What would differ if we used LDA as a base in this synthesis? Explain. What would differ
Fantastic news! We've Found the answer you've been seeking!
Question:
What would differ if we used LDA as a base in this synthesis? Explain. What would differ if we used LDA as a base and ethanol as a solvent in this synthesis? Explain.
Transcribed Image Text:
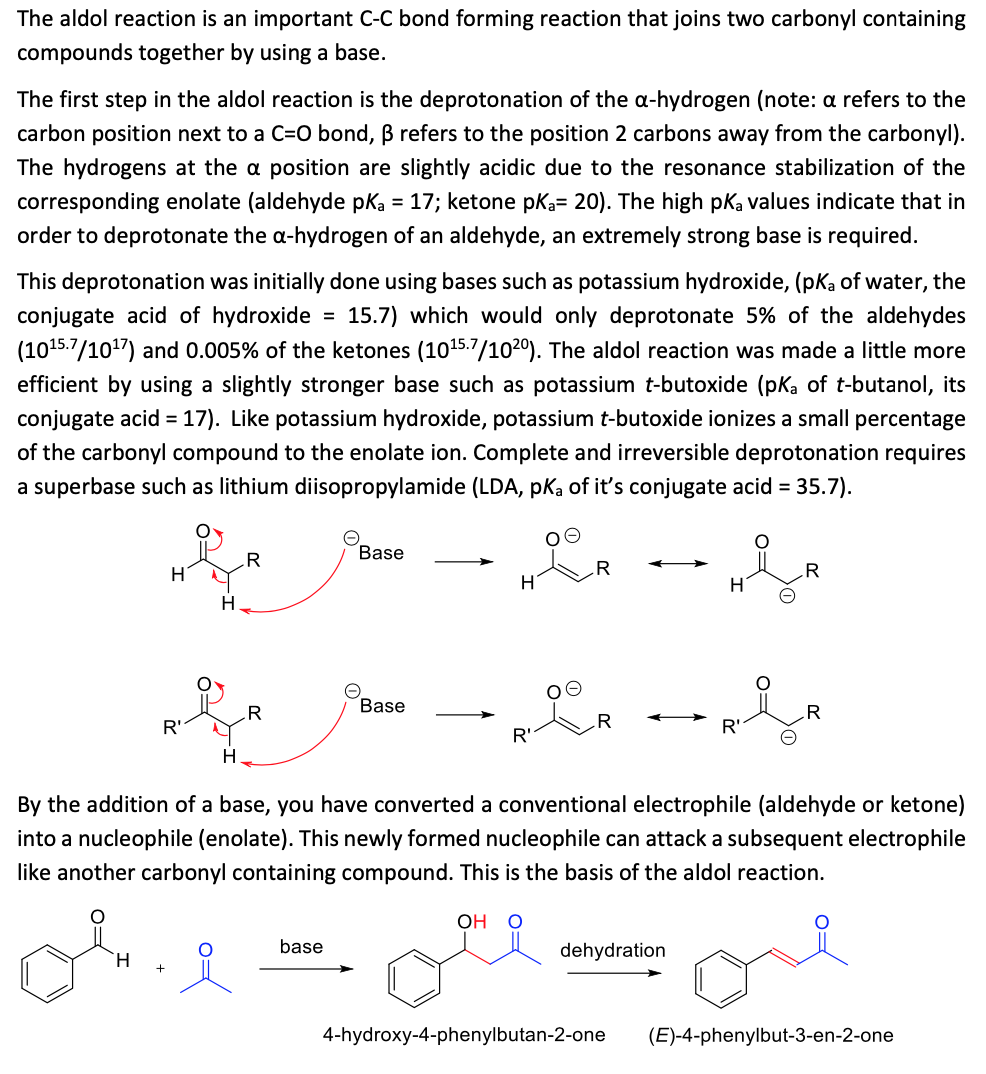
The aldol reaction is an important C-C bond forming reaction that joins two carbonyl containing compounds together by using a base. The first step in the aldol reaction is the deprotonation of the a-hydrogen (note: a refers to the carbon position next to a C=0 bond, B refers to the position 2 carbons away from the carbonyl). The hydrogens at the a position are slightly acidic due to the resonance stabilization of the corresponding enolate (aldehyde pka = 17; ketone pka= 20). The high pka values indicate that in order to deprotonate the a-hydrogen of an aldehyde, an extremely strong base is required. This deprotonation was initially done using bases such as potassium hydroxide, (pKa of water, the conjugate acid of hydroxide = 15.7) which would only deprotonate 5% of the aldehydes (1015.7/1017) and 0.005% of the ketones (1015.7/1020). The aldol reaction was made a little more efficient by using a slightly stronger base such as potassium t-butoxide (pką of t-butanol, its conjugate acid = 17). Like potassium hydroxide, potassium t-butoxide ionizes a small percentage of the carbonyl compound to the enolate ion. Complete and irreversible deprotonation requires a superbase such as lithium diisopropylamide (LDA, pKa of it's conjugate acid = 35.7). Base .R .R H Base .R R' R' H. By the addition of a base, you have converted a conventional electrophile (aldehyde or ketone) into a nucleophile (enolate). This newly formed nucleophile can attack a subsequent electrophile like another carbonyl containing compound. This is the basis of the aldol reaction. ОН О base dehydration H. 4-hydroxy-4-phenylbutan-2-one (E)-4-phenylbut-3-en-2-one The aldol reaction is an important C-C bond forming reaction that joins two carbonyl containing compounds together by using a base. The first step in the aldol reaction is the deprotonation of the a-hydrogen (note: a refers to the carbon position next to a C=0 bond, B refers to the position 2 carbons away from the carbonyl). The hydrogens at the a position are slightly acidic due to the resonance stabilization of the corresponding enolate (aldehyde pka = 17; ketone pka= 20). The high pka values indicate that in order to deprotonate the a-hydrogen of an aldehyde, an extremely strong base is required. This deprotonation was initially done using bases such as potassium hydroxide, (pKa of water, the conjugate acid of hydroxide = 15.7) which would only deprotonate 5% of the aldehydes (1015.7/1017) and 0.005% of the ketones (1015.7/1020). The aldol reaction was made a little more efficient by using a slightly stronger base such as potassium t-butoxide (pką of t-butanol, its conjugate acid = 17). Like potassium hydroxide, potassium t-butoxide ionizes a small percentage of the carbonyl compound to the enolate ion. Complete and irreversible deprotonation requires a superbase such as lithium diisopropylamide (LDA, pKa of it's conjugate acid = 35.7). Base .R .R H Base .R R' R' H. By the addition of a base, you have converted a conventional electrophile (aldehyde or ketone) into a nucleophile (enolate). This newly formed nucleophile can attack a subsequent electrophile like another carbonyl containing compound. This is the basis of the aldol reaction. ОН О base dehydration H. 4-hydroxy-4-phenylbutan-2-one (E)-4-phenylbut-3-en-2-one
Expert Answer:
Answer rating: 100% (QA)
Lithium Diisopropyl Amide LDA is a Strong Bulky base It forms Eno... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Would ethanol (CH 3 CH 2 OH) be a suitable solvent in which to perform the following proton transfer? Explain your answer: ONH2 + NH3 NH2 H.
-
