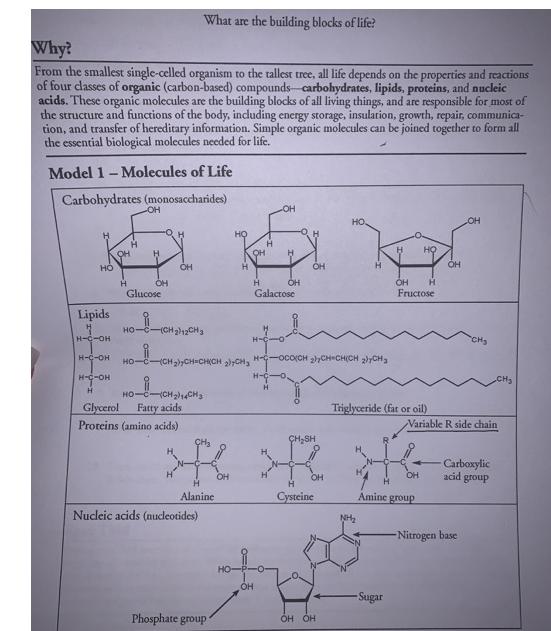
Why? From the smallest single-celled organism to the tallest tree, all life depends on the properties...
Fantastic news! We've Found the answer you've been seeking!
Question:




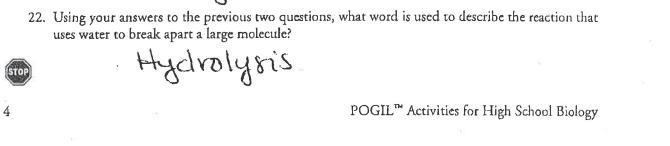


Transcribed Image Text:
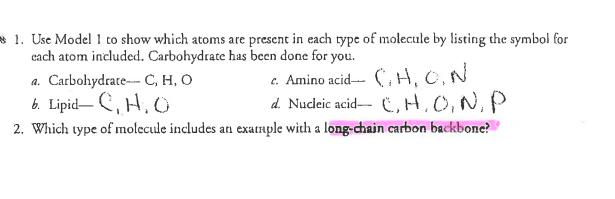
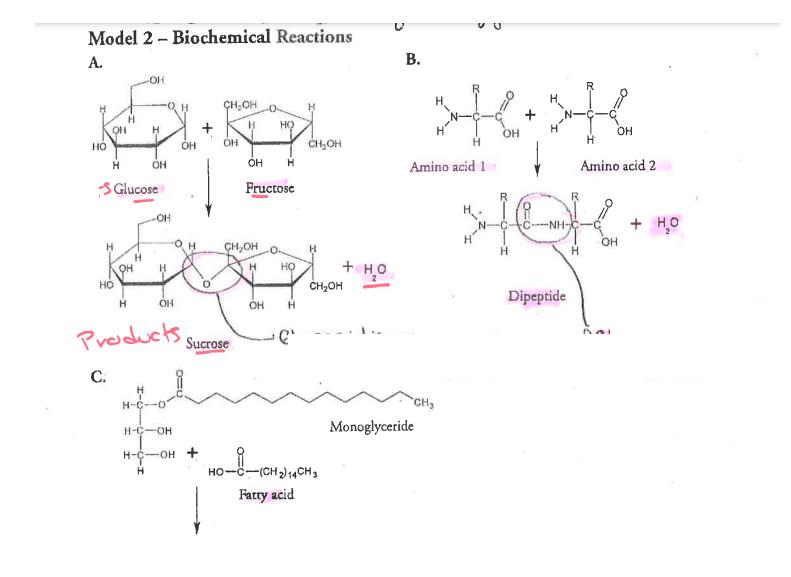
Why? From the smallest single-celled organism to the tallest tree, all life depends on the properties and reactions of four classes of organic (carbon-based) compounds-carbohydrates, lipids, proteins, and nucleic acids. These organic molecules are the building blocks of all living things, and are responsible for most of the structure and functions of the body, including energy storage, insulation, growth, repair, communica- tion, and transfer of hereditary information. Simple organic molecules can be joined together to form all the essential biological molecules needed for life. Model 1 - Molecules of Life Carbohydrates (monosaccharides) HO Lipids H-C-OH H-C-OH H-C-OH H H -OH OH Glucose What are the building blocks of life? OH HO C-(CH2)12CH3 Glycerol Proteins (amino acids) HỌC ỊCH Cy Fatty acids Nucleic acids (nucleotides) Phosphate group- H OH OH HO HỌ Ở CH2)CH CHỊCH, CH, HC000ỊCH LỊCH CHỊCH CH H-C H-C OH H H Galactose OH OH OH CH₂SH > >R R OH OH H Alanine Cysteine Amine group OH OH H NH₂ Triglyceride (fat or oil) -Sugar НО OH H Fructose OH OH CH₂ Variable R side chain -Nitrogen base Carboxylic acid group CH₂ 1. Use Model 1 to show which atoms are present in each type of molecule by listing the symbol for each atom included. Carbohydrate has been done for you. a. Carbohydrate C, H, O b. Lipid-C,H,O c. Amino acid- CH. C. N d. Nucleic acid-C, H, O, N, P 2. Which type of molecule includes an example with a long-chain carbon backbone? 3. In the molecule referred to in the previous question, what is the dominant element attached to the carbon backbone? 4. The fatty acid chain of the lipids is often referred to as a hydrocarbon chain. Discuss with your group why the chain is given this name and write a one-sentence definition for a hydrocarbon. 5. Which molecule has a central carbon atom with four different components around it? K-group. I C 6. Which molecule has a sugar, nitrogenous base, and phosphate group? 7. Discuss with your group members some similarities among all four types of molecules. List as many as you can. 8. What is the chemical formula of the first carbohydrate molecule shown? 9. What three structural groups shown do all amino acids have in common? 10. There are 20 naturally-occurring amino acids, and each one only varies in the structure of the R side chain. Two amino acids are shown in Model 1. What are the R side chains in each? Read This! During chemical reactions, the bonds in molecules are continually broken and reformed. To break a bond, energy must be absorbed. When bonds are formed, energy is released. If more energy is released than absorbed during a chemical change, the process can be used as a source of energy. A general rule for processes such as respiration is the more carbon atoms there are in a molecule, the more energy that molecule can provide to the organism when it is used as food. POCI™ Accivision for Mist Citiem. 