Two of the following four compounds are more acidic than CH 3 OH (i.e., two of these
Question:
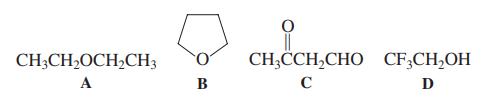
Two of the following four compounds are more acidic than CH3OH (i.e., two of these have Ka greater than methanol). Which ones?
(a) A and B;
(b) B and C;
(c) C and D;
(d) D and A;
(e) D and B.
Transcribed Image Text:
CH;CH,OCH,CH3 CH;CCH,CHO CF;CH,OH A B C D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
c C O are more acidic than CH 3 OH Method i Acidity depends upon the s...View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Two items are omitted in each of the following four lists of income statement data. Determine the amounts of the missing items, identifying them byletter. Sales Sales returns and allowances Sales...
-
Two items are omitted in each of the following four lists of income statement data. Determine the amounts of the missing items, identifying them byletter. igl Sas0,000 Sales Seles returns and...
-
Two items are omitted in each of the following four lists of income statement data. Determine the amounts of the missing items, identifying them byletter. Sales Sales returns and allowances Sales...
-
Upland Co.'s inventory records showed the following data accounted for in a perpetual inventory system. Date Units Unit Cost June 1 Inventory 1,250 $8.00 June 3 Purchases 2,500 8.40 June 7 Sales (at...
-
Consider another Ricardian example, using standard Ricardian assumptions: Vintland has 30 million hours of labor in total per year. Moonited Republic has 20 million hours of labor per year. a. Which...
-
List and quantify some of the benefi ts that could be derived from the acquisition of a clinical IT system. If possible, estimate the costs of these systems, and develop a return on investment (ROI),...
-
Steven Sanchez worked as a warehouse employee for Gruma Corporation. In December 2016, Sanchez was presented an arbitration agreement during a meeting with George Tate, the human resources manager...
-
Sharon Feldman, president of Allied Products, considers $20,000 to be a minimum cash balance for operating purposes. As can be seen from the following statements, only $15,000 in cash was available...
-
3. (L)Explain the four terms in the equation below, and why this equation has to be true by definition. Explain the assumption that allows us to use this equation to determine the agricultural...
-
McMullen Company purchased tool sharpening equipment on May 1 for $162,000. The equipment was expected to have a useful life of three years, or 12,000 operating hours, and a residual value of $3,600....
-
Split your team in two, each group to analyze one of the following reaction sequences by a mechanism ( 13 C 5 carbon-13 isotope).
-
Two of the following four compounds are more acidic than CH 3 OH (i.e., two of these have K a greater than methanol). Which ones? (a) A and B; (b) B and C; (c) C and D; (d) D and A; (e) D and B....
-
Although not all students have debt after graduating from college, more than half do. The College Boards 2008 Trends in Student Aid addresses, among other topics, the difference in the average...
-
10:00 Module name: Taxation Planning 2A Module code: TXP02A2 Year: 2022 Semester: First Assessment: Continuous Assessment 3 Release date: 1 April 2022 Submission deadline: 22 April 2022 INFORMATION:...
-
1. What could be the reasons for the price increase in offline retail stores? 2. From the information given in the case, can one conclude that there was a differential price response from the online...
-
On July 1, a company sells inventory on account to a customer for $40,000, with terms of 1/15, n/30. The company uses the net method to account for sales discounts. The customer pays the amount due...
-
What is one way that Netflix has changed the film/television industry? Can social media (Facebook, Twitter, Instagram, etc.) affect what movie/TV/song we might watch or listen to? Why or why not?...
-
A firm's end-of-year free cash flow is anticipated to be $14 million. The company's free cash flows are expected to grow 15 percent a year forever. The firm's weighted average cost of capital is 18...
-
Your client, a large construction firm organized as a C corporation, allows certain employees (including the president of the corporation) to use its company owned airplane for non-business flights....
-
Starr Co. had sales revenue of $540,000 in 2014. Other items recorded during the year were: Cost of goods sold ..................................................... $330,000 Salaries and wages...
-
Which of the following pairs of structures represent resonanceforms? (b) :0: (a) :0: and and (d) :: (c) :0: :0: and and
-
Draw as many resonance structures as you can for the followingspecies: (b) :NH2 H2N-C=NH2 (c) (a) :0: H --- () HC $H2 (e) H %H
-
Cyclobutadiene is a rectangular molecule with two shorter double bonds and two longer single bonds. Why do the following structures not represent resonance forms?
-
An investment that pays you $3,500, five years from now, is worth $2,495 today, if the discount rate is 7%. If the discount were 4%, what would you be willing to pay for this investment?
-
Using Ampol Australia's Dividend, Prices and Share Valuation excel sheets provided and: 1. Calculate percentage returns per month 2. Estimate beta using the monthly returns 3. Look up the risk-free...
-
letter to your former 12-year old self. Address each of the 4 Quadrants of the Johari Window by writing about the advice you would give to your former self. Consider the following questions in your...

Study smarter with the SolutionInn App


