Calculate the self-diffusion coefficient in the primitive cubic -Po (Figure 1.23; Figure 1.23 3.36 ) at 500
Question:
Calculate the self-diffusion coefficient in the primitive cubic α-Po (Figure 1.23;
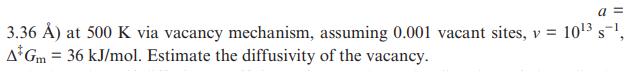
Figure 1.23

Transcribed Image Text:
3.36 ) at 500 K via vacancy mechanism, assuming 0.001 vacant sites, v = 10 s, A Gm = 36 kJ/mol. Estimate the diffusivity of the vacancy. a=
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Equation 310 Equation 318 Po has Nn 6 nearest neighbors and the probability pavail th...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
The drug patch shown in the figure in the next column releases a water-soluble epidermal growth factor (species A) to repair a specific region of wounded tissue on the human body. A slow release of...
-
The Arnold Diffusion Cell shown in Figure 26.5 is a simple device used to measure gas-phase diffusion coefficients for volatile substrates in air. In the present experiment, liquid acetone is loaded...
-
The mass transfer device shown in the figure at the top of the next column is used to carry out the controlled release of a vapor-phase pheromone drug used in pest control. The solid drug sublimes at...
-
Why is ultraviolet light, but not infrared light, effective in making certain materials fluoresce?
-
Determine the moment of inertia and the radius of gyration of the shaded area with respect to the y axis. 10 mm- - 10 mm 50 inm C. 50 mm 10 mm 90 mm
-
A put option with a strike price of $50 sells for $3.20. The option expires in two months and the current stock price is $51. If the risk-free interest rate is 5 percent, what is the price of a call...
-
Discuss the sampling strategy and technique to be used. Develop a hypothetical research scenario that would necessitate the use of the Action Research Approach and a Posttest With a Historical...
-
Pension Plan Financial Statements. The State of Nodak operates a Public Employees Retirement System (PERS) for all employees of the state. The preclosing trial balance of the PERS as of June 30,...
-
Let M be a metric space. If {x} and {y} are sequences in M such that x x and y y, show that d(xn, yn) d(x,y).
-
Calculate the self-diffusion coefficient of -Po along the direction of the cell edge (Figure 1.23; a = 3.36 ) at 500 K via an interstitial mechanism with full availability of interstitial sites,...
-
Verify the statement that p dir 2 = 2 /6 = a 2 /N n , in which the distance a is the unit-cell edge, the actual jump length, its projection onto the unit cell edge direction, and N n is the...
-
Suppose that in a certain drug the concentration of a particular chemical is a random variable with a continuous distribution for which the p.d.f. g is as follows: Suppose that the concentrations X...
-
Textotape Inc. (the "Company") is the largest and most technically advanced producer of pressure-sensitive labels and associated products in Texas. Its sales volume in the fiscal year ended December...
-
Provide a recommendation for improvement if Instructions to Assessors on how to assess candidates are not clear and concise such as missing information regarding reasonable adjustment, LLN levels for...
-
Benchmarking in Risk Management: Refer to Chapter 3 in the required text and create your blog entry that demonstrates your understanding of how benchmarking is used to identify risk. Can benchmarking...
-
Accurately reflecting the revised depreciation expense in financial statements is important. Inaccurate financial statements can mislead investors and stakeholders. How might investors react if they...
-
UML is a publicly traded corporation that makes various control devices used in manufacturing mechanical equipment. Joe is the president of UML and Tim is the purchasing agent, both have worked at...
-
The following information pertains to Family Video Company. 1. Cash balance per bank, July 31, $8,228.98. 2. July bank service charge not recorded by the depositor $31.72. 3. Cash balance per books,...
-
Below is a sample of the data in the file NFLAttendance which contains the 32 teams in the National Football League, their conference affiliation, their division, and their average home attendance....
-
Propose the structure of a compound consistent with the following data: (a) C 5 H 10 O, broadband-decoupled 13 C NMR: 7.1, 34.6, 210.5 (b) C 6 H 10 O, broadband-decoupled 13 C NMR: 70.8, 116.2, 134.8
-
Determine the structure of an alcohol with molecular formula C 6 H 14 O that exhibits the following DEPT-135 spectrum: DEPT-135 70 60 50 40 Chemical Shift (ppm) 30 20 10
-
Deduce the structure of a compound with molecular formula C 6 H 14 O 2 that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. 100 80 60 40 20 3000 2500 Wavenumber (cm-1) 4000 3500 2000 1500...
-
On January 1, 20X8, Potter Corporation acquired 90 percent of Shoemaker Companys voting stock, at underlying book value. The fair value of the noncontrolling interest was equal to 10 percent of the...
-
Jones Industries received $800,000 from issuing shares of its common stock and $700,000 from issuing bonds. During the year, Jones Industries also paid dividends of $90,000. How are the effects of...
-
Barnes Company reports the following operating results for the month of August: sales $320,000 (units 5,000); variable costs $216,000; and fixed costs $70,500. Management is considering the following...

Study smarter with the SolutionInn App


