A few drops of each of the indicators shown in the accompanying table were placed in separate
Question:
A few drops of each of the indicators shown in the accompanying table were placed in separate portions of a 1.0-M solution of a weak acid, HX. The results are shown in the last column of the table. What is the approximate pH of the solution containing HX? Calculate the approximate value of Ka for HX.
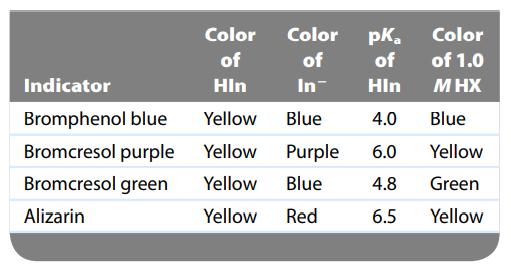
Transcribed Image Text:
Color Color pka of of of Indicator Hin In Hin Bromphenol blue Yellow Blue 4.0 Bromcresol purple Yellow Purple 6.0 Bromcresol green Yellow Blue 4.8 Alizarin Yellow Red 6.5 Color of 1.0 M HX Blue Yellow Green Yellow
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Solution a The approximate pH of the solution contai...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The data shown in the accompanying table were obtained from a tensile test of high-strength steel. The test specimen had a diameter of 0.505 in. and a gage length of 2.00 in. (see figure for Prob....
-
The observations shown in the accompanying table were drawn from a randomized block design with no interaction. a. Calculate SST, SSA, SSB, and SSE. b. Calculate MSA, MSB, and MSE. c. Construct an...
-
A 0.01 M solution of a weak acid in water is 0.05% ionized at 25C. What is its pK?
-
A steam generator consists of a bank of stainless steel (k = 15 W/m K) tubes having the core configuration of figure and an inner diameter of 13.8 mm. The tubes are installed in a plenum whose...
-
Two different companies are considering rights offerings. The current market price per share is $48 in both cases. To allow for fluctuations in market price, company X wants to set a subscription...
-
Calculate the expectation values of p and p2 for a particle in the state 11= 2 in a square-well potential.
-
Event A: Randomly select a U.S. citizen of Indian origin.. Event B: Randomly select a U.S. citizen of Chinese origin. Determine whether the events are mutually exclusive. Explain your reasoning.
-
Three years after receiving their degrees, graduates of a universitys MBA program have reported their annual salary rates, with a portion of the data listed in file XR11097. Graduates in one of the...
-
In Thunder Bay, a public survey was done that suggests that people are generally supportive of using automated, AI-based video analysis software [1]. Yet, elsewhere in Canada, such software has come...
-
In BE2-2, Caterpillar's current assets consisted primarily of cash and short-term investments of $8.3 billion, accounts receivable of $17.9 billion, inventory of $11.1 billion, and miscellaneous...
-
A 10.00-g sample of the ionic compound NaA, where A - is the anion of a weak acid, was dissolved in enough water to make 100.0 mL of solution and was then titrated with 0.100 M HCl. After 500.0 mL...
-
A buffer is made using 45.0 mL of 0.750 M HC 3 H 5 O 2 (K a = 1.3 10 -5 ) and 55.0 mL of 0.700 M NaC 3 H 5 O 2 . What volume of 0.10 M NaOH must be added to change the pH of the original buffer...
-
An individual and a group applied to the Chicago Park District for permits to hold rallies advocating the legalization of marijuana. The Park District, a municipal agency, required a permit to...
-
Cash is debited, and Notes Payable ( a liability account ) is credited. Which transaction A . Borrowing money Buying equipment by signing a note ( a liability ) Receiving money from stockholders...
-
Layton Corp. has a $4,000 par value bond outstanding with a coupon rate of 4.1 percent paid semiannually and 15 years to maturity. The yield to maturity of the bond is 4.3 percent. What is the dollar...
-
Jason and his parents reside in Florida and have a pre-paid college plan. They are researching college options for Jason. What would keep education costs the lowest? A money market account A state...
-
Questions (inclusive) are structured essay questions. Please USE the full ILAC method when answering questions . Make sure cite relevant sections and sub-sections of legislation and/or case law to...
-
On the sheet called Income Statement, in cell E14 create the formula for the Income Tax. On the sheet called Income Statement, make cell C14 is the only cell that the enduser can type in. Protect the...
-
What are three alternative approaches to measuring VAR and their benefits?
-
One study found that the elderly who do not have children dissave at about the same rate as the elderly who do have children. What might this finding imply about the reason the elderly do not dissave...
-
How might you replace a halogen substituent by a deuterium atom if you wanted to prepare a deuteratedcompound? Br , CHH-H CHCH2CH
-
How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase. (a) "CH (b) HH2CH2CHBr CH3CH2CH2CH2CH2CH2CH2CH3 (c)...
-
Rank each of the following series of compounds in order of increasing oxidationlevel: CI (a) (b) CH3CN CH3CH2NH2 H2NCH2CH2NH2
-
How may price floors and ceilings destroy market equilibrium? How does the shape of the indicator line on the graph show diminishing returns? Monthly rent A Price Ceiling on Rent B $900 $800...
-
Provide a comprehensive analysis of the regulatory frameworks governing financial institutions, delineating the intricate interplay between prudential supervision, market conduct standards, and...
-
How does the forum meet organisational objectives? What vocabulary, tone, structure and style suits the audience? How will I: build trust develop positive working relationships ...

Study smarter with the SolutionInn App