11. Using the information from above, is a carbohydrate or a lipid more likely to be a good source of energy for an organism? Model 2 - Biochemical Reactions A. H CH₂OH H H HO OH OH BD OH OH H HO OH H C. H H Glucose HO -OH OH H H-C-0 Fructose H-C-OH H-C-OH + H H -OH + H₂O CH₂OH OH H H H HO Bug 4g. OH Dipeptide Products, Sucrose OH H CH₂OH H CH₂OH HỌ-C(CH2)CHy Fatty acid B. + H₂O Amino acid 1 HIGH+ OH Monoglyceride D CH₂ OH Н Amino acid 2 Bai H H-CIO H-C-OH H-Co Diglyceride Biological Molecules +214 CH3 + H₂O Ester bond (fatty acids join to a 3lycrol) 12. What are the reactants of reaction A? CH₂ Glucose & Fructose 13. What are the products of reaction A? Sucrose & water 3 14. Each of the reactants in reaction A is a single sugar molecule, also called a monosaccharide. What prefix before saccharide would you use to describe sucrose? 15. What are the reactants of reaction B? 1 16. When the two molecules in reaction B are joined together, what other two molecules are pro- duced? 1 l'antido 17. What product do all three reactions in Model 2 have in common? STOP Read This! When sugars are joined together the new bond that form is a glycosidic bond. When amino acids are joined the new bond that forms is a peptide bond. When fatty acids are joined to a glycerol the bond that holds them is an ester bond. 18. On the diagrams in Model 2, circle and label the glycosidic, peptide, and ester bonds. $19. These reactions are all referred to as dehydration synthesis or condensation reactions. With your group develop an explanation for why these terms are used to describe these reactions. for to be LAXILICE 20. These reactions can also be reversed, breaking the large molecule into its individual molecules. What substance would need to be added in order to reverse the reaction? 21. Lysis means to split or separate. What prefix would you add to lysis to mean separate or split using water? 22. Using your answers to the previous two questions, what word is used to describe the reaction that uses water to break apart a large molecule? Hydrolysis. STOP POGIL™ Activities for High School Biology Extension Questions 23. Metabolism is the collective term used to describe all the chemical reactions taking place inside living organisms. Why is water so important for metabolic reactions? ( 24. We store excess food in our body either in the form of carbohydrates (in muscles and the liver) o or as fat (adipose tissue). When our body needs additional energy it uses the carbohydrate source first as a source of "quick" energy, then the fat. Why do you think carbohydrates are used as a source of quick energy rather than fat? Use complete sentences and scientific terminology in your response. 1 tl Inc. 1 Look at the two types of fatty acids below, saturated and unsaturated. What is the difference between the two? Jee D H H-C-H H-C-H H-CH H-C-HI H-C-H HOT Saturated Fatty Acid H-C-H C-H H-C H-C-H H-C-H HO O d W Unsaturated Fatty Acid 26. Saturated fats are solid fats, like the animal fats lard and butter, whereas unsaturated fats are more fluid and form oils, such as vegetable oil. Trans fats are plant oils that are artificially solidified to make them suitable for baking purposes. In recent years trans fats