The stream below is flashed adiabatically across a valve. Conditions upstream are 250F and 500 psia, and
Question:
The stream below is flashed adiabatically across a valve. Conditions upstream are 250F and 500 psia, and downstream are 300 psia. Compute using a process simulator the:
(a) Phase condition upstream of the valve;
(b) Temperature downstream of the valve;
(c) Molar fraction vaporized downstream of the valve; and
(d) Molefraction compositions of the vapor and liquid phases downstream of the valve.
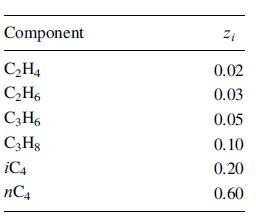
Transcribed Image Text:
Component C₂H4 C₂H6 C3H6 C3H8 iC4 nC4 Zi 0.02 0.03 0.05 0.10 0.20 0.60
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
a Phase condition upstream of the valve 43 b Temperature downstream of the valve 478...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Separation Process Principles Chemical And Biochemical Principles
ISBN: 9780470481837
3rd Edition
Authors: By J. D. Seader, Ernest J. Henley, D. Keith Roper
Question Posted:
Students also viewed these Life Sciences questions
-
A vaporliquid mixture at 250F and 500 psia contains N 2 , H 2 S, CO 2 , and all the normal paraffins from methane to heptane. Use Figure 2.4 to estimate the K-value of each component. Which...
-
A hydrocarbon vapor-liquid mixture at 250?F and 500 psia contains N2, H2S, CO2, and all the normal paraffins from methane to heptane. Use Figure to estimate the K-value of each component in the...
-
A pipe carries steam as a two phase liquid vapor mixture at 2.0 MPa. A small quantity is withdrawn through a throttling calorimeter, where it undergoes a throttling process to an exit pressure of 0.1...
-
Did Hopkins materially misrepresent his health when applying for insurance? Does Golden Rule have the right to rescind his insurance policies?
-
In Example 10.5 we used linearization to estimate V ().We can alternatively use the random group method to estimate the variance of . Calculate 11 22 12 21 for each of the seven random groups,...
-
The graphs in Fig. P.4.98 are the reflection spectra for several roses seen in white light. The flowers were white, yellow, light pink, dark pink, blue, orange, and red. Associate each graph with a...
-
The change in Gibbs' free energy for vaporization of a pure substance is (a) + ve (b) - ve (c) 0 (d) Either + ve or - ve.
-
Hart Computer Company has been purchasing carrying cases for its portable computers at a delivered cost of $68 per unit. The company, which is currently operating below full capacity, charges factory...
-
Determine the following amounts for Sprouts Ending cash balance in 2020 Ending cash balance in 2021 Increase (decrease) in cash from 2020 to 2021 Determine the following amounts for Sprouts in 2021...
-
Following are separate financial statements of Michael Company and Aaron Company as of December 31, 2013 (credit balances indicated by parentheses). Michael acquired all of Aarons outstanding voting...
-
What is the difference between adsorbent and adsorbate?
-
In crystallization, what is a eutectic? What is mother liquor? What are hydrates?
-
Consider the problem Maximize z = x1 + x2 Subject to 2x1 + x2 6 X1 + 2x2 6 X1 + x2 0 (a) Show that the optimal basic solution includes both x 1 and x 2 and that the feasibility ranges for the two...
-
A single - pass shell and tube counter flow heat exchanger uses waste gas on the shell side to heat a liquid in the tubes. The waste gas enters at a temperature of 400C at a mass flow rate of 40...
-
Q1 (a) (b) Use Hooke's law to derive an expression for the shear modulus G in terms of Young's modulus E and the Poisson's ratio v. This relationship sets a limit for admissible values of Poisson's...
-
Consider the dissolution of Calcite [CaCO3] in an aqueous solution: CaCO3 Ca+ + CO3- Does this reaction proceed spontaneously in the direction written? Explain! Species CaCO3 Ca+ CO2- AH [kcal/mole]...
-
In the combustion of Hydrogen, the following reactions involving radicals are fast in both the forward and backward directions K H+O OH+O KD k2f O + H2 OH+H Kb HO+H OH+H K3b Using that assumption of...
-
5. [20 Points] Consider the chemical reaction 3CO + 2.502 + 8 N XCO + YCO + ZO + 8N2 a. Determine the coefficients x, y, and z if an equilibrium composition exists at 2600 K and 3 atm. b. Does the...
-
Write out the singular value decomposition (8.40) of the matrices in Exercise 8.5.1. Matrices in Exercise 8.5.1 (a) (b) (c) (d) (e) (f) 2 0 0 1 (4 2) 2 0 2 1 01 0- 1 011
-
How do network effects help Facebook fend off smaller social-networking rivals? Could an online retailer doing half as much business compete on an equal footing with Amazon in terms of costs? Explain.
-
DNA-repair systems are responsible for maintaining genomic fidelity in normal cells despite the high frequency with which mutational events occur. What type of DNA mutation is generated by (a) UV...
-
What is the name given to the process that can repair DNA damage and generate genetic diversity? Briefly describe the similarities and differences of the two processes.
-
The genome of a retrovirus can integrate into the host-cell genome. What gene is unique to retroviruses, and why is the protein encoded by this gene absolutely necessary for maintaining the...
-
The sheet Inventory lists a grocery store's inventory for two months. Calculate the percent change for each item to two decimal places. If the formula gives an error, put "Initial Stock" in the cell....
-
A local manufacturer produces two different products on the same production line. Product A requires .5 hours of labor while product B requires .8 hours of labor. The total labor available in any one...
-
Hampton Company reports the following information for its recent calendar year. Income Statement Data Sales Expenses: Cost of goods sold Salaries expense Depreciation expense Net income Required:...

Study smarter with the SolutionInn App


