Write a Lewis structure for ethyl alcohol, CH,CH,OH, showing all valence electrons. You do not...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
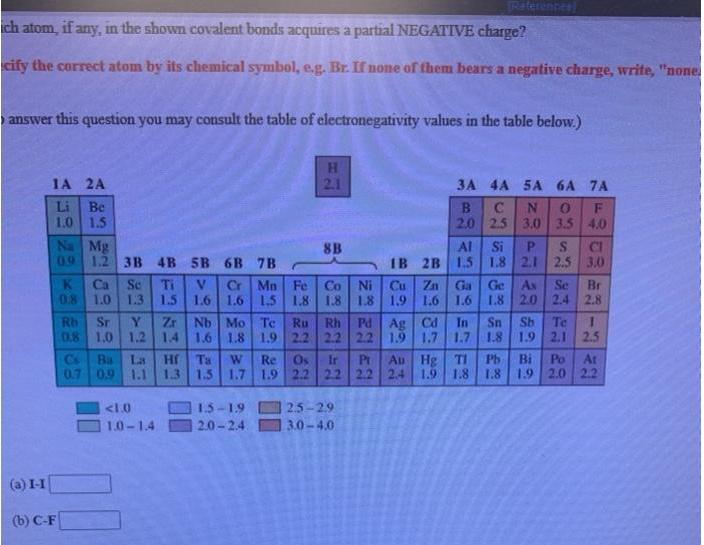
Write a Lewis structure for ethyl alcohol, CH,CH,OH, showing all valence electrons. • You do not have to consider stereochemistry. Explicitly draw all H atoms. Include all valence lone pairs in your answer. Raterences ich atom, if any, in the shown covalent bonds acquires a partial NEGATIVE charge? cify the correct atom by its chemical symbol, e.g. Br. If none of them bears a negative charge, write, "none answer this question you may consult the table of electronegativity values in the table below.) 1A 2A 2.1 3A 4A 5A 6A 7A Be 1.0 1.5 Na Mg 09 12 3B 4B 5B 6B 7B Li 2.0 2.5 3,0 3.5 4.0 Al Si P. IB 2B 1.5 1.8 2.1 2,5 3.0 8B CI Cr Mn Fe 1.6 1.6 1.5 1.8 1.8 Co Ni Cu Zn 1.8 1.9 1.6 Ga Ge K Ca 0.8 Se Ti 1.3 Se Br 2.4 2.8 As 1.0 1.5 1.6 1.8 2.0 Zr 08 1.0 1.2 14 Nb Mo Te Pd Ag Cd 1.9 Rh Te Sb 1.9 2.1 2.5 Sr Rh In Sn 1.7 1.7 1.8 Ru 1.6 1.8 1.9 2.2 2.2 2.2 Au Hg TI Cs Bs 0.7 09 1.1 La Hr Ta 1.3 1.5 1.7 1.9 2.2 22 2.2 2.4 Ir Pr Pb Bị 1.8 Po At 1.9 2.0 2.2 W Re Os 1.9 1.8 <1.0 1.5-19 2.5-29 1.0-1.4 2.0-2.4 3.0-4.0 (a) I-1 (b) C-F How many electrons are in the valence shell of each atom ? (a) Aluminum (AI) (b) Chlorine (CI) (c) Argon (Ar) Relerences Identify the corresponding elements that have ground state electron configurations as specificd. Use either full element names, e.g. Bromine, or the chemical symbol, e.g. Br. (a) 1s2 (b) Is*2s 2p°3s*3p° Write a Lewis structure for ethyl alcohol, CH,CH,OH, showing all valence electrons. • You do not have to consider stereochemistry. Explicitly draw all H atoms. Include all valence lone pairs in your answer. Raterences ich atom, if any, in the shown covalent bonds acquires a partial NEGATIVE charge? cify the correct atom by its chemical symbol, e.g. Br. If none of them bears a negative charge, write, "none answer this question you may consult the table of electronegativity values in the table below.) 1A 2A 2.1 3A 4A 5A 6A 7A Be 1.0 1.5 Na Mg 09 12 3B 4B 5B 6B 7B Li 2.0 2.5 3,0 3.5 4.0 Al Si P. IB 2B 1.5 1.8 2.1 2,5 3.0 8B CI Cr Mn Fe 1.6 1.6 1.5 1.8 1.8 Co Ni Cu Zn 1.8 1.9 1.6 Ga Ge K Ca 0.8 Se Ti 1.3 Se Br 2.4 2.8 As 1.0 1.5 1.6 1.8 2.0 Zr 08 1.0 1.2 14 Nb Mo Te Pd Ag Cd 1.9 Rh Te Sb 1.9 2.1 2.5 Sr Rh In Sn 1.7 1.7 1.8 Ru 1.6 1.8 1.9 2.2 2.2 2.2 Au Hg TI Cs Bs 0.7 09 1.1 La Hr Ta 1.3 1.5 1.7 1.9 2.2 22 2.2 2.4 Ir Pr Pb Bị 1.8 Po At 1.9 2.0 2.2 W Re Os 1.9 1.8 <1.0 1.5-19 2.5-29 1.0-1.4 2.0-2.4 3.0-4.0 (a) I-1 (b) C-F How many electrons are in the valence shell of each atom ? (a) Aluminum (AI) (b) Chlorine (CI) (c) Argon (Ar) Relerences Identify the corresponding elements that have ground state electron configurations as specificd. Use either full element