Split your team in two, each group to analyze one of the following reaction sequences by a
Question:
Split your team in two, each group to analyze one of the following reaction sequences by a mechanism (13C 5 carbon-13 isotope).
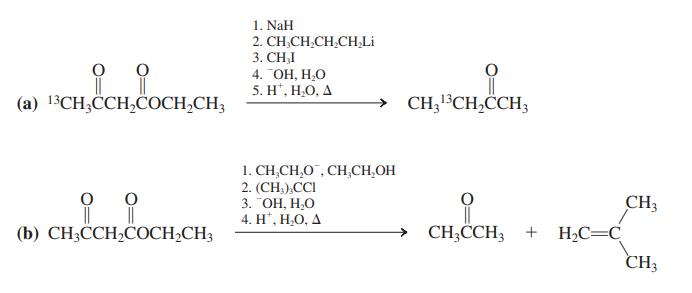
Reconvene and discuss your results. Specifi cally, address the position of the 13C label in the product of (a) and the failure to obtain alkylation in (b).
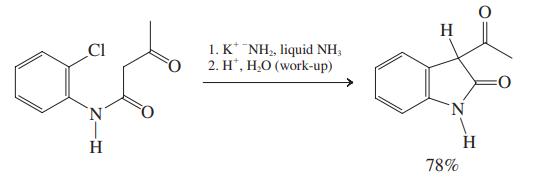
As a complete team, also discuss the mechanism of the following transformation.
Transcribed Image Text:
1. NaH 2. CH,CH,CH,CH,Li 3. СHI 4. ОН, Н.О 5. H", Н.О, Д (а) 13CH,ССH,CОСH-CH CH;13CH,CCH3 1. CH,CH,O , CH,CH,OH 2. (CH,),CCI 3. "ОН, Н.О 4. H", Н.О, А CH3 → CH;CCH3 + H2C=C CH3 (b) СH,ССH,CОCH-CH;
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The reaction sequence involves the conversion of CH3CH2CH2CH2Li 2 into CH3CH2CH2CH2OH 4 The steps in...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Your task is to prepare isopropyl methyl ether by one of the following reactions. Which reaction would give the better yield? Explain your answer. (1) Isopropyl methyl ether Or (2) Isopropyl methyl...
-
One way to prepare esters is by the reaction of an alcohol with an acid anhydride (as in this experiment). However, they also may be prepared by the reaction between an alcohol and a carboxylic acid...
-
This problem introduces two literature syntheses of indole derivatives, and you are asked to come up with plausible mechanisms for them. Divide your team in two, each group concentrating on one of...
-
What effect does a weakening in U.S dollar have on Net Income for subsidiaries of U.S companies?
-
In your answer to this question, use a diagram and start from a no-trade point like S0 with a no-trade price ratio of 2 W/C. Now trade is opened and the country can trade whatever it wants at an...
-
Discuss the pros and cons of reporting contractual adjustments directly on the monthly fi nancial reports at the department level.
-
Christopher Boling was seriously injured in 2008 when vapors escaping from a gas can ignited. He filed a products liability claim against the manufacturer. To fund the litigation, Boling entered into...
-
Multiple Choice Questions The following items apply to determining sample sizes using random sampling from large populations for attributes sampling. Select the most appropriate response for each...
-
In its simplest form, profit is: Fixed costs - Depreciation - Interest = Profit Sales/Turnover - Variable costs/Used goods = Profit (Actual retail price - cost)/ Actual retail price = Profit (Price -...
-
The cash book (bank columns) of Ella for the month of May 207 were as follows: Ellas bank statement for May 207 showed the following: The following errors were discovered: 1. Ella had overcast the...
-
Propose a synthesis of ketone C, which was central in attempts to synthesize several antitumor agents. Start with aldehyde A, lactone B, and anything else you need. H,C H2C A B C
-
Two of the following four compounds are more acidic than CH 3 OH (i.e., two of these have K a greater than methanol). Which ones? (a) A and B; (b) B and C; (c) C and D; (d) D and A; (e) D and B....
-
Calculate the value for each of Exercises. 0!
-
How can acting unprofessionally in a business social situation (for example the annual company picnic or holiday party) hurt your career? Share some examples of inappropriate behavior at business...
-
What would be the appropriate adjusting journal entries: The notes payable balance relates to a bank loan obtained in 2018 that is payable in full on august 31,2023. the loan agreement specifies that...
-
1:Explain your understanding of why it is important to be persuasive in negotiation and how you would go about being persuasive in a negotiation. 2: Research a conflict or dispute that has occurred...
-
What is going on with your site? I have searched for examples for the following papers and could not find any info on them. They are: 1. Create Visual Communication 2. Comm Regulation and Policy 3....
-
One of your staff members has been asked to give two speeches. Details are as follows: first speech - large target audience; objective: to inform them of company progress over the past year; and ...
-
What are the guidelines regarding the deductibility of luxury water travel (e.g., cruise ship) for business purposes? Refer to Chapter 1 of IRS Publication 463.
-
How can NAFTA be beneficial to suppliers of Walmart?
-
Fluoromethane (CH3F, = 1.81 D) has a smaller dipole moment than chloromethane (CH3C1, = 1.87 D) even though fluorine is more electronegative then chlorine. Explain
-
Methanethiol, CH2SH, has a substantial dipole moment ( = 1.52) even though carbon and sulfur have identical electro negativities. Explain.
-
Calculate the formal charges on the atoms shown inred. (a) (CH3)2OBF3 (b) H2C-NEN: (c) H2C=N=N: (d) :=-: (e) H (f) - CH
-
Balance at beginning of the year: $ 52 comma 000 bullet Purchased goodwill during the year for $ 18 comma 500. bullet Sold various intangible properties included in class 14.1 for $ 13 comma 000...
-
Doobey, Incorporated, has just formed a new division, and the following four investment opportunities are available to the division. The firm requires a minimum return of 8 percent. Investment...
-
If I want to buy a $500,000 and can get a 30-year mortgage with an APR = 3.5%, what would my monthly payments be? If I can get a 15 year mortgage for 2.5%, how much would I save? I've checked with my...

Study smarter with the SolutionInn App