have been associated with negative health issues and are not as widely used. Explain in simple molecular terms what would have to be done to a plant oil to transform it to a trans fat. Why? From the smallest single-celled organism to the tallest tree, all life depends on the properties and reactions of four classes of organic (carbon-based) compounds-carbohydrates, lipids, proteins, and nucleic acids. These organic molecules are the building blocks of all living things, and are responsible for most of the structure and functions of the body, including energy storage, insulation, growth, repair, communica- tion, and transfer of hereditary information. Simple organic molecules can be joined together to form all the essential biological molecules needed for life. Model 1 - Molecules of Life Carbohydrates (monosaccharides) HO Lipids H-C-OH H-C-OH H-C-OH H H -OH OH Glucose What are the building blocks of life? OH HO C-(CH2)12CH3 Glycerol Proteins (amino acids) HỌC ỊCH Cy Fatty acids Nucleic acids (nucleotides) Phosphate group- H OH OH HO HỌ Ở CH2)CH CHỊCH, CH, HC000ỊCH LỊCH CHỊCH CH H-C H-C OH H H Galactose OH OH OH CH₂SH > >R R OH OH H Alanine Cysteine Amine group OH OH H NH₂ Triglyceride (fat or oil) -Sugar НО OH H Fructose OH OH CH₂ Variable R side chain -Nitrogen base Carboxylic acid group CH₂ 1. Use Model 1 to show which atoms are present in each type of molecule by listing the symbol for each atom included. Carbohydrate has been done for you. a. Carbohydrate C, H, O b. Lipid-C,H,O c. Amino acid- CH. C. N d. Nucleic acid-C, H, O, N, P 2. Which type of molecule includes an example with a long-chain carbon backbone? 3. In the molecule referred to in the previous question, what is the dominant element attached to the carbon backbone? 4. The fatty acid chain of the lipids is often referred to as a hydrocarbon chain. Discuss with your group why the chain is given this name and write a one-sentence definition for a hydrocarbon. 5. Which molecule has a central carbon atom with four different components around it? K-group. I C 6. Which molecule has a sugar, nitrogenous base, and phosphate group? 7. Discuss with your group members some similarities among all four types of molecules. List as many as you can. 8. What is the chemical formula of the first carbohydrate molecule shown? 9. What three structural groups shown do all amino acids have in common? 10. There are 20 naturally-occurring amino acids, and each one only varies in the structure of the R side chain. Two amino acids are shown in Model 1. What are the R side chains in each? Read This! During chemical reactions, the bonds in molecules are continually broken and reformed. To break a bond, energy must be absorbed. When bonds are formed, energy is released. If more energy is released than absorbed during a chemical change, the process can be used as a source of energy. A general rule for processes such as respiration is the more carbon atoms there are in a molecule, the more energy that molecule can provide to the organism when it is used as food. POCI™ Accivision for Mist Citiem. 