Two metal rods, one silver and the other copper, are both attached to a steam chamber...
Fantastic news! We've Found the answer you've been seeking!
Question:
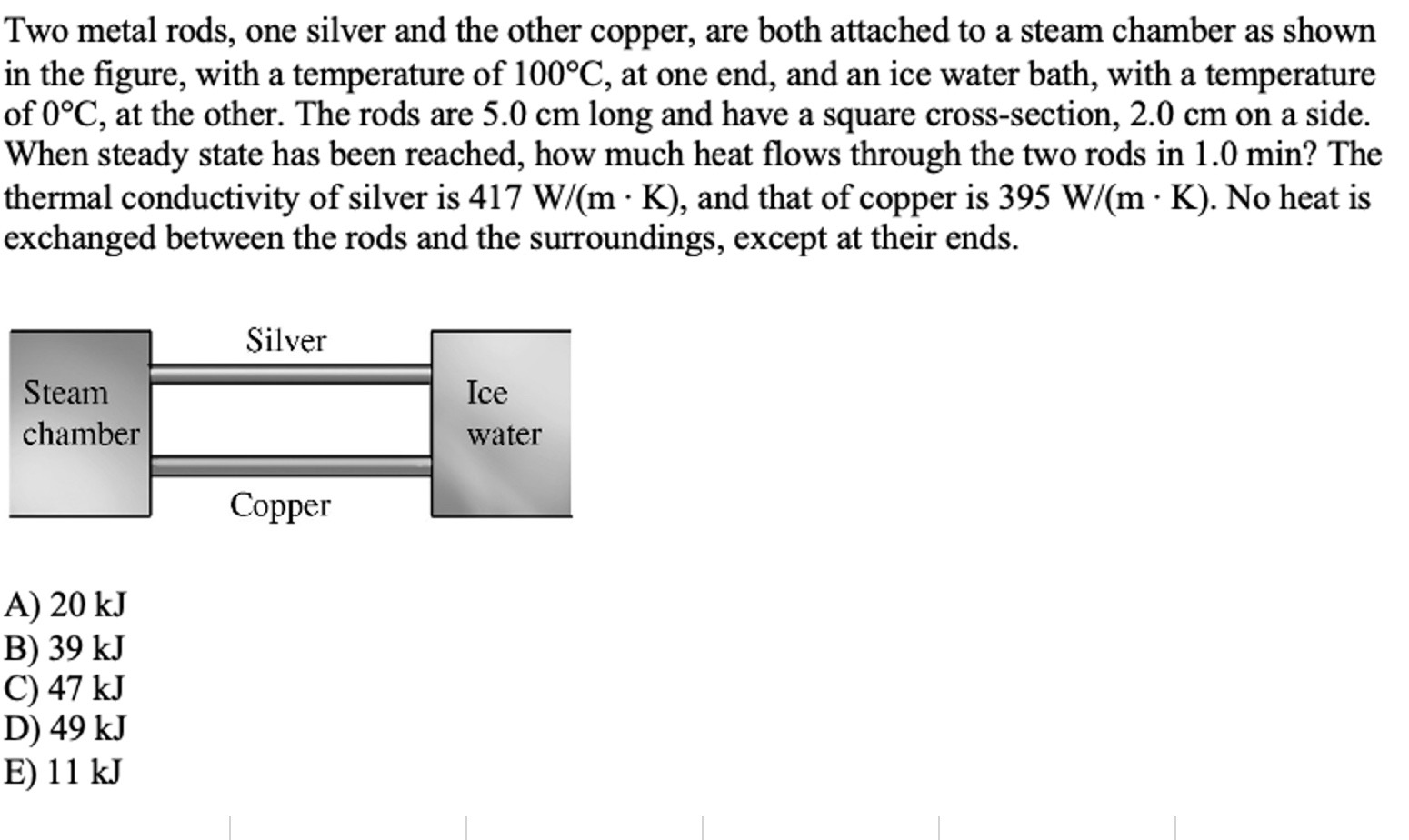
Transcribed Image Text:
Two metal rods, one silver and the other copper, are both attached to a steam chamber as shown in the figure, with a temperature of 100°C, at one end, and an ice water bath, with a temperature of 0°C, at the other. The rods are 5.0 cm long and have a square cross-section, 2.0 cm on a side. When steady state has been reached, how much heat flows through the two rods in 1.0 min? The thermal conductivity of silver is 417 W/(m K), and that of copper is 395 W/(m K). No heat is exchanged between the rods and the surroundings, except at their ends. Silver Steam chamber Ice water Copper A) 20 kJ B) 39 kJ C) 47 kJ D) 49 kJ E) 11 kJ Two metal rods, one silver and the other copper, are both attached to a steam chamber as shown in the figure, with a temperature of 100°C, at one end, and an ice water bath, with a temperature of 0°C, at the other. The rods are 5.0 cm long and have a square cross-section, 2.0 cm on a side. When steady state has been reached, how much heat flows through the two rods in 1.0 min? The thermal conductivity of silver is 417 W/(m K), and that of copper is 395 W/(m K). No heat is exchanged between the rods and the surroundings, except at their ends. Silver Steam chamber Ice water Copper A) 20 kJ B) 39 kJ C) 47 kJ D) 49 kJ E) 11 kJ
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below This problem can be solved using the concept of heat flow through a conductor Since the r... View the full answer