names, e.g. Bromine, or the chemical symbol, e.g. Br. (a) 1s2 (b) Is*2s 2p°3s*3p° Write a Lewis structure for ethyl alcohol, CH,CH,OH, showing all valence electrons. • You do not have to consider stereochemistry. Explicitly draw all H atoms. Include all valence lone pairs in your answer. Raterences ich atom, if any, in the shown covalent bonds acquires a partial NEGATIVE charge? cify the correct atom by its chemical symbol, e.g. Br. If none of them bears a negative charge, write, "none answer this question you may consult the table of electronegativity values in the table below.) 1A 2A 2.1 3A 4A 5A 6A 7A Be 1.0 1.5 Na Mg 09 12 3B 4B 5B 6B 7B Li 2.0 2.5 3,0 3.5 4.0 Al Si P. IB 2B 1.5 1.8 2.1 2,5 3.0 8B CI Cr Mn Fe 1.6 1.6 1.5 1.8 1.8 Co Ni Cu Zn 1.8 1.9 1.6 Ga Ge K Ca 0.8 Se Ti 1.3 Se Br 2.4 2.8 As 1.0 1.5 1.6 1.8 2.0 Zr 08 1.0 1.2 14 Nb Mo Te Pd Ag Cd 1.9 Rh Te Sb 1.9 2.1 2.5 Sr Rh In Sn 1.7 1.7 1.8 Ru 1.6 1.8 1.9 2.2 2.2 2.2 Au Hg TI Cs Bs 0.7 09 1.1 La Hr Ta 1.3 1.5 1.7 1.9 2.2 22 2.2 2.4 Ir Pr Pb Bị 1.8 Po At 1.9 2.0 2.2 W Re Os 1.9 1.8 <1.0 1.5-19 2.5-29 1.0-1.4 2.0-2.4 3.0-4.0 (a) I-1 (b) C-F How many electrons are in the valence shell of each atom ? (a) Aluminum (AI) (b) Chlorine (CI) (c) Argon (Ar) Relerences Identify the corresponding elements that have ground state electron configurations as specificd. Use either full element names, e.g. Bromine, or the chemical symbol, e.g. Br. (a) 1s2 (b) Is*2s 2p°3s*3p° Write a Lewis structure for ethyl alcohol, CH,CH,OH, showing all valence electrons. • You do not have to consider stereochemistry. Explicitly draw all H atoms. Include all valence lone pairs in your answer. Raterences ich atom, if any, in the shown covalent bonds acquires a partial NEGATIVE charge? cify the correct atom by its chemical symbol, e.g. Br. If none of them bears a negative charge, write, "none answer this question you may consult the table of electronegativity values in the table below.) 1A 2A 2.1 3A 4A 5A 6A 7A Be 1.0 1.5 Na Mg 09 12 3B 4B 5B 6B 7B Li 2.0 2.5 3,0 3.5 4.0 Al Si P. IB 2B 1.5 1.8 2.1 2,5 3.0 8B CI Cr Mn Fe 1.6 1.6 1.5 1.8 1.8 Co Ni Cu Zn 1.8 1.9 1.6 Ga Ge K Ca 0.8 Se Ti 1.3 Se Br 2.4 2.8 As 1.0 1.5 1.6 1.8 2.0 Zr 08 1.0 1.2 14 Nb Mo Te Pd Ag Cd 1.9 Rh Te Sb 1.9 2.1 2.5 Sr Rh In Sn 1.7 1.7 1.8 Ru 1.6 1.8 1.9 2.2 2.2 2.2 Au Hg TI Cs Bs 0.7 09 1.1 La Hr Ta 1.3 1.5 1.7 1.9 2.2 22 2.2 2.4 Ir Pr Pb Bị 1.8 Po At 1.9 2.0 2.2 W Re Os 1.9 1.8 <1.0 1.5-19 2.5-29 1.0-1.4 2.0-2.4 3.0-4.0 (a) I-1 (b) C-F How many electrons are in the valence shell of each atom ? (a) Aluminum (AI) (b) Chlorine (CI) (c) Argon (Ar) Relerences Identify the corresponding elements that have ground state electron configurations as specificd. Use either full element names, e.g. Bromine, or the chemical symbol, e.g. Br. (a) 1s2 (b) Is*2s 2p°3s*3p° Write a Lewis structure for