What is standard deviation of such a portfolio? Assume that there are only two stocks in the
Fantastic news! We've Found the answer you've been seeking!
Question:
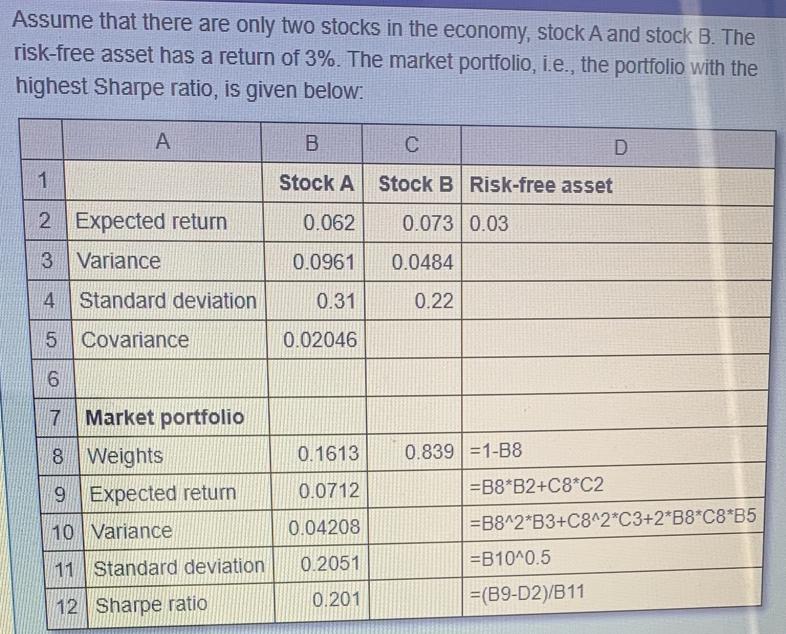
Transcribed Image Text:
Assume that there are only two stocks in the economy, stock A and stock B. The risk-free asset has a return of 3%. The market portfolio, i.e., the portfolio with the highest Sharpe ratio, is given below: A 1 2 Expected return 3 Variance 4 Standard deviation 5 Covariance 6 7 Market portfolio 8 Weights 9 Expected return 10 Variance 11 Standard deviation 12 Sharpe ratio B Stock A C Stock B Risk-free asset 0.062 0.0961 0.0484 0.31 0.22 0.02046 0.1613 0.0712 0.04208 0.2051 0.201 0.073 0.03 0.839 1-B8 D =B8*B2+C8 C2 =B8^2*B3+C8^2*C3+2*B8*C8*B5 =B10^0.5 =(B9-D2)/B11 Assume that there are only two stocks in the economy, stock A and stock B. The risk-free asset has a return of 3%. The market portfolio, i.e., the portfolio with the highest Sharpe ratio, is given below: A 1 2 Expected return 3 Variance 4 Standard deviation 5 Covariance 6 7 Market portfolio 8 Weights 9 Expected return 10 Variance 11 Standard deviation 12 Sharpe ratio B Stock A C Stock B Risk-free asset 0.062 0.0961 0.0484 0.31 0.22 0.02046 0.1613 0.0712 0.04208 0.2051 0.201 0.073 0.03 0.839 1-B8 D =B8*B2+C8 C2 =B8^2*B3+C8^2*C3+2*B8*C8*B5 =B10^0.5 =(B9-D2)/B11
Expert Answer:
Answer rating: 100% (QA)
To calculate the standard deviation of the portfolio we need to consider the weights of ... View the full answer

