A nucleophile made by deprotonation of propyne is (a) :CH,CH;; (b) :HC=CH;; (c) :C=CH; (d) :C=CCH; (e)
Question:
A nucleophile made by deprotonation of propyne is
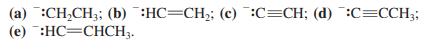
Transcribed Image Text:
(a) :CH,CH;; (b) :HC=CH;; (c) :C=CH; (d) :C=CCH; (e) :HC=CHCH3.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Here option d is the right ...View the full answer

Answered By

SUMAN MAHAPA
I am very keen in helping others to solve problems. I always try to teach in very simple & easy method, so that I can make teaching learning process easy and full of fun.Improvisation in study technique is my goal.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
The mechanism for acidic hydrolysis of a nitrile resembles the basic hydrolysis, except that the nitrile is first protonated, activating it toward attack by a weak nucleophile (water). Under acidic...
-
A somewhat unusual synthesis of cortisone-related steroids includes the following two reactions.
-
From what epoxide and what nucleophile colld each of the following compound be prepared Inppued? (Assums each is racemic.) C,H OH/H,O CH CH2 sodium azide
-
1. What trade- offs are involved in deciding to have a single large, centrally located facility instead of several smaller, dispersed facilities? 2. Who needs to be involved in facility location...
-
Build a WBS for the short case study in question 9 in Chapter 4. The project should be organized by short iterations, not trying to deploy all modules at the same time. You should first implement the...
-
Because the United States is more of a servicebased economy, manufacturing is not a contributor to the GDP. True or False
-
The Howell Corporation has the following account balances (in millions): Prepare an income statement and a supporting schedule of cost of goods manufactured for the year ended December 31, 2017. (For...
-
Lisah, Inc., manufactures golf clubs in three models. For the year, the Big Bart line has a net loss of $10,000 from sales $200,000, variable costs $180,000, and fixed costs $30,000. If the Big Bart...
-
When controlling for many factors that determine earnings differentials (age, occupation, education, etc.), there is still an 'unexplained' difference of 11% in earnings between Black and white...
-
Eagle Sports Supply has the following financial statements. Assume that Eagle's assets are proportional to its sales. INCOME STATEMENT, 2015 Sales .................. $950 Costs .................. 250...
-
The compound whose structure is H C C(CH 2 ) 3 Cl is best named (IUPAC) (a) 4-chloro-1-pentyne; (b) 5-chloropent-1-yne; (c) 4-pentyne-1-chloroyne; (d) 1-chloropent-4-yne.
-
When cyclooctyne is treated with dilute, aqueous sulfuric acid and HgSO4, a new compound results. It is best represented as () (b) () OH OH (d)
-
What types of meaning breakdowns have you experienced in your conversations with others? Be specific and identify the context, the situation, and the conversation topic.
-
Take a look at the C code (fa-pipe.c) shown below. Assume that all system and library calls succeed at runtime. (All error handling code has been omitted for brevity.) #include #include #include...
-
What are types of business disasters and why is disaster recovery important. In your example: What business disaster occurred? What was the impact of the business disaster? Did the company have a...
-
Justify why the taxonomies of behavioral concepts for managers and leaders differ.
-
A comparative income statement is given below for McKenzie Sales, Ltd., of Toronto: McKenzie Sales, Ltd. Comparative Income Statement This Year Last Year Sales $ 7,300,000 $ 5,548,000 Cost of goods...
-
1. Draft a thorough essay on "Consuming Food for Religion, Exploring Different Diets of Various Religions". a. Could you please assist me with writing an abstract for the paper "Consuming Food for...
-
Good, Inc., a 501(c)(3) exempt organization, engages in a 503 prohibited transaction. What negative tax consequences may be associated with Good engaging in a 503 prohibited transaction?
-
Respond to the ethical judgments required based on the following scenarios. Scenario 1. Assume you have collected a sample using MUS and that you have evaluated that sample to calculate a total...
-
The algebraic forms of the [orbitals are a radial function multiplied by one of the factors (a) z (5z2 - 3r2), (b) y (5y2 - 3r2), (c) x (5x2 - 3r2), (d) Z(x2 Y2), (e) Y(x2- z2), (f) x (z2 y2), (g)...
-
The NO2, molecule belongs to the group C2v' with the C2 axis bisecting the ONO angle. Taking as a basis the N2s, N2p, and 02p orbitals, identify the irreducible representations they span, and...
-
The phenanthrene molecule (29) belongs to the group C2v with the C, axis in the plane of the molecule. (a) Classify the irreducible representations spanned by the carbon 2pz orbitals and find their...
-
Convert the following decimal numbers to binary a. 256 b. 45 C. 9871
-
Explain the impact cultural diversity has on the group process in counseling.
-
task: write a 6-8 line algorithm for a program that accomplishes the following The program asks the user to input a numeric year, month, and day. The program then says what day of the week it was on...

Study smarter with the SolutionInn App