ethyl alcohol, CH,CH,OH, showing all valence electrons. • You do not have to consider stereochemistry. Explicitly draw all H atoms. Include all valence lone pairs in your answer. Raterences ich atom, if any, in the shown covalent bonds acquires a partial NEGATIVE charge? cify the correct atom by its chemical symbol, e.g. Br. If none of them bears a negative charge, write, "none answer this question you may consult the table of electronegativity values in the table below.) 1A 2A 2.1 3A 4A 5A 6A 7A Be 1.0 1.5 Na Mg 09 12 3B 4B 5B 6B 7B Li 2.0 2.5 3,0 3.5 4.0 Al Si P. IB 2B 1.5 1.8 2.1 2,5 3.0 8B CI Cr Mn Fe 1.6 1.6 1.5 1.8 1.8 Co Ni Cu Zn 1.8 1.9 1.6 Ga Ge K Ca 0.8 Se Ti 1.3 Se Br 2.4 2.8 As 1.0 1.5 1.6 1.8 2.0 Zr 08 1.0 1.2 14 Nb Mo Te Pd Ag Cd 1.9 Rh Te Sb 1.9 2.1 2.5 Sr Rh In Sn 1.7 1.7 1.8 Ru 1.6 1.8 1.9 2.2 2.2 2.2 Au Hg TI Cs Bs 0.7 09 1.1 La Hr Ta 1.3 1.5 1.7 1.9 2.2 22 2.2 2.4 Ir Pr Pb Bị 1.8 Po At 1.9 2.0 2.2 W Re Os 1.9 1.8 <1.0 1.5-19 2.5-29 1.0-1.4 2.0-2.4 3.0-4.0 (a) I-1 (b) C-F How many electrons are in the valence shell of each atom ? (a) Aluminum (AI) (b) Chlorine (CI) (c) Argon (Ar) Relerences Identify the corresponding elements that have ground state electron configurations as specificd. Use either full element names, e.g. Bromine, or the chemical symbol, e.g. Br. (a) 1s2 (b) Is*2s 2p°3s*3p° Write a Lewis structure for ethyl alcohol, CH,CH,OH, showing all valence electrons. • You do not have to consider stereochemistry. Explicitly draw all H atoms. Include all valence lone pairs in your answer. Raterences ich atom, if any, in the shown covalent bonds acquires a partial NEGATIVE charge? cify the correct atom by its chemical symbol, e.g. Br. If none of them bears a negative charge, write, "none answer this question you may consult the table of electronegativity values in the table below.) 1A 2A 2.1 3A 4A 5A 6A 7A Be 1.0 1.5 Na Mg 09 12 3B 4B 5B 6B 7B Li 2.0 2.5 3,0 3.5 4.0 Al Si P. IB 2B 1.5 1.8 2.1 2,5 3.0 8B CI Cr Mn Fe 1.6 1.6 1.5 1.8 1.8 Co Ni Cu Zn 1.8 1.9 1.6 Ga Ge K Ca 0.8 Se Ti 1.3 Se Br 2.4 2.8 As 1.0 1.5 1.6 1.8 2.0 Zr 08 1.0 1.2 14 Nb Mo Te Pd Ag Cd 1.9 Rh Te Sb 1.9 2.1 2.5 Sr Rh In Sn 1.7 1.7 1.8 Ru 1.6 1.8 1.9 2.2 2.2 2.2 Au Hg TI Cs Bs 0.7 09 1.1 La Hr Ta 1.3 1.5 1.7 1.9 2.2 22 2.2 2.4 Ir Pr Pb Bị 1.8 Po At 1.9 2.0 2.2 W Re Os 1.9 1.8 <1.0 1.5-19 2.5-29 1.0-1.4 2.0-2.4 3.0-4.0 (a) I-1 (b) C-F How many electrons are in the valence shell of each atom ? (a) Aluminum (AI) (b) Chlorine (CI) (c) Argon (Ar) Relerences Identify the corresponding elements that have ground state electron configurations as specificd. Use either full element names, e.g. Bromine, or the chemical symbol, e.g. Br. (a) 1s2 (b) Is*2s 2p°3s*3p° Write a Lewis structure for ethyl alcohol, CH,CH,OH, showing all valence electrons. • You do not have to consider stereochemistry. Explicitly draw all H atoms. Include all valence lone pairs in your answer. Raterences ich atom, if any, in the shown covalent bonds acquires a partial NEGATIVE charge? cify the correct atom by its chemical symbol, e.g. Br. If none of them bears a negative charge, write, "none answer this question you may consult the table of electronegativity values in the table below.) 