Give the product of each of the following reactions. (a) (b) (c) (d) (e) CH 1. ,
Question:
Give the product of each of the following reactions.
(a)
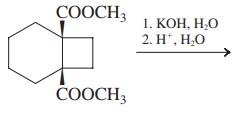
(b)
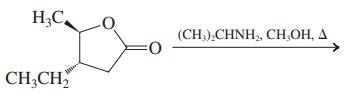
(c)
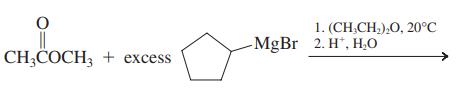
(d)
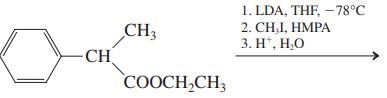
(e)
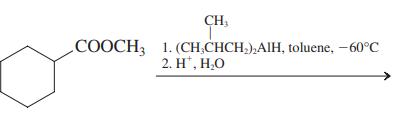
Transcribed Image Text:
СООCH 1. КОН, Н.О 2. H', Н.О СООCH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
a...View the full answer

Answered By

MANDWEEP BHUMIJ
I did my masters in Tezpur University with specialization in Organic chemistry. I cleared CSIR NET-JRF with all India ranking 74. I am currently working as Junior Research Fellowship in Central drug research institute Lucknow. I have some private tutoring experiences in multiple subjects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the product of each of the following reactions: (a) (b) (c) (d) CH3 CHCH3 Na2r207.H CH2CH3 Na,Cr0H CH3 1. NBS/A/peroxide 2. CH30 CH3 1. NBS/A /peroxide 3. H2/Ni
-
Give the product of each of the following reactions: a. b. c. d. OHH, . CHCHCH,N12 catalytic Zo outo 0 auda CH3CCI + 2 1. HCI, NaNO2 22. H20, Cu20, Cu(NO32 catalytic
-
Give the product of each of the following reactions: a. b. c. d. CH CH3 A CH2CH3 CH2CH 2CH3 -A CH2CH3 h CH2CH3 CH2CH3 CH2CHs
-
Your restaurant has five menu choices for lunch. How many ways can you order them on your menu? A 0.0749 B 0.0747 120
-
Consider the following thought experiment regarding foreign direct investment and specific factors. (a) In the pure specific-factors model in the text, suppose that a wave of foreign direct...
-
During the Launch and Understand phases, it became evident that major changes will occur for the staff. For example, there will be less focus on data entry and more on providing student service....
-
Do analytical efforts, big data examinations, and textual analyses impact compliance and fraud deterrence? Provide an explanation for your opinion.
-
Refer to Exhibit 2-11 on page 65. Required a. What is Universal Sports Exchange's operating profit equation? b. If Universal Sports Exchange sells 55,000 jerseys, what total expense will be reported...
-
If $300,000 is to be saved over 15 years, how much should be deposited monthly if the investment earns 7% interest compounded monthly?
-
Jimbo Kirtley & Brothers is a U.S. exporter of gym equipment and spandex-based athletic wear. The company's intrinsic business value (VBUSD) is USD 10,000. Assume Kirtley has a net debt (NDUSD) USD...
-
Reaction review. Suggest reagents to convert each of the following starting materials into the indicated product: (a) hexanoyl chloride into acetic hexanoic anhydride; (b) methyl hexanoate into...
-
For each of the naturally occurring lactones below, draw the structure of the compound that would result from hydrolysis using aqueous base. (a) (b) (c) Sedanenolide, major contributor to the flavor...
-
(a) In principle, do your eyes have greater resolution on a dark cloudy day or on a bright sunny day? (b) Choose the best explanation from among the following: I. Your eyes have greater resolution on...
-
A square loop with sides 1 2 cm by 1 2 cm has 2 5 turns of wire and is in a magnetic field of strength 4 . 2 x 1 0 ^ - 2 T with its axis perpendicular to the direction of the field. if it is rotated...
-
How do post-transcriptional and post-translational regulatory mechanisms, such as microRNA-mediated gene silencing and protein modification, contribute to the fine-tuning of developmental processes,...
-
Grandin Inc. is evaluating its dividend policy. It has a capital budget of $625,000, and it wants to maintain a target capital structure of 60% debt and 40% equity. The company forecasts a net income...
-
recording a lon term lease as an operating lease as opposed to a capital lease what will cause for a lease are the debt to equity will be higher than total asset turnover? Explain.
-
Consider a reaction that has a negative AH and a positive AS. Which of the following statements is true? This reaction will be nonspontaneous only at high temperatures. This reaction will be...
-
Ira Cook is planning to make a charitable contribution to the Boy Scouts of Crystal, Inc. stock worth $20,000. The stock has an adjusted basis of $15,000. A friend has suggested that Ira sell the...
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
Identify these pairs of compounds as identical, structural isomers, enantiomers, or diastereomers: CH3 CH3 H3 CH Br - - - - -Cl - b) Br- a) - - CI - - CH3 CH CH3 CH3 CH3 CH3 CH CI Br - - CH3...
-
(a) A solution of 0.2g/mL of a compound in a 1 dm cell rotates plane-polarized light + 13.3 o at the sodium D line. What is the specific rotation of this compound? (b) What is the rotation caused by...
-
Describe how this amine could be resolved by using this carboxylic acid? CH3 CNH, H HC H CI C COH
-
Explain for each of the following scenarios whether they are: Tax planning; Tax avoidance; or Tax evasion and why. Michael Brown is the sole director of Brown Pty Ltd. Brown Pty Ltd received...
-
Butler Lumber Company The file Butler - Spring'12.xls contains Exhibit 1 and Exhibit 2 from the case. As Mr. Butler's financial advisor, he has asked you to help him negotiate banking arrangements...
-
Give an explanation- and usean example - ofwhy we may need to convert an Account Receivable to a Note Receivable and why we need to keep track of the days of the notes.

Study smarter with the SolutionInn App