11. Using the information from above, is a carbohydrate or a lipid more likely to be a good source of energy for an organism? Model 2 - Biochemical Reactions A. H CH₂OH H H HO OH OH BD OH OH H HO OH H C. H H Glucose HO -OH OH H H-C-0 Fructose H-C-OH H-C-OH + H H -OH + H₂O CH₂OH OH H H H HO Bug 4g. OH Dipeptide Products, Sucrose OH H CH₂OH H CH₂OH HỌ-C(CH2)CHy Fatty acid B. + H₂O Amino acid 1 HIGH+ OH Monoglyceride D CH₂ OH Н Amino acid 2 Bai H H-CIO H-C-OH H-Co Diglyceride Biological Molecules +214 CH3 + H₂O Ester bond (fatty acids join to a 3lycrol) 12. What are the reactants of reaction A? CH₂ Glucose & Fructose 13. What are the products of reaction A? Sucrose & water 3 14. Each of the reactants in reaction A is a single sugar molecule, also called a monosaccharide. What prefix before saccharide would you use to describe sucrose? 15. What are the reactants of reaction B? 1 16. When the two molecules in reaction B are joined together, what other two molecules are pro- duced? 1 l'antido 17. What product do all three reactions in Model 2 have in common? STOP Read This! When sugars are joined together the new bond that form is a glycosidic bond. When amino acids are joined the new bond that forms is a peptide bond. When fatty acids are joined to a glycerol the bond that holds them is an ester bond. 18. On the diagrams in Model 2, circle and label the glycosidic, peptide, and ester bonds. $19. These reactions are all referred to as dehydration synthesis or condensation reactions. With your group develop an explanation for why these terms are used to describe these reactions. for to be LAXILICE 20. These reactions can also be reversed, breaking the large molecule into its individual molecules. What substance would need to be added in order to reverse the reaction? 21. Lysis means to split or separate. What prefix would you add to lysis to mean separate or split using water? 22. Using your answers to the previous two questions, what word is used to describe the reaction that uses water to break apart a large molecule? Hydrolysis. STOP POGIL™ Activities for High School Biology Extension Questions 23. Metabolism is the collective term used to describe all the chemical reactions taking place inside living organisms. Why is water so important for metabolic reactions? ( 24. We store excess food in our body either in the form of carbohydrates (in muscles and the liver) o or as fat (adipose tissue). When our body needs additional energy it uses the carbohydrate source first as a source of "quick" energy, then the fat. Why do you think carbohydrates are used as a source of quick energy rather than fat? Use complete sentences and scientific terminology in your response. 1 tl Inc. 1 Look at the two types of fatty acids below, saturated and unsaturated. What is the difference between the two? Jee D H H-C-H H-C-H H-CH H-C-HI H-C-H HOT Saturated Fatty Acid H-C-H C-H H-C H-C-H H-C-H HO O d W Unsaturated Fatty Acid 26. Saturated fats are solid fats, like the animal fats lard and butter, whereas unsaturated fats are more fluid and form oils, such as vegetable oil. Trans fats are plant oils that are artificially solidified to make them suitable for baking purposes. In recent years trans fats have been associated with negative health issues and are not as widely used. Explain in simple molecular terms what would have to be done to a plant oil to transform it to a trans fat.