1A 2A 2.1 3A 4A 5A 6A 7A Be 1.0 1.5 Na Mg 09 12 3B 4B 5B 6B 7B Li 2.0 2.5 3,0 3.5 4.0 Al Si P. IB 2B 1.5 1.8 2.1 2,5 3.0 8B CI Cr Mn Fe 1.6 1.6 1.5 1.8 1.8 Co Ni Cu Zn 1.8 1.9 1.6 Ga Ge K Ca 0.8 Se Ti 1.3 Se Br 2.4 2.8 As 1.0 1.5 1.6 1.8 2.0 Zr 08 1.0 1.2 14 Nb Mo Te Pd Ag Cd 1.9 Rh Te Sb 1.9 2.1 2.5 Sr Rh In Sn 1.7 1.7 1.8 Ru 1.6 1.8 1.9 2.2 2.2 2.2 Au Hg TI Cs Bs 0.7 09 1.1 La Hr Ta 1.3 1.5 1.7 1.9 2.2 22 2.2 2.4 Ir Pr Pb Bị 1.8 Po At 1.9 2.0 2.2 W Re Os 1.9 1.8 <1.0 1.5-19 2.5-29 1.0-1.4 2.0-2.4 3.0-4.0 (a) I-1 (b) C-F How many electrons are in the valence shell of each atom ? (a) Aluminum (AI) (b) Chlorine (CI) (c) Argon (Ar) Relerences Identify the corresponding elements that have ground state electron configurations as specificd. Use either full element names, e.g. Bromine, or the chemical symbol, e.g. Br. (a) 1s2 (b) Is*2s 2p°3s*3p° Write a Lewis structure for ethyl alcohol, CH,CH,OH, showing all valence electrons. • You do not have to consider stereochemistry. Explicitly draw all H atoms. Include all valence lone pairs in your answer. Raterences ich atom, if any, in the shown covalent bonds acquires a partial NEGATIVE charge? cify the correct atom by its chemical symbol, e.g. Br. If none of them bears a negative charge, write, "none answer this question you may consult the table of electronegativity values in the table below.) 1A 2A 2.1 3A 4A 5A 6A 7A Be 1.0 1.5 Na Mg 09 12 3B 4B 5B 6B 7B Li 2.0 2.5 3,0 3.5 4.0 Al Si P. IB 2B 1.5 1.8 2.1 2,5 3.0 8B CI Cr Mn Fe 1.6 1.6 1.5 1.8 1.8 Co Ni Cu Zn 1.8 1.9 1.6 Ga Ge K Ca 0.8 Se Ti 1.3 Se Br 2.4 2.8 As 1.0 1.5 1.6 1.8 2.0 Zr 08 1.0 1.2 14 Nb Mo Te Pd Ag Cd 1.9 Rh Te Sb 1.9 2.1 2.5 Sr Rh In Sn 1.7 1.7 1.8 Ru 1.6 1.8 1.9 2.2 2.2 2.2 Au Hg TI Cs Bs 0.7 09 1.1 La Hr Ta 1.3 1.5 1.7 1.9 2.2 22 2.2 2.4 Ir Pr Pb Bị 1.8 Po At 1.9 2.0 2.2 W Re Os 1.9 1.8 <1.0 1.5-19 2.5-29 1.0-1.4 2.0-2.4 3.0-4.0 (a) I-1 (b) C-F How many electrons are in the valence shell of each atom ? (a) Aluminum (AI) (b) Chlorine (CI) (c) Argon (Ar) Relerences Identify the corresponding elements that have ground state electron configurations as specificd. Use either full element names, e.g. Bromine, or the chemical symbol, e.g. Br. (a) 1s2 (b) Is*2s 2p°3s*3p°
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
You do not have to agree with Bogles planning strategy. Playing the devils advocate (one who argues for the opposite viewpoint), persuasively present another perspective. (You may want to do some...
