GaAs (E g = 1.4 eV) and ZnSe (E g = 2.6 eV) are isoelectronic with Ge.
Question:
GaAs (Eg = 1.4 eV) and ZnSe (Eg = 2.6 eV) are isoelectronic with Ge. Their band structures are shown below. Are they direct- or indirect-gap semiconductors?
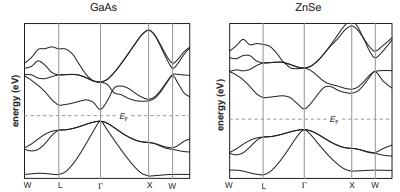
Transcribed Image Text:
energy (ev) W L GaAs r X W energy (eV) W ZnSe T X W
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
For each compound the vale...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
The red pigment vermillion (HgS) is a semiconductor with a band gap of 2.0 eV. It was replaced in the late nineteenth and early twentieth century by CdS 1x Se x pigments that are less toxic and more...
-
In many semiconductors such as GaAs, the conduction band has a conduction band minimum at zone center, and an indirect gap with higher energy at another minimum, at a critical point on the zone...
-
Which semiconductors in Table 7.8 will have colors other than white or black? For each compound in your list predict the approximate color. Table 7.8 Si Ge Elemental semiconductors Bond Band gap...
-
Describe how you plan to allocate your budget to each promotional activity. Provide a brief rationale. First, when developing a marketing budget plan, you want to start with the total budget...
-
Using Mohrs circle, determine for the quarter ellipse of Prob. 9.67 the moments of inertia and the product of inertia with respect to new axes obtained by rotating the x and y axes about O (a)...
-
The Gladys Corporation buys office equipment costing $658,000 on May 12, 2017. In 2020, new and improved models of the equipment make it obsolete, and Gladys sells the old equipment for $34,000 on...
-
John and Jennifer Margeson entered into a contract to sell a weight-loss franchise business called Inches-A-Weigh to Theresa Artis. The parties memorialized their agreement in an Asset Purchase...
-
Sales of industrial vacuum cleaners at R. Lowenthal Supply Co. over the past 13 months are as follows: (a) Using a moving average with three periods, determine the demand for vacuum cleaners for next...
-
1) Write a Python function to convert mass values from gram to ounce and return the conversion result. The conversion formula is ounce = gram x 0.035274. The function header is: def gm_to_ounce...
-
Consider LaCrSb 3 , whose structure is shown below (left). To a reasonable approximation, this structure can be described as independent CrSb 2 2 layers and Sb layers separated by La 3+ ions [3]. If...
-
NbN crystallizes with the NaCl type structure. Sketch out an MO diagram for octahedrally coordinated niobium and use it to sketch a DOS plot for NbN. Indicate the relative area of the *, *, and N 2p...
-
The direct material cost of manufacturing a product is $103.95, direct labour cost is $46.20, and overhead is $57.75. (a) What is the percent content of each element of cost in the product? (b) What...
-
Susan has $180,000 to invest and is in the 35% tax bracket. Charles, Susan's son, is in the 15% tax bracket. Instead of investing the funds herself, she loans the $180,000 to Charles to invest on...
-
Mrs. Jimenez makes monogrammed fleece blankets. It costs her$5.95 for the fabric for each blanket and $175 per month to rentthe embroidery machine. She sells the blankets for $25.95 each.Find b, the...
-
Suburbia Company manufactures a product through a continuous single-step process. All materials are added at the beginning of processing. Production and cost data for the company for February 2016...
-
John Slow is driving from Boston to the New York area, a distance of 180 miles ata constant speed, whose value is uniformly distributed between 30 and 60 miles per hour.What is the PDF of the...
-
A donut store has 15 different types of donuts. You can only buy a bag of 3 of them, where each donut has to be of a different type from the rest. How many different bags of 3 can you make?
-
Selected sales and operating data for three divisions of different structural engineering firms are given asfollows: Sales Average operating Net operating income Minimum required rate of retur...
-
Gopher, Inc. developing its upcoming budgeted Costs of Quality (COQ) with the following information: Expense Item Budget Raw Materials Inspection $ 15,000 EPA Fine 200,000 Design Engineering 15,000...
-
Determine the electron configuration for each of the following ions: a. A carbon atom with a negative charge b. A carbon atom with a positive charge c. A nitrogen atom with a positive charge d. An...
-
Identify which of the following two reactions you would expect to occur more rapidly: (1) addition of HBr to 2-methyl-2- pentene or (2) addition of HBr to 4-methyl-1-pentene. Explain your choice.
-
With current spectroscopic techniques (discussed in Chapters 1517), chemists are generally able to determine the structure of an unknown organic compound in just one day. These techniques have only...
-
Printing Plus, Lynn Sanders' printing service company. We will analyze and record each of the transactions for her business and discuss how this impacts the financial statements. More detail for each...
-
You are an expatriate employee completing your overseas assignment and you just received your final performance appraisal. You feel it did not provide an accurate assessment of your performance and...
-
Landscape, Inc., is a lawn and garden service. The company originally specialized in serving small residential clients; recently it has started contracting for work on larger office building grounds....

Study smarter with the SolutionInn App


