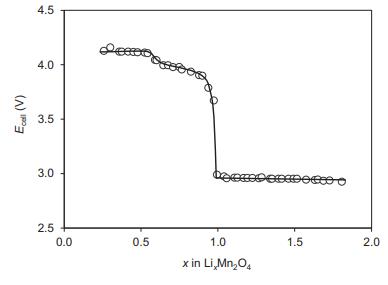
The figure below shows E against x at 30 C for a cell with a spinel-based LixMn
Question:
The figure below shows E against x at 30 °C for a cell with a spinel-based LixMn2O4 cathode (0
(a) Describe the structure you might expect for a LiMn2O4 spinel.
(b) State what can you conclude from Ecell versus x in the regions 0
(c) State what could cause the marked drop in Ecell at x = 1.
Transcribed Image Text:
Ecal (V) = 4.5 4.0 3.5 3.0 2.5 0.0 0.5 bet 1.0 x in Li,Mn>O4 1.5 2.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The electrochemical data are taken from Ohzuku et al J Electrochem Soc 137 1990 769775 a Spinel has ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
Figure out why the following induction should hold, and show that it does. The values have many beautiful mathematical properties. Here are just a few. In k- 1
-
Instructions: Assume you work in the accounting department of Caesars Entertainment and your supervisor, Logan Lacy, provides you with the above information and asks you to use the What-if Analysis...
-
Armidale Company Ltd (ALC) 2022 books contain the following information (budgeted): Activities Activity Cost Relevant cost driver Account Inquiry costs $198,000 Cost of Billing accounts (billing...
-
Find the mass and center of mass of the lamina that occupies the region D and has the given density function p. D is the triangular region with vertices (0, 0), (2, 1), (0, 3); (x, y) = x + y
-
Using the Hund rules, find the basic term of the atom whose partially filled sub shell contains (a) Three p electrons; (b) Four p electrons.
-
Represent in polar form and graph in the complex plane as in Fig. 325. Do these problems very carefully because polar forms will be needed frequently. Show the details. -4 + 4i pl+i /4
-
In 2014, Air Asia Flight No. 8501 crashed in the Java Sea while flying from Indonesia to Singapore. The crash resulted in the deaths of all passengers and crew on board. The plane involved in the...
-
Ideal Manufacturing Company of Sycamore, Illinois, has supported a research and development (R&D) department that has for many years been the sole contributor to the company's new farm machinery...
-
1. Solve the double inequality below. Then graph the solution set on the real number line. -81+3(x-2) < 13
-
Li 1 Mn 2 O 4 (a = 8.2495 ) is cubic, whereas Li 2 Mn 2 O 4 is tetragonal (a = 5.653 and c = 9.329 ). Suggest a reason for lower symmetry of Li 2 Mn 2 O 4 . Calculate the volume change upon its...
-
Calculate the energy density in W h/kg for a battery based on a Li x C 6 anode and a Li 1x CoO 2 cathode, operating at an average voltage of 3.6 V. Assume that the electrolyte and casing make up 50%...
-
The following extracts are taken from the website of the Chartered Institute of Management Accountants. The leading global professional body of Management Accountants. CIMA is the world's largest...
-
Considering the wars, terrorist attacks, and other violations of international law around the world, discuss the function of international law in regulating the relations between sovereign nations in...
-
What is the Present Value for $15,000 in 30 years at discount rate of 12%? XYZ mutual fund has an average 6% annual rate of return. How much money you could make in 20 years if you invest 80,000 at...
-
Why not think that women are better able to reflect critically on masculinity and male experience than men since they do not need to imagine the perspective of the oppressed? I think there is a lot...
-
Sea P(A) = 0,52, P(B) = 0,27 y P(A|B) = 0,47. una. Calcular P(ANB) b. Calcular P(AUB) C. Calcular P(BIA)
-
Provide the table below answer these questions. Income Balance Revenue $10,000,000 Cash $200,000 COGS $7,500,000 Accounts receivable $1,200,000 Gross profit $2,500,000 Inventory $400,000 Total...
-
A check drawn by Cullen for $500 is made payable to the order of Jordan and issued to Jordan. Jordan owes his landlord $500 in rent and transfers the check to his landlord with the following...
-
Parkin Industries, a U.S. company, acquired a wholly-owned subsidiary, located in Italy, at the beginning of the current year, for 200,000. The subsidiary's functional currency is the euro. The...
-
A 1.50 mole sample of an ideal gas at 28.5C expands isothermally from an initial volume of 22.5 dm 3 to a final volume of 75.5 dm 3 . Calculate w for this process a. For expansion against a constant...
-
Derive the equation (H/T) V = C V + V/ from basic equations and definitions.
-
Consider the reaction TiO 2 (s) + 2 C(graphite) + 2 Cl 2 (g) 2 CO(g) + TiCl 4 (l) for which ÎH o R ,298 K = 80. kJ mol 1 . Given the following data at 25°C, Assume that the heat capacities...
-
A company provides the following account balances at year end ( except for Retained Earnings, which is the beginning balance ) . The CFO has asked you to use this information to prepare an income...
-
As a winner of a local competition, you can choose one of the following prizes:(a) $100,000 now(b) $170,000 at the end of 4 years(c) $18,000 per year starting next year for 10 yearsWhich one is the...
-
1. Should prison inmates be allowed a free college education while noncriminals must pay tuition? Why or why not? Do you believe in less parole eligibility for prisoners? 2. Define parole, including...

Study smarter with the SolutionInn App


