A synthesis of the sesquiterpene farnesol requires the conversion of a dichloro compound into an alkynol, as
Question:
A synthesis of the sesquiterpene farnesol requires the conversion of a dichloro compound into an alkynol, as shown below. Suggest a way of achieving this transformation.
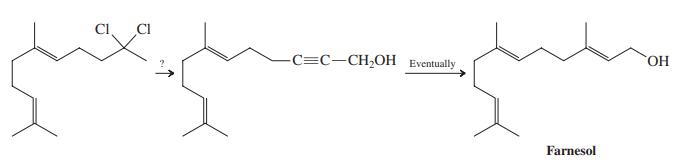
Transcribed Image Text:
CI. CI -C=C-CH;OH Eventually HO, Farnesol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
First we can use a bulky base such as Potassium tertbutoxide KOCH33 to ...View the full answer

Answered By

Akop Yepremyan
My aim would be to adequately walk through a given problem and find a solution through logic, rather than memorizing each question. With my strong background in tutoring and research as a PhD chemist from Texas Christian University (TCU) I will provide both theoretical and practical knowledge for your success.
I have been studying, teaching and researching in the field of chemistry for over 15 years. I have mentored and conducted multiple review sessions for students. Because of my extensive laboratory experience, I allow the student the visualize a given problem, thus making them much more enjoyable and easier to solve.
I am happy to teach you the fundamentals of chemistry in a way that will be very enjoyable for you and you are not counting the minutes to stop studying. Chemistry takes time to learn, I will make sure you push through the barrier that makes chemistry a difficult subject, and reach a level where you are very comfortable with solving any problem that is presented to you. I am available to help anytime and will respond to your need promptly.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Synthesis of the sesquiterpene bergamotene proceeds from the alcohol shown here. Suggest a sequence to complete the synthesis. HOH,C CH3 CH3 Bergamotene
-
Using benzene, acetic anhydride and 1-propanethiol as the source of all the carbon atoms, along with any necessary inorganic reagents, outline a synthesis of the compound shown.
-
Using benzene, acetic anhydride, and 1-propanethiol as the source of all the carbon atoms, along with any necessary inorganic reagents, outline a synthesis of the compound shown. 0 lonscn.oua CCH...
-
Estimates have been presented to Holly Farms, which is considering two environmental chambers for a project that will detail laboratory confirmations of on-line bacteria tests in chicken meat for the...
-
Build a WBS for one of the chapter case study projects other than asset management; either customer relationship management or Collection Management. The Project should use an iterative approach and...
-
Which marketing era occurred during the Great Depression when manufacturers almost had to force consumers to purchase their products? a. Production concept era b. Sales concept era c. Marketing...
-
A Pepsi promotion encouraged consumers to collect Pepsi points and redeem them for merchandise. If they did not have quite enough points for the prize they wanted, they could buy additional points...
-
KinderKids provides daycare for children Mondays through Fridays. Its monthly variable costs per child are as follows: Lunch and snacks ................. $ 100 Educational supplies ...................
-
A co-worker makes a joke with you about a client, saying they 'dress up' on the days you are on shift. The next week you receive a friend request on a social networking site from that client - do you...
-
Go-Go Industries is growing at 30% per year. It is all-equity-financed and has total assets of $1 million. Its return on equity is 25%. Its plowback ratio is 40%. a. What is the internal growth rate?...
-
Formulate a plausible mechanism for the hydration of ethyne in the presence of mercuric chloride. MR 3 H IH 1H 1 H (CH3)4Si 6.0 5.5 5.0 4.5 4.0 3.5 3.0 2.5 2.0 1.5 1.0 0.5 0.0 300-MHz 'H NMR...
-
Your team is studying the problem of an intramolecular ring closure of enediyne systems important in the total synthesis of dynemicin A, which exhibits potent antitumor activity. One research group...
-
Explain why the elementary row operations of types #2 and #3 do not change the corange of a matrix.
-
Job satisfaction is a fundamental concept of ETHICS. Explain why job satisfaction is considered an important aspect of the design process. Describe the five different aspects that job satisfaction is...
-
Sheridan Express is a coffee bean roaster who specializes in dark roasting beans grown in California. During the month, Sheridan added the following costs: Conversion Costs (CC), $5450; and Direct...
-
At t = 0, the instantaneous position of two pulses moving along a taut string with a speed v = 1.82 cm/s are as shown in the diagram below. Each unit on the horizontal axis is 5.0 cm and each unit on...
-
One of the objectives of an infrastructure management system IMS is to provide information for deciding whether to maintain, rehabilitate, or replace. Two other objectives are to plan and schedule...
-
Explain why the first meeting of the team is important for the team's success. Describe the approach you would implement to your initial meeting with your team: to meet collectively with its members...
-
Allied Fund, a charitable organization exempt under 501(c)(3), has branches located in each of the 50 states. Allied is not a private foundation. Rather than having each of the state units file an...
-
Eleni Cabinet Company sold 2,200 cabinets during 2011 at $160 per cabinet. Its beginning inventory on January 1 was 130 cabinets at $56. Purchases made during the year were as follows: February . 225...
-
The CIO2 molecule (which belongs to the group C, 2v) was trapped in a solid. Its ground state is known to be BJ Light polarized parallel to the y-axis (parallel to the 00 separation) excited the...
-
What states of (a) Anthracene, (b) Coronene (24) may be reached by electric dipole transitions from their (totally symmetrical) ground states? 24 Coronene
-
Determine whether the integral over /J and /, in Exercise 12.15a is zero over a symmetrical range about e = 0 in the group C3v
-
Apply the Test for Divergence to each series. (Remember that the only conclusions from the Test for Divergence are that the series diverges or the test is inconclusive.) (a) 6 cos (n + 4) 2 6 cos 3n...
-
Suppose that a firm manufactures espresso machines. The firm's fixed cost is $1650587, and the variable cost is $89 per machine. If each machine sells for $438, how many espresso machines need to be...
-
What are the advantages of cultural diversity within an organization and how can it gain a competitive advantage from the cultural differences?

Study smarter with the SolutionInn App


