Fifty mol% propylene in propane is separated with silica gel. The products are to be 90 mol%
Question:
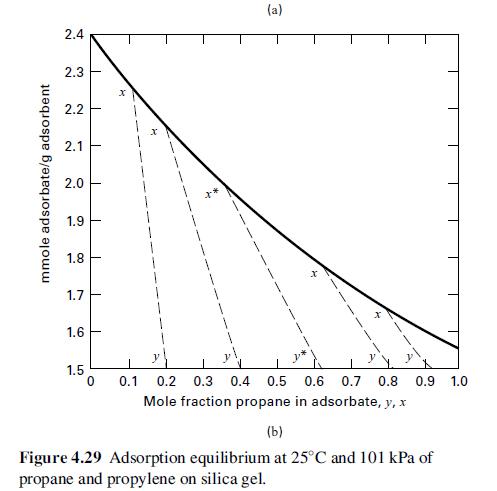
Fifty mol% propylene in propane is separated with silica gel. The products are to be 90 mol% propylene and 75 mol% propane. If 1,000 lb of silica gel/lbmol of feed gas is used, can the desired separation be made in one stage? If not, what separation can be achieved? Use Figure 4.29.
Transcribed Image Text:
mmole adsorbate/g adsorbent 2.4 2.3 2.2 2.1 2.0 1.9 1.8 1.7 1.6 1.5 T T 1 1 1 I 1 0 X (a) 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 Mole fraction propane in adsorbate, y, x (b) Figure 4.29 Adsorption equilibrium at 25°C and 101 kPa of propane and propylene on silica gel.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
To determine if the desired separation of 50 mol propylene in propane into 9...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Separation Process Principles Chemical And Biochemical Principles
ISBN: 9780470481837
3rd Edition
Authors: By J. D. Seader, Ernest J. Henley, D. Keith Roper
Question Posted:
Students also viewed these Life Sciences questions
-
A gas containing 50 mol% propylene in propane is to be separated with silica gel having the equilibrium properties shown in Figure. The final products are to be 90 mol% propylene and 75 mol% propane....
-
One lbmol of methane (CH4) undergoes complete combustion with stoichiometric amount of air in a rigid container. Initially, the air and methane are at 14.4 psia and 77oF. The products of combustion...
-
Propane gas is burned steadily at 1 atm pressure with a 10 percent excess oxygen supplied by atmospheric air. The reactants enter a steady flow combustor at 25C. Determine the final temperature of...
-
On January 1, 2012, Albert invested $1,000 at 6 percent interest per year for three years. The CPI (times 100) on January 1, 2012, stood at 100. On January 1, 2013, the CPI was 105; on January 1,...
-
Covariance of estimated population totals in a cluster sample. Suppose a one-stage cluster sample is taken from a population of N psus, with inclusion probabilities Ïi. Let tx and ty be the...
-
A simple soft drink system relies on pressurized CO 2 to force the soft drink (sg = 1.08) from its tank sitting on the floor up to the outlet where cups are filled. Determine the required CO 2...
-
Calculate \(\Delta G^{0}\) for the hydrogenation of ethylene at \(298 \mathrm{~K}\) using the following data: \(\Delta G_{f, 298}^{0}\) for \(\mathrm{C}_{2} \mathrm{H}_{4}=68.12 \mathrm{~kJ} /...
-
Finney Inc. has conducted an analysis of overhead costs related to one of its product lines using a traditional costing system (volume-based) and an activity-based costing system. Here are its...
-
Calculate Clean Price Given inputs. i . Instrument name: Security A ii . Issuer description: Issuer designs and manufactures wristwatches and timepieces for men and women. It sells its watches...
-
Determine the internal normal force, shear force, and moment at points E and F of the compound beam. Point E is located just to the left of 800 N force. 1200 N 800 N 400 N/m 3. A |E B F -1.5 m--1 m-2...
-
A liquid of 30 mol% toluene, 40 mol% ethylbenzene, and 30 mol % water is subjected to a continuous flash distillation at 0.5 atm. Assuming that mixtures of ethylbenzene and toluene obey Raoults law...
-
A vapor mixture of equal volumes NH 3 and N 2 is contacted at 20 C and 1 atm (760 torr) with water to absorb some of the NH 3 . If 14 m 3 of this mixture is contacted with 10 m 3 of water, calculate...
-
The Hermite polynomials are orthogonal with respect to the inner product Find the first five monic Hermite polynomials. 00 (f. 8) = ! f(1)8(1) e-* dr. dt. J-00
-
you will assume the role of Chief Financial Officer of Monsanto Corporation. Bayer is considering a bid for United States seed company Monsanto. Assume that relevant tax rate is 35%. EBIT,...
-
Calculate the treynor, sharpe, and jensen measures with a portfolio beta of 1.12 and a risk free rate of 2.90 Symbol Description AAPL Apple Inc ALA ALTAGAS INCOME TRUST AMC Entertainment Holdings...
-
Please give answer on urgent base Read the following case scenario and Prepare a 1,500 word financial report ASSIGNMENT SCENARIO: Faisal and Sarah are in their late 50's and considered themselves...
-
Find lim 0 2 sin(20) 1 - sin(0)
-
QUESTION 3 [20 Marks] 1. Figure 1 illustrates the short-run profit/loss condition of a typical firm in a given market. Figure 1 P 245 210 175 0 129 MC ATC AVC D (3) (1) (1) (1) (a) Calculate the...
-
Find all eigenvalues and eigenvectors of (a) The n x n zero matrix O; (b) The n x n identity matrix I.
-
Use the information given about the angles and to find the exact value of: (a) sin( + ) (b) cos( + ) (c) sin( - ) (d) tan ( + ) (e) sin(2) (f) cos (2) (g) sin /2 (h) cos/2 cos = 4/5, 0 < < /2; cos =...
-
How do the RNA molecules of these two viruses differ in number and function? In your response, consider the role of both in the formation of mRNA.
-
A single virus particle of influenza A is insufficient to initiate disease in humans. It is estimated that between 100 and 1,000 influenza A virus particles are necessary to cause the flu in an...
-
Compare and contrast the reproductive life cycle of WNV (See the following figure) to that of HIV. The virus binds to a protein on the surface of a cell (not shown here) and enters the cell. e The...
-
Is China's rapid economic growth, spurred by Deng Xiaoping's reforms in the 80s, sustainable in the long term, or does the current threat of deflation indicate potential risks that could undermine...
-
The market for Big Macs in Canada is initially in equilibrium with supply and demand curves of the usual shape. Whoppers are a close substitute for Big Macs; Fries and Big Macs are complements. Use...
-
The COVID-19 pandemic has adversely affected the lives of people around the world in millions of ways. Due to this severe epidemic, all countries in the world have been affected by all aspects,...

Study smarter with the SolutionInn App


