A Nb-W alloy held at 2800C is partly liquid and partly solid. (a) If possible, determine the
Question:
A Nb-W alloy held at 2800°C is partly liquid and partly solid.
(a) If possible, determine the composition of each phase in the alloy, and
(b) If possible, determine the amount of each phase in the alloy.
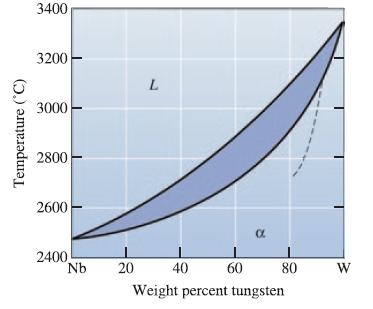
Transcribed Image Text:
Temperature (°C) 3400 3200 3000 2800 2600 2400 Nb L 20 α 40 60 Weight percent tungsten 80 W
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 28% (7 reviews)
a The composition of each phase in the al...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the eutectoid temperature, the composition of each phase in the eutectoid reaction, and the amount of each phase present in the eutectoid microconstituent for the following systems. For the...
-
Determine the composition of the products of the disassociation reaction CO2 CO + O when the products are at 1 atm and 2500 K.
-
A Nb-60 wt% W alloy is heated to 2800 C. Determine (a) The composition of the solid and liquid phases in both wt% and at%; (b) The amount of each phase in both wt% and at%; and (c) Assuming that the...
-
k) Assume that one of these portfolio's is the Market Portfolio and all portfolios, except Portfolio G, are fairly priced according to the CAPM. What is the highest utility score that can be achieved...
-
Starting from your graphical results of Problem 7b, show graphically the effect on YE and on XM of a depreciation of the nations currency from a position of full employment and a trade deficit.
-
Joy's Bar and Grill completed the following note-payable transactions: Answer these questions for Joy's Bar and Grill: 1. How much interest expense must be accrued at December 31, 2020? 2. Determine...
-
Refer to Exercise 9. For the result that was statistically significant, the P-value was 0.01. What P-value would be needed to conclude at the = 0.05 level that the time spent studying differs...
-
Home Town Supply does not segregate sales and sales taxes at the time of sale. The register total for March 16 is $10,388. All sales are subject to a 6% sales tax. Compute sales taxes payable and...
-
Machine A costs $800, requires annual maintenance of $150, and will last for 10 years. Machine B costs $600, requires annual maintenance of $200, and will last for 5 years. What is the calculated...
-
Write a statement that creates the concatenation of the three strings namel, name 2, and name 3, separated by commas, and assigns the result to a variable named roster. So if name 1, name2, and name...
-
Determine the phases present, the compositions of each phase, and the amount of each phase in mol% for the following NiO-MgO ceramics at 2400C: (a) NiO- 30 mol% MgO; (b) NiO-45 mol% MgO; (c) NiO-60...
-
A Bi-60 at% Sb alloy is cooled under equilibrium conditions from the liquid state. Determine (a) The temperature at which the first solid nucleus forms and its composition; (b) The compositions and...
-
A study was made of frequent fliers, and it was found that 53% had an income over $65,000 a year. a. Fifteen frequent fliers were selected at random, and their incomes recorded. What is the...
-
An astronaut who recently landed on an unfamiliar planet wants to measure the acceleration due to gravity. He constructs a simple pendulum of length \(40.0 \mathrm{~cm}\) and then finds that the...
-
TSMC Corporation is considering selling one of its old wafer fabrication machines. The machine, purchased for \($3,000,000\) 5 years ago, had an expected life of 10 years and an expected salvage...
-
Two vertical springs, one with spring constant \(k\) and the other with spring constant \(2 k\), each have a ball of mass \(m\) hanging from them. Compare the period of oscillation of the two springs...
-
At present, Global Skateboards Limited is considering expanding its product line to include solar powered skateboards; however, it is questionable how well they will be received by skateboarders....
-
Decathlon Stores is expanding operations with the introduction of a new distribution center. Not only will sales increase but investment in inventory will decline due to increased efficiencies in...
-
The Bombay Company, Inc., sold a line of home furnishings that included furniture, wall decor, and decorative accessories. Bombay operated through a network of retail locations throughout the United...
-
1. Advertising for eyeglasses _________ (increases/decreases) the price of eyeglasses because advertising promotes _________. 2. An advertisement that succeeds in getting consumers to try the product...
-
The codons (words) in DNA (that specify which amino acid should be at a particular point in a protein) are three bases long. How many such three-letter words can be made from the four bases adenine,...
-
Part of a certain DNA sequence is GGTCTATAC. What is the complementary sequence?
-
Which of the noncyclic isomers of C 4 H 7 F are optically active?
-
The recent collapse of two major banks, Silicon Valley Bank and Signature Bank, and the subsequent federal intervention to protect uninsured depositors, has reignited debates about government...
-
Privatizing Social Security in the United States has been a topic of debate for many years, with strong opinions on both sides since its creation as part of President Franklin D. Roosevelt's New...
-
Economists measure the price level by using a basket of goods and services and calculating how the total cost of buying that basket of goods will increase over time. Economists often express the...

Study smarter with the SolutionInn App


