...
-
Use Matlab to solve the simultaneous linear algebraic equations to solve for all currents: 4 R = 15 R = 10 R = 15 www 2 R = 50 ww R = 20 0 R = 50 ww OV = 200 volts O V = 0 volts 6
-
What is the run-time for the following algorithm? for (int i = 0; i < size/2; i++) { } System.out.println(arr[i] + " ") ;
-
Sold a 500,000 5 year bond, semi-annual interest at 6% when market interest rate was 8% Prepare the journal entry.
-
A girl who is initially running at 1 . 6 m / s increases her velocity to 3 . 4 0 m / s in 5 . 0 0 seconds, find her acceleration.
-
Anthony and Claire Anthony has not updated his will since his marriage to his first wife, Sophie. Sophie is listed as his Executor and Power of Attorney for both Personal Care and Property. Claire...
-
If we were admitting those students whose IQ score fell in the top 10 % of the distribution (assume normal distribution), what would be the cut-off point (raw score cut-off) for admission (=100, =...
-
Write a declaration for each of the following: a. A line that extends from point (60, 100) to point (30, 90) b. A rectangle that is 20 pixels wide, 100 pixels high, and has its upper-left corner at...
-
How did the SarbanesOxley Act impact corporations financial reports?
-
Betty Bronson has just retired after 25 years with the electric company. Her total pension funds have an accumulated value of $180,000, and her life expectancy is 15 more years. Her pension fund...
-
Assume a firm has earnings before depreciation and taxes of $440,000 and depreciation of $140,000. a. If it is in a 35 percent tax bracket, compute its cash flow. b. If it is in a 20 percent tax...
-
The comparative financial statements for CyberOptic Corporation are in the Working Papers. The financial statements have been completed up to the trend analysis section. The following information is...
-
The comparative financial statements for CyberOptic Corporation are in the Working Papers. The financial statements have been completed up to the comparative analysis section. Instructions: 1....
-
Use the comparative statements from Application Problem 17-1 to complete this problem. Instructions: 1. Based on CyberOptic's comparative financial statements prepared in Application Problem 17-1 and...

Study smarter with the SolutionInn App


