Because of cyclic delocalization, structures A and B shown here for o-dimethylbenzene (o-xylene) are simply two resonance
Question:
Because of cyclic delocalization, structures A and B shown here for o-dimethylbenzene (o-xylene) are simply two resonance forms of the same molecule. Can the same be said for the two dimethyl-cyclooctatetraene structures C and D? Explain.
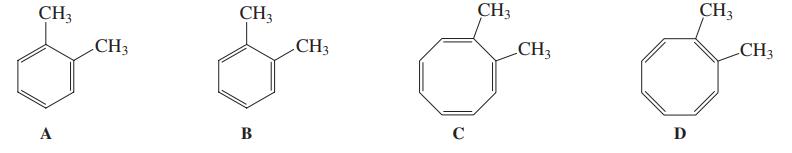
Transcribed Image Text:
CH3 CH3 CH3 CH3 CH3 CH3 CH3 CH3 A В C D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
As the structure a and b are two canonical forms of xylene but this is not true in c...View the full answer

Answered By

Saher bibi
I have done my Master of philosophy in the field of chemistry of natural products my major was (Isolation, Identification and Bioevaluation of natural products from medicinal plant) from Quaid-i-Azam University Islamabad, Pakistan, ranked as No. 1 university of Pakistan. I got scholarships and a laptop from the government of Pakistan on the basis of good academic performance
I am proficient in identification and screening of bioactive phytochemicals using HPLC-MS & MS/MS techniques. My M.phil research work consisted of identification of phytoconstituents by using HPLC MS/MS profiling, 80% of the compounds from the crude extract has been identified without the need for purification, which makes it a useful tool for screening and dereplication. Isolation and structure elucidation of secondary metabolites from Medicago Polymorpha for drug discovery using a combination of MS and NMR techniques. Identification of the metabolites involves the spectroscopic techniques using NMR, MS, GCMS, UV-VIS, and classical wet chemical techniques. Troubleshoot and maintenance of analytical instruments as needed. Structure elucidation of novel natural products from Medicago Polymorpha by using NMR spectroscopy, LCMS, isolation and purification of compounds by using silica gel chromatography.
I have experience of teaching at university of Wah. I have taught different courses such as inorganic chemistry, biochemistry and general chemistry to BS 4th and BS 2nd. I have taught these courses in funny and creative manner. Several people think that chemistry is rough and tough subject but i want to say no its not like that because no subject becomes difficult when teacher makes it easy. All those who are facing difficulty in studying chemistry should come to me and see how i am making chemistry easy. thanks.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
(a) Describe the molecule xenon trioxide, XeO3, using four possible Lewis structures, one each with zero, one, two, or three Xe-O double bonds. (b) Do any of these resonance structures satisfy the...
-
The following three Lewis structures can be drawn for N2O: (a) Using formal charges, which of these three resonance forms is likely to be the most important? (b) The N-N bond length in N2O is 1.12...
-
The reaction of two equivalents of Mg with 1, 4-dibromobutane produces compound A. The reaction of A with two equivalents of CH3CHO (acetaldehyde). followed by work-up with dilu aqueous acid,...
-
Describe the typical terrorist cell.
-
Describe what happened during the 1970s to spur the growth in project management and compare and contrast this with the events that occurred during the 1990s and that are occurring today.
-
In Young's double-slit experiment, light intensity is a maximum when the two waves interfere constructively. This occurs when where \(d\) is the separations of the slits, \(\lambda\) the wavelength...
-
Refer to the information in Exercise 17-7 to answer the following requirements. Required 1. Using ABC, compute the overhead cost per unit for each product line. 2. Determine the total cost per unit...
-
Interest during Construction Grieg Landscaping began construction of a new plant on December 1, 2010. On this date the company purchased a parcel of land for $139,000 in cash. In addition, it paid...
-
Stage duration (hours) for immatures of 2 species of blow fly when reared at several temperatures; e.g., "time to complete" the stage. The threshold temperature for all species is 10C.--------1. What...
-
You plan to retire in exactly 20 years. Your goal is to create a fund that will allow you to receive $20,000 at the end of each year for the 30 years between retirement and death (a psychic told you...
-
Vanillin, whose structure is shown in the margin and is the subject of the Chapter Opening, is a benzene derivative with several functional groups, each one of which displays its characteristic...
-
The energy levels of the 2-propenyl (allyl) and cyclopropenyl p systems (see margin) are compared qualitatively in the diagram below. (a) Draw the three molecular orbitals of each system, using plus...
-
Marketers need to be extra sure their product works as promised when they first introduce it. How does this statement relate to what we know about consumers evoked sets?
-
Choose the below option: The timetable management set to complete our audit engagement was unreasonably short considering failure by the company's personnel to complete schedules on a timely basis...
-
"The company developed a Price Master File which captures the price in which inventory should be purchased from suppliers and the price is in which it should be sold to customers. Employees do not...
-
I require an example of how to develop two work systems for a cash based business. One for banking and another for bill handling. I am struggling to write the step bt step instructions required.
-
Solve the following problems about Power (P): 2. At what rate can a 1.5 hp motor raised a 100-lb. box?
-
Calculate the support reactions for this simply supported beam, a component of the bridge. Key Values A 15 kN B 20 KN C 3 kN/m X 1.5 m y 2.5 m R1 X A 1m B Y UDL = C R2 1m
-
Heron Company purchases commercial realty on November 13, 1996, for $650,000. Straight-line depreciation of $287,492 is claimed before the property is sold on February 22, 2014, for $850,000. What...
-
Prepare a stock card using the following information A company is registered for GST which it pays quarterly, assume GST was last paid on the 30th of June 2019. It uses weighted average cost...
-
Explain how X-ray diffraction can be used to determine the helical configuration of biological molecules.
-
Describe the characteristics of the Fermi-Dirac distribution. Why is it appropriate to call the parameter u. a chemical potential?
-
Equivalent lattice points within the unit cell of a Brava is lattice have identical surroundings. What points within a body-centred cubic unit cell are equivalent to the point (1/2, 0, 1/2)?
-
What are the key historical factors supporting the building of relationships between indigenous communities and settlers from Britain and France? What were the strengths and difficulties? What is an...
-
Let n be a positive integer, n2. The graph G illustrated below has vertices a1, a2, ..., an, b1, b2, ..., bn. C1, C2, ..., C, and d, and edges ab, bici, cia, and, bnd, cnd and aja+1, bibi+1, CiCi+1...
-
How should an organization deal with third-party network access?

Study smarter with the SolutionInn App