Related Book For 

Principles Of Heat Transfer
ISBN: 9781305387102
8th Edition
Authors: Frank Kreith, Raj M. Manglik, Mark S. Bohn
Posted Date:
Students also viewed these physics questions
-
A long rod, insulated to prevent heat loss along its sides, is in perfect thermal contact with boiling water (at atmospheric pressure) at one end and with an ice-water mixture at the other (Fig....
-
The HaberBosch process for the production of ammonia is one of the key industrial processes in developed countries. N 2 (g) + 3 H 2 (g) 2 NH 3 (g) (a) Calculate r G for the reaction at 298 K, 800...
-
On January 1, 2018, the Haskins Company adopted the dollar-value LIFO method for its one inventory pool. The pool's value on this date was $660,000. The 2018 and 2019 ending inventory valued at...
-
Kesha, a sole proprietor, is engaged in a cash basis service business. In the current year, she incorporates the business to form Kiwi Corporation. She transfers assets with a basis of $500,000 (fair...
-
The figure below shows the age structures for the world human population in 1950 and 2010 as well as a projection for the year 2100. How did the age structure of the world population change between...
-
In 2018, Ginzel Corporation agreed to provide a client with 20 detailed marketing analyses of its client's key products for a total fee of $110,000. The fee was computed as $5,000 per report x 20...
-
Recommend a qualitative analysis technique and give reasons for your selection. Then, please comment and critique on 2 fellow students recommendations. 2. Please discuss whether you agree or disagree...
-
Capstone Case: Sunrise Bakery Expansion The Sunrise Bakery Corporation was originally founded in Houston, TX in 1991 by Griffin Harris, who currently serves as the company's Chief Executive Officer....
-
Mr. and Mrs. Fox acquired a rental property on January 2, 2021, in the City of Ottawa. Although it is generally difficult to rent a property during the coldest winter months in Ottawa, the homeowners...
-
Determine the kinetic energy of a 1 2 4 0 - kg car moving with a speed of 1 2 . 6 m / s .
-
In Figure 1, the heat exchanger (HX) has a pressure drop of 25 psi on the water side. The heat exchanger inlet pipe velocity is 10 ft/s. Assume the properties of water are constant at 70 F. What is a...
-
What is the significance of Rochin v. California and Mapp v. Ohio? And, do you agree or disagree with the Supreme Court's decision in both these cases?
-
What is your stance on the efficacy and desirability of community policing as a policy approach? Please elucidate the rationale behind your position, considering its potential impacts on crime...
-
With the use of Excel, calculate the worth of each alternative accordingly. 2. At what Exit price would the two alternatives be identical in their worth Discount rate = 9% for both projects Year Free...
-
The times required to complete each of eight jobs in a two-machine flow shop are shown in the table that follows. Each job must follow the same sequence, beginning with machine A and moving to...
-
Suppose that the electrical potential at the point (x, y, z) is E(x, y, z) = x + y - 2z. What is the direction of the acceleration at the point (1,3,2)?
-
The heat transfer coefficient between a surface and a liquid is 57 W/ (m 2 K). How many watts per square meter will be transferred in this system if the temperature difference is 10C?
-
In the shell of a shell-and-tube heat exchanger with two shell passes and eight tube passes, 12.6 kg/s of water is heated from 80C to 150C. Hot exhaust gases having roughly the same physical...
-
An aniline-alcohol solution is flowing at a velocity of 3 m/s through a long, 2.5-cm-ID thin-wall tube. Steam is condensing at atmospheric pressure on the outer surface of the tube, and the tube wall...
-
Parents with a child in subsidized childcare in the province of Qubec, Canada, pay a basic amount and, depending on family income, may pay an additional amount. As of January 1, 2017, families with a...
-
A firm in the state of Karnataka in India can source one of its factors of production either within the state, \(F_{K}\), or from the neighboring state of Maharashtra, \(F_{M}\). Assume the quality...
-
In the Application A Beer Manufacturer's ShortRun Cost Curves," the short-run variable cost function for Japanese beer is \(V C=0.55 q^{1.67}\). If the fixed cost is 600 and the firm produces 550...

Study smarter with the SolutionInn App