-
Write a Lewis structure for each of the following organic molecules All the Diagrams are included in the Solution
-
For the previous question, assume that you do not have sufficient savings to cover the entire amount required to start your sun-block business. You are going to have to get external financing. A...
-
Della Simpson Inc. sells two popular brands of cookies, Dellas Delight and Bonnys Bourbon. Dellas Delight goes through the Mixing and Baking Departments and Bonnys Bourbon, a filled cookie, goes...
-
Prescott Co. management has committed to a plan to dispose of a group of assets associated with the manufacture of railroad cars. This group of assets qualifies as a component of an entity for...
-
Suppose Donahue Brothers purchases $ 500,000 of 6% annual bonds of Clarkson Corporation at face value on January 1, 2014. These bonds pay interest on June 30 and December 31 each year. They mature on...
-
Presented below are summary financial data from the Pfizer, Inc., 2015 annual report. Using the ratio definitions from Exhibit 4.6, calculate the following ratios: return on equity, return on assets...
-
Prepare a physical DFD based on the output from Problem 4-4. Use Microsoft Visio or other diagraming software.
-
Civil Rights & Civil Liberties : How are civil rights different from civil liberties? How might the United States be different today if the Fourteenth Amendment had not been ratified and added to the...
-
Julio and Milania are owners of Falcons Corporation, an S corporation. They each own 50 percent of Falcons Corporation. In year 1, Julio and Milani received distributions of $20,000 and $10,000,...
-
A firm has Free Cash Flow to Equity (FCFE) of $1.3 billion. Their Weighted Average Cost of Capital (WACC) is 11%, and the firm's Cost of Equity is 13%. FCFE is expected to increase indefinitely by...
-
The company found that the current net operating income is equal to 16% of current sales revenue. What is the company's current Margin of Safety (in dollars)?
-
Provide services to customers on account for $42,000. Purchase equipment by signing a note with the bank for $34,000. Repay $27,000 of the note in (2) above. Pay utilities of $4,200 for the current...
-
Lauder Company manufactures and distributes various fixtures used primarily in new building construction. At the company's Bayside plant, Lauder produces two models of one widely used fixture...
-
Case Questions: Recall from class that components (in the WACC formula) are based on the (long-term) target debt-equity ratio. Assumptions: The beta of debt (for both Midland and its divisions) is...
-
The first production department of Stone Incorporated reports the following for April Direct Materials Conversion Units Beginning work in process inventory 79,000 Percent Complete 80% Percent...
-
eBook Break-even sales The Boston Beer Company Inc. (SAM) produces Samuel Adams beer and other alcoholic beverages. Boston Beer reported the following operating information for a recent year (in...
-
You are planning to purchase your first home five years from today. The required down payment will be $50,000. You currently have $20,000. but you plan to contribute $500 each quarter to a special...
-
Pentafluorophenol is readily prepared by heating hexafluorobenzene with potassium hydroxide in tert-butyl alcohol: What is the most reasonable mechanism for this reaction? Comment on the comparative...
-
Cyclopentyl methyl ketone has been prepared from 1,4-dibromobutane and ethyl acetoacetate. Outline the steps in this synthesis by writing a series of equations showing starting materials, reagents,...
-
Write a series of equations describing a synthesis of 1-bromo-2- methyl-2-propanol from tert-butyl bromide.
-
Fayols principle of esprit de corps refers to: A. Subordination of individual interest to the common good. B. Development of a high level of employee morale. C. Encouragement of initiative and risk...
-
Dee Hock recommends that managers spend what percentage of their time managing the employees for whom they have direct responsibility? A. 0%. B. 50%. C. 75%. D. 100%.
-
According to Herzbergs theory, which job factor would be considered a motivator? A. Money. B. Vacation policies. C. Quarterly parties. D. Recognition of achievement through a clinical ladder.

Study smarter with the SolutionInn App



