Prove that Equation (15.6) reduces to the LorenzLorentz equation (15.4) in the case of purely ionic bonding.
Question:
Prove that Equation (15.6) reduces to the Lorenz–Lorentz equation (15.4) in the case of purely ionic bonding.
Equation (15.6)
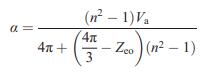
Equation (15.4)
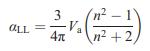
Transcribed Image Text:
a = 4x + (n - 1) Va 4 3 -Zeo (1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Purely ionic bonding implie...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
To calculate variance and standard deviation, we take the deviations from the mean. At times, we need to consider the deviations from a target value rather than the mean. Consider the case of a...
-
A registered dealer, based in Chandigarh, makes a supply to another registered dealer located in Chandigarh, valuing rupees 1,20,000. The applicable rate of GST is 12%. Calculate the amount of tax...
-
In aqueous solution, hydrogen sulfide reduces (a) Fe3+ to Fe2+ (b) Br2 to Br- (c) MNO4- to Mn2+ (d) HNO3 to NO2. In all cases, under appropriate conditions, the product is elemental sulfur. Write a...
-
Suppose that the probability that a mechanic fixes a car correctly is 0.9. Determine the odds against the mechanic fixing a car correctly.
-
Calculate the Lande g factor for atoms (a) In S states; (b) In singlet states.
-
H&R Block Inc. provides tax preparation services throughout the United States and other parts of the world. These services are provided through two segments: company-owned offices and franchised...
-
The June 30, 2010, adjusted trial balance of Energized Espresso, Inc., is shown next. Requirements 1. Prepare the June closing entries for Energized Espresso, Inc. 2. Calculate the ending balance in...
-
Digital Software Inc., has two product lines. The income statement for the year ended December 31 shows the following: The products, Num 1 and Num 2, are sold in two territories, North and South, as...
-
Consider an existing capacity resource such as a machine whose capacity is measured in hours it can be realistically operated in a year. In some months, the machine is idle for a few hours because of...
-
Use the AndersonEggleton relationship to calculate the refractive index of the mineral orthoclase (KAlSi 3 O 8 ) with a unit-cell volume 720.4 3 containing four formula units. The cation...
-
Yellow light of the Na doublet with a wavelength of 589.30.3 nm and frequency of 5.0910 14 Hz in vacuum enters Fe 2 O 3 that has a refractive index of 3.00. Calculate the speed, wavelength, and...
-
a. Use the following data and = .10 to test the stated hypotheses. Assume x is normally distributed in the populations and the variances of the populations are approximately equal. H0: 1 - 2 = 0...
-
What is the price of a bond with a coupon rate of 4.50%, a yield to maturity of 4.25%, a future value of $1000, semi-annual coupon payments, and 9 years until maturity?
-
Explain the responsibilities of the chairperson in bringing meetings to a close.? What information should be covered when briefing the minute taker? A)What are minutes of meetings? (b) What purpose...
-
rader-Instructions tep roject Description: ou have been hired as a finance manager at Granger vacation homes, a regional construction company that finances nd builds vacation properties. As part of...
-
1. What are insulators and conductors? How are they different from one another? 2. What is a capacitor? How does it work? How is it different from a battery? 3. What are the different parts in a...
-
1: Conduct a resource search for an article about your future career. 2: Respond to several questions using information from the resource you selected in Part 1. The selected article? What is the...
-
Your firm uses a periodic review system for all SKUs classified, using ABC analysis, as B or C items. Further, it uses a continuous review system for all SKUs classified as A items. The demand for a...
-
During the month, services performed for customers on account amounted to $7,500 and collections from customers in payment of their accounts totaled $6,000. At the end of the month, the Accounts...
-
An alkyne with molecular formula C 5 H 8 was treated with sodium in liquid ammonia to give a disubstituted alkene with molecular formula C 5 H 10 . Draw the structure of the alkene.
-
Predict the major product expected for each of the following reactions: (a) (b) (c) (d) (e) (f) ? XS HCI CI 1) xs NANH/NH3 2) H,0 CI
-
Suggest reagents that would achieve the following transformation: CI CI CI
-
All animals are heterotrophs that ingest their food. Somebiologists have speculated that the first animal to actively huntwas a flatworm-like animal. How are flatworms different from otherearly...
-
Select at least three leadership theories evaluate their impact on professional relationships. discuss which leadership theories, characteristics, or competences you believe are necessary to be an...
-
Do you agree that self-awareness is essential for being a good leader? Can you think of some specific negative consequences that might result from a leader not having self-awareness? Leaders of many...

Study smarter with the SolutionInn App


