Ignoring small electron binding energies and the very small mass of the neutrino, show that the mass
Question:
Ignoring small electron binding energies and the very small mass of the neutrino, show that the mass of a nucleus increases when it decays by electron capture if the Q-value of the decay is less than mec2 ? 0.511 MeV. Verify that this is the case for the electron capture decay of the longest-lived isotope of technetium:
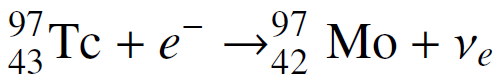
Transcribed Image Text:
97 Тс + e 43 Tc + e¯ →% 97 42 Мо + Ve
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
When a nucleus A Z decays to A1 Z 1 by electron captureenergy conservation gives where we have ig...View the full answer

Answered By

Talha Talib
I am a member of IEEE society. As i am a student of electrical engineering badge 17 but beside of this i am also a tutor in unique academy. I teach calculus, communication skills, mechanics and economics. I am also a home tutor. My student Muhammad Salman Alvi is a brilliant A-level student and he performs very well in academics when i start to teach him. His weak point was mathematics but now he is performing well in mathematics. I am a scholarship holder in Fsc as i scored 1017 marks in metric out of 1100. Later on i got scholarship in Punjab Group of Colleges. I got 2nd position in robotics competition in 2018 as my project home automation select for the exhibition in Expocentre.
4.60+
23+ Reviews
62+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
Electron-donating groups on benzene promote electrophilic aromatic substitution and lead preferentially to so-called ortho and para products over meta products, whereas electron-withdrawing groups...
-
The binding energies of K-shell and L-shell electrons in copper are 8.979 and 0.951 keV, respectively. If a Ka x ray from copper is incident on a sodium chloride crystal and gives a first-order Bragg...
-
The binding energy of a valence electron in a Li atom in the states 2S and 2P is equal to 5.39 and 3.54 eV respectively. Find the Rydberg corrections for S and P terms of the atom.
-
______________ is an approach to doing business that attempts to maximize an organization's competitiveness through the continual improvement of the quality of its products, services, people,...
-
You step onto a hot beach with your bare feet. A nerve impulse, generated in your foot, travels through your nervous system at an average speed of 110 m/s. How much time does it take for the impulse,...
-
Use the Gauss-Seidel method to solve the following system of equations. 10x12x2 x3 = 27 - - -3x1 6x2 + 2x3 = -61.5 x1 + x2 +5x3 = -21.5 Compute the approximate relative percent error for each...
-
What is the payback period for the project described in Problem 4.21 if the maintenance increases by $\$ 500 /$ year (after the first year)? Data from in problem 4.21 What is the payback period for...
-
a. Calculate the centroids for each group in the analysis sample. What do these values indicate? b. Use the regression approach to two-group DA to develop a rule for predicting whether or not a bank...
-
Your company is excited with the information discussed regarding the cloud technologies that are available to Get-Sole Shoes. Now the management team wants to examine what its cloud infrastructure...
-
Exclusive-OR (XOR) is one of the most used operations in the calculation of codewords. Apply the exclusive-OR operation on the following pairs of patterns. Interpret the results. a. (10001) (10001)...
-
Verify the expressions for the Q-value in ? ? -decay and electron capture, eq. (17.34)? Q(B) = A(Z, A) A(Z + 1, A), Q(B+) = A(Z, A) A(Z 1, A) 2Ae Q(EC) = A(Z, A) - A(Z 1, A),
-
97% of naturally occurring calcium is calcium-40, 40 20 Ca. This may seem surprising, since if we use the semi-empirical mass formula to estimate the most stable nuclide with A = 40 we find Z 18....
-
What are the types of opinions and the conclusion of each type of opinion?
-
Busaba Domitila (Domi) invented a new digital camera and data-storage system that is smaller than a grain of rice. She has named it The Camerice. She designed a unique typeface and logo for all...
-
Please give a real-life example of a civil claim(Pleading) that contains the proper name(Legal Name) of the two parties and the amount of the claim.
-
Perform the following actions to complete the accounting cycle for the first month of operations at Mother Molly's Childcare. Record adjusting journal entries for these items: A physical count of...
-
What is a tort? Choose one intentional tort against the person (other than battery) and describe briefly how it might occur.
-
Should a court recognize a common-law duty of social hosts to prevent drunken guests from getting on the road?
-
Which GAAP states that recorded financial transactions must have a confirmable (provable) basis in fact? a. Objectivity Principle b. Conservatism Principle c. Matching Principle d. Consistency...
-
Identify Thank You mission, strategy and core competencies. Identify strategy changes that have taken place at Thank You since its founding in 2008. Your answer must in text references and must be...
-
(a) Calculate the density of the atmosphere at the surface of Mars (where the pressure is 650 Pa and the temperature is typically 253 K, with a CO 2 atmosphere), Venus (with an average temperature of...
-
A large cylindrical tank contains 0.750 m 3 of nitrogen gas at 27 o C and 7.50 10 3 (absolute pressure). The tank has a tight-fitting piston that allows the volume to be changed. What will be the...
-
Digesting fat produces 9.3 food calories per gram of fat, and typically 80% of this energy goes to heat when metabolized. (One food calorie is 1000 calories and therefore equals 4186 J.) The body...
-
respond to the following : Hi Kassim, "Given that you have successfully applied these steps to pay down your car loan faster, how would you adapt this financial plan if you were to encounter a...
-
The Impact of Artificial Intelligence in the Accounting Profession: Challenges and Opportunities: The accounting profession has been undergoing significant transformations with the advent of...
-
The following is an alphabetical list of accounts and balances for J. Robinson, proprietorship at June 30, 2024 Accounts Payable $13,300 Notes Payable $31, 800, Accounts Receivable $10, 850 Prepaid...

Study smarter with the SolutionInn App


