Specify whether you expect the benzene rings in the following compounds to be activated or deactivated. NO2
Question:
Specify whether you expect the benzene rings in the following compounds to be activated or deactivated.
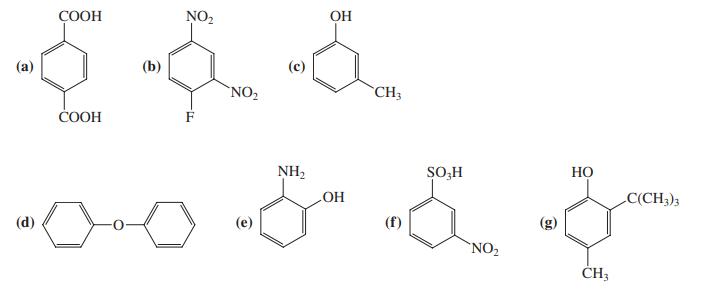
Transcribed Image Text:
СООН NO2 OH (a) (b) 'NO2 CH3 COOH NH2 SO-H Но HO C(CH3)3 (d) (e) (f) 'NO2 ČH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
a In compound a COOH group is deactivating Hence benzene ring is expect...View the full answer

Answered By

Aditi Deswal
Currently , I am doing post graduation ( MSc.) in Chemistry . I want to bacome a lecturer . I feel happy when I teach student and solve their problem . It is my passion as well as hobby . I feel blessed If I got a chance to share my knowledge as much as I have . I teach student at my home also. I love teching and want to spend my whole life to teach the students and explores new things.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Biphenyl is two benzene rings joined by a single bond. The site of substitution for a biphenyl is determined by (1) which phenyl ring is more activated (or less deactivated), and (2) which position...
-
Biochemical oxidation of aromatic rings is catalyzed by a group of liver enzymes called aryl hydroxylases. Part of this chemical process is the conversion of toxic aromatic hydrocarbons such as...
-
The following compound has a pentasubstituted benzene ring. (a) Starting with benzene and using any other necessary reagents of your choice, design a synthesis for this compound. (b) It is very...
-
What is meant by date alignment? Does it exist on the consolidated worksheet under the following methods, and if not, how is it created prior to elimination of the investment account under each of...
-
During a criminal trial for extortion, a psychiatrist testified regarding defendant. His testimony and opinion were based primarily on information and reports from other doctors. Defendant was...
-
Consider two bundles of coffee and chocolate and how Ted feels about them. The first bundle consists of two cups of coffee and two chocolate bars. The second bundle consists of one cup of coffee and...
-
1. Working in small groups, complete the Pay Raise Worksheet. 2. After the class reconvenes, group spokespersons present group findings. Pay Raise Worksheet April Knepper is the new supervisor of an...
-
The information that follows is from Jasons Hardware Companys June 30, 2014, post-closing trial balance. Required 1. Prepare a classified balance sheet for Jasons Hardware Company. 2. Compute Jasons...
-
Bleeker Street Pizza and Keste Pizza & Vino are located close to each other on Bleeker Street in the West Village. There are 450 potential customers every day, and suppose that each of them is...
-
The homogeneous circular cylinder of mass m and radius R carries a slender rod of mass m/2 attached to it as shown. If the cylinder rolls on the surface without slipping with a velocity v O of its...
-
The rate of nitration of (chloromethyl)benzene, shown below is 0.71 relative to the rate of nitration of benzene (=1). The (chloromethyl)nitrobenzene product mixture that results contains 32% ortho,...
-
Rank the compounds in each of the following groups in order of decreasing reactivity toward electrophilic aromatic substitution. Explain your answers. CH3 () (b) H; H3
-
Big Bend Co. fixed budget for the year is shown below: Prepare a flexible budget for Big Bend Co. that shows a detailed budget for its actual sales volume of 42,000 units. Use the contribution margin...
-
Shi Import-Export's balance sheet shows $400 million in debt, $60 million in preferred stock, and $260 million in total common equity. Shi's tax rate is 15%, rd = 9%, rps = 8.9%, and rs = 13%. If Shi...
-
A firm has sales of $1.3 million, and 15 percent of the sales are for cash. The year-end accounts receivable balance is $150,000. What is the average collection period? (Use a 360-day year. Do not...
-
If the 1-year rate of return is 20% and the interest rates are constant, what is the 5-year holding rate of return? Q 2-11 If you invest $2,000 today and it earns 25% per year, how much will you have...
-
Village Finance Co . advanced three loans to Kamiko $ 3 , 1 0 0 on June 9 , $ 2 , 7 0 0 on August 1 4 , and $ 2 , 2 0 0 on October 1 8 . Simple interest at 7 . 7 0 % was charged on all three loans,...
-
A company has issued preferred stock with an annual dividend of $2.42 that will be paid in perpetuity. The current price of the stock is $24.6. What is the expected rate of return on the preferred...
-
A law practice was incorporated on January 1, 2014, and expects to earn $25,000 per month before deducting the lawyer's salary. The lawyer owns 100% of the stock. The corporation and the lawyer both...
-
How does Kant answer Humes bundle theory of self? Do you think he is successful?
-
Determine the ratios of (a) The mean speeds, (b) The mean kinetic energies of His atoms and Hg atoms at 25e.
-
The best laboratory vacuum pump can generate a vacuum of about 1 n Torr. At 25C and assuming that air consists of N, molecules with a collision diameter of395 pm, calculate (a) The mean speed of the...
-
At what pressure does the mean free path of argon at 25C become comparable to the diameters of the atoms themselves?
-
A business provides services valued at 1 1 0 0 and sends an invoice to the client. The client has promised to pay in one month. which two accounts will be affected?
-
Rushing's financial statements were finalized on March 3, 2025. Required: 1. What amount(s) if any, related to the situations described should Rushing report among current liabilities in its balance...
-
Monroe Company produces and sells three products. Information regarding these three products for 2024, the most recent year, is provided below: selling price per unit Product #1 $26 variable costs...

Study smarter with the SolutionInn App