Related Book For 

Data Analysis and Decision Making
ISBN: 978-0538476126
4th edition
Authors: Christian Albright, Wayne Winston, Christopher Zappe
Posted Date:
Students also viewed these accounting questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Assume that there are only two shareholders in a company; the controlling shareholder holds 90% of the equity shares of the company whilst the other non-controlling shareholder holds the remaining...
-
A 1000-ft-long 2-ft-diameter concrete pipe (sharp-edge entrance) is used to convey 60F water from Reservoir A to B. At the end of the pipe, a diffuser with an exit diameter of 3.5 ft is installed to...
-
Addison, Inc. makes a single product, an indoor fireplace. Data for last year is as follows: Selling price per fireplace Manufacturing costs: Variable per unit produced: Direct materials Direct labor...
-
Because of two hydrogen bonds, carboxylic acids show a very strong attractive force between two molecules that persists even in the gas phase. Show this hydrogen bonding between two carboxylic acid...
-
What is the primary factor that distinguishes what may be described as an auditing "mechanic" from the true auditor?
-
Olympic Village Enterprises lost its entire inventory in a hurricane that occurred on July 31,2010. Over the past five years, gross profit has averaged 30% of net sales. The companys records reveal...
-
Baldacci Inc. has a casting machine that is used for three of the companys products. Each machine setup costs $ 20,445, and the machine was set up in June for six different production runs. The...
-
Explore three multiple Internet sites for providers of home mortgages or consumer credit. create a mind map and analysis What are the terms of the mortgage or credit agreement? What are the...
-
A year ago, Rebecca purchased 100 shares of Havad stock for $25 per share. Yesterday, she placed a limit order to sell her stock at a price of $30 per share before the market opened. The stocks price...
-
Assume that in Bolivia it takes 9 0 hours of labor to produce a ton of salt and 6 0 hours of labor to produce a ton of soybean oil. In addition, assume that in Brazil it takes 8 0 hours of labor to...
-
How much should Town report as unrestricted contribution revenue for 20X6 with respect to the pledges? a. $0 b. $360,000 c. $720,000 d. $800,000 Town Service Center is a voluntary health and welfare...
-
Which of the following accounts of a governmental unit is credited to close it out at the fiscal year-end? a. Appropriations Control b. RevenueProperty Tax c. Budgetary Fund BalanceAssigned for...
-
What do we mean by the term location economies and why is it relevant to international business?
-
On January 2, 20X2, a not-for-profit botanical society received a gift of an exhaustible fixed asset with an estimated useful life of 10 years and no salvage value. The donors cost of this asset was...
-
The initial transfer of cash from the general fund to establish an internal service fund requires the general fund to credit Cash and debit a. Accounts ReceivableInternal Service Fund. b. Transfers...
-
For the given parallel wiper mechanism, find the amount of area wiped in one stroke by graphical method. Assume AB = 30 mm, BC = 200 mm, CD = 80 mm, EF = DG = 20 mm, DE = GF = 300 mm, 8 = 10 150 mm...
-
State whether each of the following will increase or decrease the power of a one-way between-subjects ANOVA. (a) The effect size increases. (b) Mean square error decreases. (c) Mean square between...
-
Do the absolute magnitudes of the monetary outcomes matter in the risky venture example? Consider the following two possibilities. In each case, multiply all monetary values in the example by a...
-
The U.S. government is attempting to determine whether immigrants should be tested for a contagious disease. Assume that the decision will be made on a financial basis. Furthermore, assume that each...
-
The file P08_77.xlsx contains data on 856 customers who have either tried or not tried a companys new frozen lasagna dinner. The manager of the company would like to compare the proportion of...
-
A hydrocarbon \(\mathrm{H}_{2}\left(\mathrm{CH}_{2} ight)_{n}\) is dissolved in a solvent \(S\) which freezes at \(9.0^{\circ} \mathrm{C}\). A solution which contains \(0.90 \mathrm{~g}\) of...
-
Estimate the depression in the freezing point if \(58.5 \mathrm{~g}\) of \(\mathrm{NaCl}\) is added to \(1 \mathrm{~L}\) of water at atmospheric pressure.
-
Calculate the mass of methyl alcohol which, when dissolved in \(100 \mathrm{~g}\) of water, would just prevent the formation of ice at \(-10^{\circ} \mathrm{C}\), given that \(K_{\mathrm{f}}\) is...

Study smarter with the SolutionInn App