Expert Answer:

Related Book For 

Business Statistics In Practice
ISBN: 9780073401836
6th Edition
Authors: Bruce Bowerman, Richard O'Connell
Posted Date:
Students also viewed these biology questions
-
Amino acids are biological compounds with the following structure, where the R group can vary. The structure and biological function of amino acids will be discussed in Chapter 25. Identify the total...
-
Why are amino acids so named? Write a general structure for an amino acid. What is the importance of the R group to final protein structure? Why does the amino acid cysteine have special significance...
-
Nucleic acids can be denatured by heat, as proteins can. What bonds are broken when a DNA molecule is denatured? Would DNA of greater percentage composition of guanine and cytosine denature more or...
-
Write a program that takes a start string and a stop string as commandline arguments and prints all substrings of a given string that start with the first, end with the second, and otherwise contain...
-
Did the transaction described between DMVW and MNVA constitute a fraudulent transfer or a breach of fiduciary duty by the MNVA board to MNVAs creditors?
-
A zero-coupon bond with 8 years to maturity, trading for $941.73 and is expected to have it's annual YTM jump by 1% when the Federal Reserve announces its rate adjustments. What is the duration...
-
0.8365 Use the Standard Normal Table or technology to find the z-score that corresponds to the cumulative area or percentile. Table 4-Standard Normal Distribution Arca Z 0 Z .09 .08 .07 .06 .05 .04...
-
Larned Corporation recorded the following transactions for the just completed month. a. $ 80,000 in raw materials were purchased on account. b. $ 71,000 in raw materials were requisitioned for use in...
-
A projectile if fired from 19.6m above the ground with an initial speed of 55.6m/s at an angle of 41.2 degrees above the horizontal. a) Determine the time necessary for the projectile to reach its...
-
In this lab, we will use the inverse kinematics to identify two functions to generate an animation of a two-link robot. As mentioned in the previous section, the lengths of the two links are a = 3 ft...
-
Select a well-known company with which you have some familiarity (specify the type of industry like Abbott Laboratories). Then, select 1 of the contemporary management techniques. Why and how do you...
-
Kirinyaga Hydraulic Contractors in partnership with the World Bank are embarking on a water project in Mukurweini Division. They are contemplating to gauge the benefits of the project through cost-...
-
Ali, a small company, is currently considering a major capital investment project for which additional finance will be required. It is not currently feasible to raise additional equity finance;...
-
Explain Demings quality principles? with Example
-
1. What generalization does Rosenblattsupport with examples? Where is it stated most directly? 2. How do the examples of censorship relate to the author's thesis? 3. Who do you believe is the...
-
Green Caterpillar Garden Supplies Inc. is analyzing a project that requires an initial investment of $550,000. The project's expected cash flows are: Year Year 1 Year 2 Year 3 Year 4 Cash Flow...
-
What are the 5 Cs of marketing channel structure?
-
Consider the bottle design study situation in Exercise 11.4. and let A, B and C represent mean daily sales using bottle designs A. B, and C. respectively. Figure 11.5 gives the Excel output of a...
-
In the book Making Hard Decisions: An Introduction to Decision Analysis, Robert T. Clemen presents a decision tree for a research and development decision (note that payoffs are given in millions of...
-
The Tastee Bakery Company supplies a bakery product to many supermarkets in a metropolitan area. The company wishes to study the effect of the height of the shelf display employed by the supermarkets...
-
You are the product manager of Lexcon Fitness and are considering the production of new elliptical trainers. You feel you can sell 5,500 of these per year for 6 years (after which time this project...
-
At present, Global Skateboards Limited is considering expanding its product line to include solar powered skateboards; however, it is questionable how well they will be received by skateboarders....
-
Assume that Fiskars Finland plans to invest in a new project that will annually generate revenues of \($2.3\) million. Cash expenses including both fixed and variable costs will be \($950,000,\) and...

Study smarter with the SolutionInn App