What nucleophile or base and what type of solvent could be used for the conversion of isobutyl bromide into each of the following compound? (CH3)TCHCH2SCH2CH3
-
What would be the effect on DNA synthesis if the telomerase enzyme were inactivated?
-
Shown here is a list published by Electronics Weekly.com of the top five semiconductor companies in the United States by revenue ($ billions). a. Construct a bar chart to display these data. b....
-
During its scal year ending June 30, 2014, the Parkville Independent School District enters into a two-year lease for ofce space covering the period May 1, 2014, through April 30, 2016. Annual rent...
-
Wonderland Preschool operates a not-for-profit morning preschool. Each family pays a non- refundable registration fee of $100 per child per school year. Monthly tuition for the eight- month school...
-
You are the president of a non-profit organization that has grown quickly over the last few years. For the first time, you have had an independent accountant audit your financial records. Upon the...
-
Morgan Companys balance sheet at December 31, 2016, is presented below. During January 2017, the following transactions occurred. (Morgan Company uses the perpetual inventory system.) 1. Morgan paid...
-
Profit a person can earn by opening his own sleep center
-
In the circuit of Fig. 5.59 , calculate v o of v s = 2 V. 8 k2 2 k2 4 k2 4 k2 Vo Vs (+I) + I
-
What is the largest number of burritos that the student could afford to purchase in one month? 2 0 burritos in one month Indicate a bundle that is unaffordable by labeling a point A . Indicate a...
-
In the fall of2009, Kraft Foods attempted to buy Cadbury plc. Data for each of the two companies are given in the table. Both companies are all equity financed. The CEO of Kraft Foods estimated that...
-
Where did I go wrong? You can see the code I have applied and it's almost there, but something isn't correct in my code and I can't figure out where I went wrong. Please help me find the error and...
-
Wehner Company is currently manufacturing Part ABS-43, producing 53,400 units annually. The part is used in the production of several products made by Wehner. The cost per unit for ABS-43 is as...
-
Harem company uses an annual cost formula for overhead of 72,000 + 1.60 for each direct labor hour worked. For the upcoming month, Karla plans to manufacture 96,000 units. Each units requires five...
-
What role does the Chief Risk Officer play in the credit process and how does it differ from that of the Chief Credit Officer? Distinguish between the account officer, credit specialists and...
-
How much work in joules is done in lifting a 9.63 newtons box from the floor to a table that is 2.77 meters above the floor? Round up to two decimal places
-
The executor of Gina Purcells estate has recorded the following information: Assets discovered at death (at fair value): Cash . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Calculate the percentage of dissociated and un-dissociated forms present in the following solutions: (a) 0.0010M glycolic acid (HOCH2CO2H; pKa = 3.83) at ph = 4.50 (b) 0.0020M propanoic acid (pKa =...
-
Draw the structure of L-galactose, and then answer the following questions: (a) Which other aldohexose gives the same aldaric acid as L-galactose on oxidation with warm HNO3? (b) Is this other...
-
Propose a synthesis of p-(dim-ethylamine) azobenzene from benzene as your only organic starting material.
-
The Arcadia Company is contemplating a large capital investment of \($32\) million in new productionline equipment, which is expected to have a useful life of six years, a residual value of \($2\)...
-
San Joaquin Company began exploration of oil in Texas by paying \($600,000\) for the drilling rights on a large tract of land. The company drilled 10 oil wells on the land. Seven of the wells were...
-
The Arcadia Company is contemplating a large capital investment of \($32\) million in new productionline equipment, which is expected to have a useful life of six years and a residual value of \($2\)...

Study smarter with the SolutionInn App


