Which of the two correlation diagrams shown below would be appropriate for tetrahedral [CoCl 4 ] 2
Question:
Which of the two correlation diagrams shown below would be appropriate for tetrahedral [CoCl4]2−? For the applicable correlation diagram, what are the correct term symbols for the lines labeled W, X, Y, and Z?
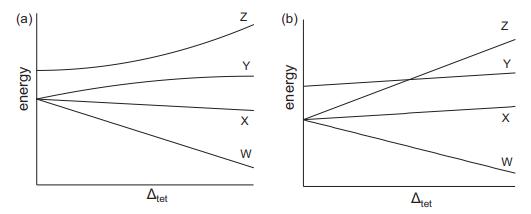
Transcribed Image Text:
(a) energy Atet energy Alet N Y X W
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Because the Cl ligands are negatively charged the Co oxidation state must be ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
The accompanying table gives the dry weights (Y) of 11 chick embryos ranging in age from 6 to 16 days (X). Also given in the table are the values of the common logarithms of the weights (Z). a....
-
Which of the following IRS pronouncement citations is INCORRECT: a. Notice 0710 2007 CB 324 b. Publication 519 c. Rev Proc 201114 2011 IRB 532 d. Action on Decision 2010002 e all of these citations...
-
Davida purchased a simple ordinary annuity that earns 2.4% annual interest. She will receive monthly payments of $250 over 10 years. What is the present value of her annuity?
-
What minimum force f , applied horizontally to the wheel axis, is needed to bring the wheel up to the height step h ? Tomer as the wheel radius and w as its weight. Tip: apply the equilibrium...
-
A packing crate of mass 40 kg is pulled by a rope as shown. The coefficient of static friction between the crate and the floor is 0.35. If α =40°, determine (a) The magnitude of the...
-
Employers are required to withhold social security taxes from wages paid to employees. What is the amount of the social security wage limitation for 2017? a. $127,200. b. $118,500. c. $113,700. d....
-
True or False: If MARR \(=10 \%\) and IRR \(=9 \%\), then ERR \( <10 \%\) and \(\mathrm{PW}>\$ 0\).
-
For the past several years, Shane Banovich has operated a part-time consulting business from his home. As of October 1, 2012, Shene decided to move to rented quarters and to operate the business as...
-
5. What effect do each of the following have on the regular payment and the total interest paid on a mortgage? Change Effect on Regular Payments (up/down) Effect on Total Interest Paid (up/down)...
-
For which of the following ions is the d-to-d transition spin-forbidden, Laporteforbidden, both or neither: (a) Tetrahedral Co 2+ , (b) Square-planar Cu 2+ , (c) Octahedral Mn 2+ (high spin), (d)...
-
The UVvisible spectrum of the octahedral complex [Ni(NH 2 CH 2 CH 2 NH 2 ) 3 ] 2+ is shown below. (a) Given that ethylenediamine ligand, NH 2 CH 2 CH 2 NH 2 , is a neutral bidentate ligand (both...
-
What is meant by the term culture? In what way can measuring attitudes about the following help to differentiate between cultures: centralized or decentralized decision making, safety or risk,...
-
Jack rides his scooter 155 m, stops and turns 38.0 to his left and goes 120 m. What is Jacks displacement?
-
Your factory has been offered a contract to produce a part for a new printer. The contract would last for 3 years and your cash flows from the contract would be $4 86 million per year. Your upfront...
-
Is Florida's zero-tolerance law implemented administratively?
-
The yield on a pure-discount instrument maturing in 200 days is equal to 6.5% per year, compounded annually. What would the yield on this instrument be equal to if it were quoted using the ACT/360...
-
You need to program the following classes: MyTravel.java - Class storing the "main" method. Travel.java - Superclass storing common travel information. AirTravel.java Subclass of Travel specific...
-
Presented below are a number of balance sheet accounts of Deep Blue Something, Inc. (a) Investment in Preferred Stock. (b) Treasury Stock. (c) Common Stock. (d) Cash Dividends Payable. (e)...
-
A copper rod of length L =18.0 in is to be twisted by torques T (see figure) until the angle of rotation between the ends of the rod is 3.08. (a) If the allowable shear strain in the copper is 0.0006...
-
Compound A has molecular formula C 5 H 10 . Hydroboration-oxidation of compound A produces an alcohol with no chirality centers. Draw two possible structures for compound A.
-
A perfectly insulating box is partially filled with water in which an electrical resistor is immersed. An external electrical generator converts the change in potential energy of a mass m which falls...
-
Predict the product(s) for each of the following reactions: a. b. c. d. e. f. Ni Pd
-
Classify the following medicines. Write OTC if they are Over-the-Counter- Drugs and Rx if they are Prescription Medicines. 1. Codeine 2. Fentanyl 3. Kremil-S 4. Morphine 5. Bonamine 6. Penicillin 7....
-
Where does C4 carbon fixation get its name, and what is its purpose? In plants that do C4 carbon fixation, where do the light-dependent reactions take place and where does the Calvin cycle take place?
-
The company is Walmart and show your reference! SWOT analysis thoroughly addresses the strengths, weaknesses, opportunities, and threats of the corporation, assessing how the company might maximize...

Study smarter with the SolutionInn App


