Devise a synthesis for each of the following four compounds by using the malonic ester synthesis. (a)
Question:
Devise a synthesis for each of the following four compounds by using the malonic ester synthesis.
(a)
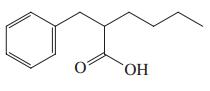
(b)
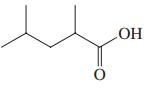
(c)
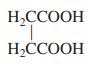
(d)

Transcribed Image Text:
HO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
a b c d u EtO OEt C...View the full answer

Answered By

KULDEEP MITTAL
CSIR UGC NET with AIR 46 and AIR 19
GATE 2020 with AIR 5
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Design a synthesis for each of the following compounds a. b. c. d. CCH3 CH3OCCH2CH2COCH3 CH2CH3 CH CH CHCOH CH30CCH2COCH,-
-
Propose a synthesis for each of the following compounds, using a Robinson annulations: a. b. c. d. CH CH3 CH3 CHs H-C
-
Devise a synthesis for each of the following, starting with benzene: a. M-bromobenzenesulfonic acid b. P-nitrotoluene
-
Apply the Western Electric rules to the control chart presented in Exercise 5.16. Would these rules result in any out-of-control signals?
-
"A country whose trade has almost no impact on world prices is at great risk of immiserizing growth." Do you agree or disagree? Why?
-
Experiment with both the size of your views as well as the confidence you assign to them to see how they affect portfolio allocation and adjusted return.
-
Washington Tennis & Education Foundation, Inc. (WTEF) is a nonprofit organization operating in the District of Columbia that provides athletic and academic programs for children from low-income...
-
1. Complete Problem 17-36 using the FIFO method of process costing. In Problem 17-36, McKnight Handcraft is a manufacturer of picture frames for large retailers. Every picture frame passes through...
-
3. (5 Marks) An engineering project requires $25,000 as first cost and has a planning horizon of six years. Operating costs are $2,300 per year, and expected annual revenue is $9,200 (both real...
-
An 80-kg man standing on a frozen lake tosses a 0.500-kg football to his dog. (a) If the ball leaves his hands at 15 m/s relative to Earth, what minimum source energy did the man supply? (b) His...
-
Devise a preparation of each of the following ketones by using the acetoacetic ester synthesis. (a) (b) (c) (d) OCH,CH3
-
Use the Michael Addition methods , with other reactions if necessary, to synthesize each of the following compounds. In each case, your starting materials should include one aldehyde or ketone and...
-
A wheel centered at the origin and of radius 10 centimeters is rotating counter-clock-wise at a rate of 4 revolutions per second. A point P on the rim of the wheel is at position (10.0) at time t =...
-
The Top Thrill Dragster travels at 120 mph and the steelrollercoaster mean speed is 61.67 mph. How many standard deviationsis the speed of the Top Thrill Dragster from the mean speed? 14.67standard...
-
XYZ Corporation has $200M in debt that yields 8%, its stock trades at $20/share and there are 20M shares outstanding. The book value of equity is $300M. XYZ's beta is 1.3. The risk free rate is 3.0%...
-
(a) Using a geometric series (and no other tricks), find a fraction that has the exact decimal expansion 0.13. Verify your answer on a calculator. (b) Using a geometric series (and no other tricks),...
-
An M&M candy dispenser has 25 red, 25 blue, and 25 greenM&Ms. The dispenser dispenses 20 M&Ms each time. What isthe probability, that on the first use, you will get at most 8 redM&Ms out of the 20...
-
Suppose f (x) = 3x^2 +1 and g (x) =2x-4 Find the value of f (g(1)).
-
In May, Rebecca's daughter, Susan, sustained a serious injury that made it impossible for her to continue living alone. Susan, who is a novelist, moved back into Rebecca's home after the accident....
-
An environmentalist wants to determine if the median amount of potassium (mg/L) in rainwater in Lincoln County, Nebraska, is different from that in the rainwater in Clarendon County, South Carolina....
-
Convert the following line-bond structures into molecularformulas: CH CH- (a) (b) . Hi H. c=C Vitamin C Aspirin (acetylsalicylic acid) (ascorbic acid) (c) H. - (d) H~ CH c-o H. . HOI I H H OH...
-
Convert the following molecular formulas into line-bond structures that are sonsistent with valence rules: (a) C3H8 (b) CH5N (c) C2H6O (2 possibilities) (d) C3H7Br (2 possibilities) (e) C2H4O (3...
-
What kind of hybridization do you expect for each carbon atom in the followingmolecules? (a) Propane, CH3CH2CH3 (b) 2-Methylpropene, CH CH3C=CH2 (c) 1-Butene 3 yne, H2C=CH-C=CH (d) Acetic acid, CHC
-
A manufacturer of consumer-packaged goods has hired your consulting (marketing analytics/research) firm to better understand and quantify consumers price sensitivity. What data would you need from...
-
The Online Cooking Community needs to redesign their website to enable registered users to rate recipes with a rating out of 5 . Develop a distributed web application that can be used as a recipe...
-
II) The total daily cost (in dollars) of producing x boxes of chocolate-dipped bars, at the Confections Candy Company, is given by C(x) = 100+ 18x 0.03x2 (10pts) 3. If the Company is to increase...

Study smarter with the SolutionInn App


